 Back
BackReactions of Aromatic Compounds: Electrophilic Aromatic Substitution and Substituent Effects
Study Guide - Smart Notes

Reactions of Aromatic Compounds
Electrophilic Aromatic Substitution (EAS)
Aromatic compounds, such as benzene, are characterized by their stability due to delocalized π electrons. Despite this stability, they can undergo substitution reactions with electrophiles, a process known as electrophilic aromatic substitution (EAS). In EAS, an aromatic hydrogen is replaced by an electrophile, preserving the aromaticity of the ring.
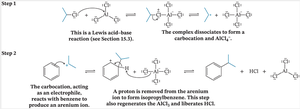
Step 1: Formation of the sigma complex (arenium ion) – The electrophile attacks the aromatic ring, temporarily disrupting aromaticity.
Step 2: Restoration of aromaticity – A proton is removed from the carbon bearing the electrophile, restoring the aromatic π system.
Example: Halogenation, nitration, sulfonation, alkylation, and acylation are common EAS reactions.
Halogenation of Benzene
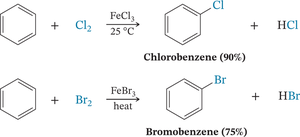
Benzene reacts with halogens (Cl2 or Br2) in the presence of a Lewis acid catalyst (FeCl3 or FeBr3) to form halogenated benzenes. The Lewis acid activates the halogen, making it a stronger electrophile.
Chlorination: Benzene + Cl2 (FeCl3, 25°C) → Chlorobenzene + HCl
Bromination: Benzene + Br2 (FeBr3, heat) → Bromobenzene + HBr
Nitration of Benzene
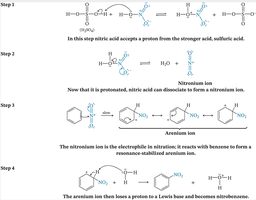
Nitration introduces a nitro group (-NO2) onto the aromatic ring using a mixture of concentrated nitric acid and sulfuric acid. Sulfuric acid acts as a catalyst, generating the nitronium ion (NO2+), the active electrophile.
Step 1: Protonation of nitric acid by sulfuric acid.
Step 2: Formation of the nitronium ion.
Step 3: Electrophilic attack on benzene to form the arenium ion.
Step 4: Deprotonation to restore aromaticity and yield nitrobenzene.
Sulfonation of Benzene
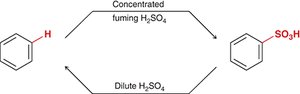
Sulfonation introduces a sulfonic acid group (-SO3H) onto benzene using fuming sulfuric acid (H2SO4 with SO3). The reaction is reversible; concentrated acid favors sulfonation, while dilute acid favors desulfonation.
Friedel–Crafts Alkylation
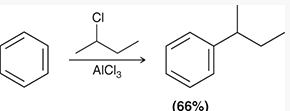
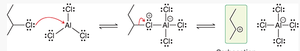
Friedel–Crafts alkylation installs an alkyl group onto an aromatic ring using an alkyl halide and a Lewis acid (AlCl3). The Lewis acid generates a carbocation or a carbocation-like complex, which acts as the electrophile.
Primary alkyl halides often form complexes with AlCl3 rather than free carbocations.
Carbocation rearrangements can occur, leading to mixtures of products.
Polyalkylation is a common side reaction.
Friedel–Crafts Acylation
Friedel–Crafts acylation introduces an acyl group (RCO-) onto an aromatic ring using an acyl chloride and a Lewis acid. The acylium ion (RCO+) is the active electrophile. This reaction avoids carbocation rearrangements and polyacylation.
The product is an aryl ketone, which can be further reduced to an alkylbenzene.
Substituent Effects on Electrophilic Aromatic Substitution
Activating and Deactivating Groups
Substituents on the aromatic ring influence both the rate and the regiochemistry of EAS reactions. They are classified as activating (increase reactivity) or deactivating (decrease reactivity), and as ortho/para directors or meta directors based on where they direct new substituents.
Activating groups: Donate electron density to the ring (e.g., -OH, -OCH3, -NH2, -CH3), usually ortho/para directors.
Deactivating groups: Withdraw electron density (e.g., -NO2, -SO3H, -COOH), usually meta directors.
Halogens: Are deactivating but ortho/para directors due to their resonance donation.
Regiochemistry of Substitution
The position where a new group is introduced depends on the nature of the existing substituent:
Ortho/para directors: Direct new groups to the ortho and para positions (relative to themselves).
Meta directors: Direct new groups to the meta position.
Examples of Substituent Effects
Methyl group (-CH3): Activates the ring and directs to ortho/para positions.
Methoxy group (-OCH3): Strongly activates the ring, also ortho/para director.
Nitro group (-NO2): Strongly deactivates the ring, meta director.
Summary Table: Activators and Deactivators
Type | Examples | Directing Effect |
|---|---|---|
Strong Activators | -OH, -NH2 | Ortho/Para |
Moderate Activators | -OCH3, -NHCOR | Ortho/Para |
Weak Activators | -CH3 | Ortho/Para |
Weak Deactivators | -F, -Cl, -Br, -I | Ortho/Para |
Moderate Deactivators | -COOH, -COOR, -SO3H | Meta |
Strong Deactivators | -NO2, -CF3 | Meta |
Practical Considerations in Synthesis
The order of introducing substituents is crucial for obtaining the desired product.
Blocking groups (e.g., -SO3H) can be used to control regiochemistry and are removable after the desired substitution.
Some reactions (e.g., Friedel–Crafts) are not possible on strongly deactivated rings.
Example: Synthesis Strategy
To synthesize a meta-disubstituted benzene, introduce a meta-directing group first, then the ortho/para-directing group.
Summary
Electrophilic aromatic substitution is the key reaction type for functionalizing aromatic rings.
Substituents affect both the rate and position of further substitution.
Understanding the interplay of activation, deactivation, and directing effects is essential for designing synthetic routes to substituted aromatic compounds.
