 Back
BackPredicting Products of Alkene Reactions in Organic Chemistry
Study Guide - Smart Notes

Q1. Predict the product(s) of the following reactions. Show proper stereochemistry whenever possible.
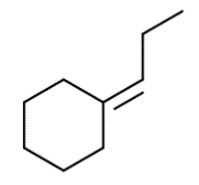
Background
Topic: Alkene Addition Reactions
This question tests your understanding of how alkenes react with various reagents to form new products, including regioselectivity and stereochemistry. You are expected to predict the outcome of reactions such as halogenation, hydrohalogenation, hydration, and others, based on the structure of the starting alkene.
Key Terms and Formulas
Alkene: A hydrocarbon containing a carbon-carbon double bond.
Markovnikov's Rule: In addition reactions, the electrophile adds to the carbon with more hydrogens.
Anti/ Syn Addition: Refers to the stereochemistry of the addition (opposite sides or same side).
Common Reagents: Br2, H2O, H2SO4, mCPBA, 9-BBN/THF, O3, (CH3)2S, HBr, CH2Cl2
Step-by-Step Guidance
Identify the functional group present in the starting material (in this case, an alkene attached to a cyclohexane ring).
Determine the reagent(s) used in the reaction (refer to your worksheet for specific reagents for each part).
Recall the mechanism for the reaction with the given reagent. For example, Br2 leads to anti addition of bromine across the double bond, while HBr follows Markovnikov's rule.
Draw the product(s) with correct connectivity and stereochemistry. Consider whether the addition is syn or anti, and if multiple stereoisomers are possible.
Check for regioselectivity (which carbon gets which group) and stereoselectivity (cis/trans or R/S configuration).
Try solving on your own before revealing the answer!
