 Back
BackReactions of Aromatic Compounds: Electrophilic Aromatic Substitution and Related Mechanisms
Study Guide - Smart Notes

Reactions of Aromatic Compounds
Electrophilic Aromatic Substitution (EAS)
Electrophilic aromatic substitution is a fundamental class of reactions in which an atom, usually hydrogen, attached to an aromatic ring is replaced by an electrophile. This mechanism preserves the aromaticity of the ring, making it distinct from addition reactions seen in alkenes.
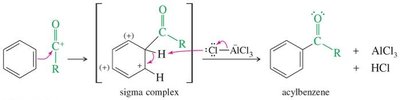
General Mechanism: The reaction proceeds via two main steps: (1) slow addition of the electrophile to the aromatic ring to form a resonance-stabilized carbocation intermediate (arenium ion), and (2) rapid deprotonation to restore aromaticity.
Key Intermediate: The arenium ion (also called σ-complex or cyclohexadienyl cation) is resonance-stabilized but less stable than benzene itself.

Halogenation of Benzene
Halogenation is a classic example of EAS, where a halogen atom replaces a hydrogen atom on the benzene ring. The reaction requires a Lewis acid catalyst to generate the electrophilic halogen species.
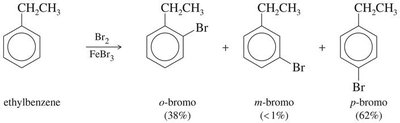
Bromination: Benzene reacts with Br2 in the presence of FeBr3 or Fe to yield bromobenzene and HBr.
Chlorination: Similar to bromination, but typically uses AlCl3 as the catalyst.
Iodination: Requires an oxidizing agent (e.g., HNO3) to generate the electrophilic iodine species.
Comparison to Alkenes: Unlike benzene, alkenes undergo addition reactions with halogens, resulting in dihalide products and loss of double bond character.

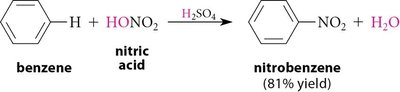
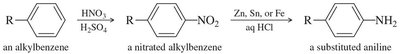
Nitration of Benzene
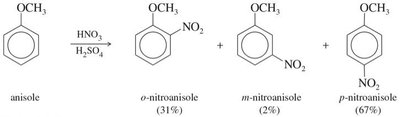
Nitration introduces a nitro group (–NO2) onto the aromatic ring using a mixture of concentrated nitric and sulfuric acids. The active electrophile is the nitronium ion (NO2+).
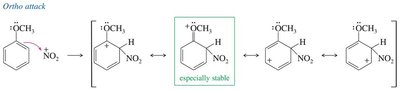
Mechanism: Formation of the nitronium ion, followed by its attack on the benzene ring and subsequent deprotonation.
Reduction of Nitro Group: The nitro group can be reduced to an amino group (–NH2) using Zn, Sn, or Fe in dilute acid, providing a route to aromatic amines.
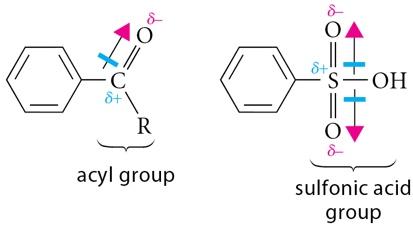
Sulfonation of Benzene
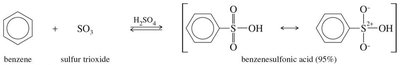
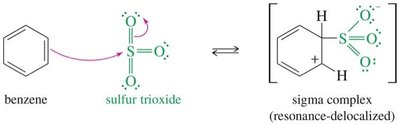
Sulfonation introduces a sulfonic acid group (–SO3H) onto the aromatic ring using fuming sulfuric acid (a mixture of SO3 and H2SO4). The reaction is reversible and can be used to temporarily block a position on the ring.
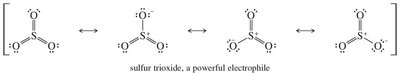
Electrophile: Sulfur trioxide (SO3), a powerful electrophile, is resonance-stabilized.
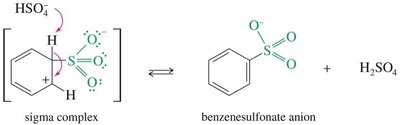
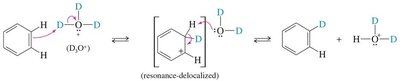
Mechanism: Involves attack of benzene on SO3, formation of a sigma complex, and deprotonation to yield benzenesulfonic acid.
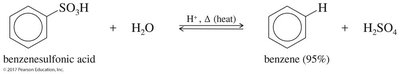
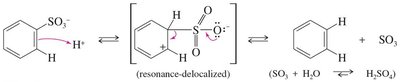
Reversibility: The sulfonic acid group can be removed by heating with dilute acid.
Hydrogen-Deuterium Exchange: Demonstrates the reversibility and mechanism of EAS using deuterium ions (D+).

