 Back
BackPericyclic Reactions and Aromaticity: Study Guide for CHEM 2425 Module 08
Study Guide - Smart Notes

Pericyclic Reactions
Cycloaddition Reactions
Cycloaddition reactions are a class of pericyclic reactions where two or more unsaturated molecules (or parts of the same molecule) combine to form a cyclic product. The most common example is the Diels-Alder reaction, a [4+2] cycloaddition. - Definition: Cycloaddition involves the joining of π systems to form rings. - Thermal vs Photochemical Conditions: The outcome and stereochemistry of cycloadditions depend on whether the reaction is performed under heat or light. - Suprafacial and Antarafacial Interactions: These terms describe how the orbitals interact during the reaction. Suprafacial means the interaction occurs on the same face of the π system, while antarafacial means it occurs on opposite faces.
Electrocyclic Reactions
Electrocyclic reactions involve the conversion of a π system into a σ bond (or vice versa) through a cyclic movement of electrons. - Definition: Electrocyclic reactions are pericyclic processes where a ring closure or opening occurs via electron movement. - Thermal vs Photochemical: The stereochemistry of the product depends on the conditions. Under thermal conditions, the reaction follows the Woodward-Hoffmann rules for conrotatory or disrotatory motion.
Sigmatropic Rearrangements
Sigmatropic rearrangements are pericyclic reactions where a σ bond migrates across a π system. - Definition: A sigmatropic rearrangement involves the shift of a σ bond adjacent to one or more π systems, resulting in a new σ bond and a rearranged π system. - Classification: These are classified by the number of atoms involved in the migration, e.g., [1,5]- or [3,3]-sigmatropic shifts.
Aromaticity
Discovery and Structure of Benzene
Benzene is the prototypical aromatic compound, known for its exceptional stability due to delocalized π electrons. - Definition: Aromatic compounds are cyclic, planar molecules with a ring of resonance-stabilized π electrons. - Stability: Benzene's stability arises from its fully conjugated π system and adherence to Hückel's rule.
Criteria for Aromaticity
Aromaticity is determined by several criteria: - The molecule must be cyclic. - The molecule must be planar. - The molecule must be fully conjugated (every atom in the ring must have a p orbital). - The molecule must have π electrons (Hückel's rule), where n is a non-negative integer. 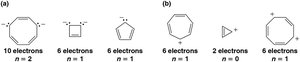
Antiaromaticity and Nonaromaticity
- Antiaromatic: Cyclic, planar, fully conjugated molecules with π electrons are antiaromatic and destabilized. - Nonaromatic: Molecules that do not meet the criteria for aromaticity or antiaromaticity.
Molecular Orbital Theory of Aromaticity
Molecular orbital (MO) diagrams help explain the stability of aromatic compounds. - Frost Circles: Used to visualize the energy levels of π molecular orbitals in cyclic systems. - Electron Filling: Aromatic compounds fill all bonding MOs, while antiaromatic compounds have unpaired electrons in non-bonding or antibonding MOs. 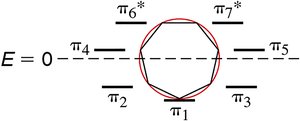
Nomenclature of Benzene Derivatives
IUPAC Naming
Benzene derivatives are named by identifying the substituents and their positions on the ring. - Example: 1,2-diiodobenzene 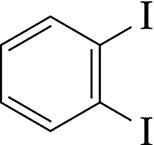 - Example: 3-chlorobenzoic acid
- Example: 3-chlorobenzoic acid 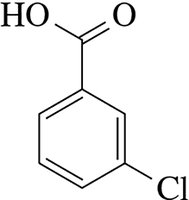
Ortho, Meta, Para Convention
The o/m/p system is used for disubstituted benzenes: - Ortho (o-): Substituents at positions 1 and 2 - Meta (m-): Substituents at positions 1 and 3 - Para (p-): Substituents at positions 1 and 4 
Classification of Aromatic Compounds
Fused Aromatic Rings
Polycyclic aromatic hydrocarbons (PAHs) consist of fused benzene rings. - Examples: Naphthalene, phenanthrene, anthracene 
Heteroaromatic Compounds
Compounds like imidazole contain heteroatoms (e.g., nitrogen) in the aromatic ring. - Basicity: The basicity of nitrogen atoms in heteroaromatic rings depends on their involvement in the aromatic π system. 
Effects of Aromaticity on Reactivity
Acidity and Basicity
Aromaticity can enhance or reduce the acidity/basicity of compounds. - Example: Aromatic carboxylic acids are often more acidic due to resonance stabilization.
Reaction Rates
Aromaticity affects the rates of reactions such as SN1, where carbocation stability is enhanced by aromatic systems.
Practice Problems and Applications
Classifying Aromatic, Antiaromatic, and Nonaromatic Compounds
- Example: Cyclopentadienyl cation is antiaromatic due to 4 π electrons. 
Frost Circles for Aromatic and Antiaromatic Systems
- Example: Frost diagrams illustrate electron filling in cyclic π systems. 
Comparing Nucleophilicity in Heterocycles
- Example: Pyrrole vs. pyrrolidine: Pyrrolidine is more nucleophilic because its nitrogen is not involved in aromaticity. 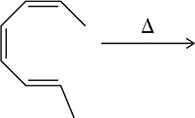
Polycyclic Aromatic Hydrocarbons
- Example: Chrysenes and pyrenes are larger PAHs with multiple fused rings. 
Summary Table: Aromaticity Classification
Compound | π Electrons | Classification |
|---|---|---|
Benzene | 6 | Aromatic |
Cyclobutadiene | 4 | Antiaromatic |
Cyclooctatetraene | 8 | Nonaromatic |
Cyclopentadienyl cation | 4 | Antiaromatic |
Naphthalene | 10 | Aromatic |
Phenanthrene | 14 | Aromatic |
Imidazole | 6 | Heteroaromatic |
Key Equations
Hückel's Rule
π electrons (where n = 0, 1, 2, ...)
Frost Circle Construction
- Place the polygon with one vertex pointing down. - Draw energy levels at each vertex. - Fill electrons according to the number of π electrons.
Conclusion
This module covers the advanced concepts of pericyclic reactions and aromaticity, including the prediction of products, classification of compounds, and the effects of aromaticity on chemical properties. Mastery of these topics is essential for understanding organic reaction mechanisms and the stability of aromatic systems.