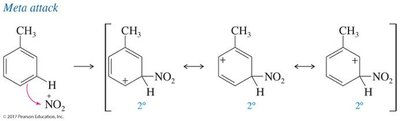
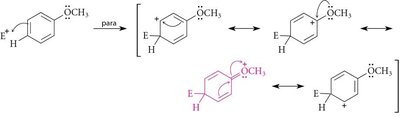
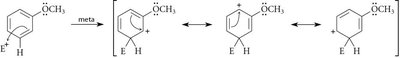
Regioselectivity in EAS: Ortho, Meta, and Para Substitution
The position at which a new substituent is introduced onto a monosubstituted benzene ring depends on the nature of the existing substituent. Substituents are classified as ortho/para-directing or meta-directing based on their electronic effects.
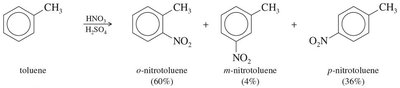
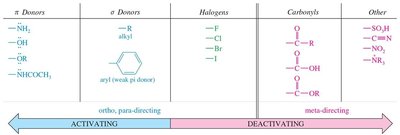
Ortho/Para Directors: Typically electron-donating groups (alkyl, –OH, –OR, –NH2), which activate the ring and direct new substituents to the ortho and para positions.
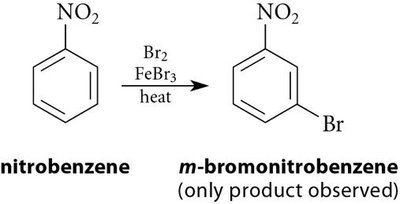
Meta Directors: Electron-withdrawing groups (–NO2, –SO3H, –COOH, –CN) deactivate the ring and direct new substituents to the meta position.
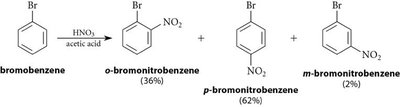
Halogens: Unique in being deactivating but still ortho/para-directing due to their ability to stabilize the sigma complex via resonance.
Activating and Deactivating Effects of Substituents
Substituents on the benzene ring influence both the rate and orientation of EAS reactions through resonance and inductive effects.
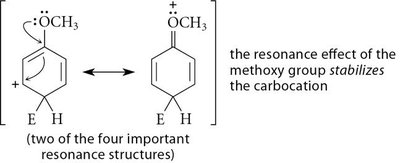
Activating Groups: Increase the rate of EAS by donating electron density to the ring (e.g., alkyl, methoxy, amino groups).
Deactivating Groups: Decrease the rate of EAS by withdrawing electron density (e.g., nitro, carbonyl, sulfonic acid groups).
Resonance Effect: Groups with lone pairs adjacent to the ring can stabilize the carbocation intermediate via resonance.
Polar Effect: Electron-withdrawing groups destabilize the carbocation intermediate, making the ring less reactive.

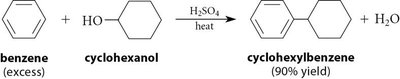
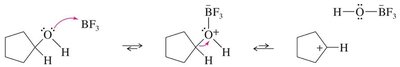
Friedel-Crafts Alkylation and Acylation
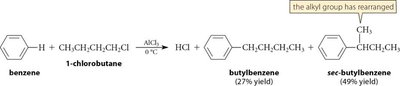
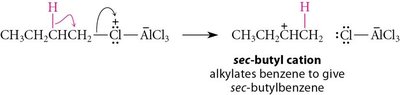
Friedel-Crafts reactions are important methods for introducing alkyl or acyl groups onto aromatic rings using alkyl or acyl halides and a Lewis acid catalyst (usually AlCl3).
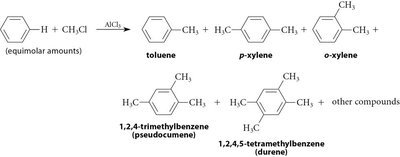
Alkylation: Involves the formation of a carbocation intermediate, which can rearrange, leading to mixtures of products. Polyalkylation is possible due to increased reactivity of the product.
Acylation: Introduces an acyl group, forming a ketone. No rearrangement occurs, and the product is less reactive than benzene, preventing polyacylation.
Limitations: Friedel-Crafts reactions do not work with strongly deactivating groups on the ring.

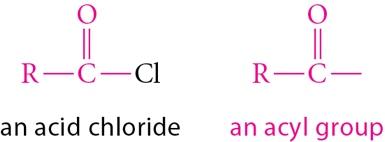
Summary Table: Activating and Deactivating Groups
Type | Examples | Directing Effect |
|---|---|---|
Strong Activators | –OH, –OR, –NH2, –NHR, –NR2 | Ortho/Para |
Moderate Activators | –NHCOR, –OCOR | Ortho/Para |
Weak Activators | –R, –C6H5 | Ortho/Para |
Halogens | –F, –Cl, –Br, –I | Ortho/Para (but deactivating) |
Weak Deactivators | –COOR, –COR, –CHO | Meta |
Strong Deactivators | –NO2, –SO3H, –CN, –NR3+ | Meta |
Applications and Examples
Polysubstitution: The presence of multiple substituents affects both the reactivity and regioselectivity of further substitutions. The most activating group usually dominates the orientation.
Blocking Groups: Sulfonation can be used to temporarily block a position on the ring to control the outcome of multi-step syntheses.
Additional info: These notes cover the core concepts and mechanisms of electrophilic aromatic substitution, including the effects of substituents on reactivity and orientation, and the practical applications of these reactions in organic synthesis.
