 Back
BackReactivity and Spectroscopy of Aldehydes and Ketones
Study Guide - Smart Notes

Chemistry of Carbonyl Compounds: Aldehydes and Ketones
Nucleophilic Addition of Amines
The nucleophilic addition of amines to carbonyl compounds is a fundamental transformation in organic chemistry, leading to the formation of imines and enamines. Both processes proceed through a tetrahedral intermediate, followed by the elimination of water.
Imine Formation: Primary amines react with aldehydes or ketones to form imines (Schiff bases). The reaction is optimal under weakly acidic conditions (pH ≈ 4.5), as strong acid or base slows the process. The mechanism involves nucleophilic attack, proton transfers, and dehydration.
Other Imine Derivatives: Oximes, semicarbazones, and 2,4-dinitrophenylhydrazones are formed by reaction with hydroxylamine, semicarbazide, and 2,4-dinitrophenylhydrazine, respectively.
Enamine Formation: Secondary amines react with aldehydes or ketones to form enamines. The mechanism is identical to imine formation until the iminium ion intermediate, which then loses a proton to yield the enamine.
Example: Reaction of acetone with methylamine yields an imine; reaction with pyrrolidine yields an enamine.
Deoxygenation Reactions
Wolff-Kishner Reaction: Converts aldehydes and ketones to alkanes via hydrazone intermediates under strongly basic conditions. Mechanism involves imine formation and subsequent reduction.
Clemmensen Reduction: Also reduces aldehydes and ketones to alkanes, but under strongly acidic conditions using zinc amalgam. Mechanistically complex and complementary to Wolff-Kishner.
Nucleophilic Addition of Alcohols: Acetal Formation
Alcohols add to carbonyl compounds to form hemiacetals and acetals. The reaction is reversible and acid-catalyzed.
Mechanism: Involves nucleophilic attack by alcohol, proton transfers, and loss of water. All steps are reversible.
Reaction Conditions: Forward reaction is favored by removal of water (e.g., distillation); reverse (hydrolysis) is favored by excess aqueous acid.
Scope: Only aldehydes and ketones form acetals; carboxylic acids and esters do not.
Cyclic Acetals: Diols can react to form cyclic acetals, which are useful as protecting groups for carbonyls.
Protecting Groups: Acetals are stable to hydride reducing agents, making them valuable for selective transformations.
The Wittig Reaction
The Wittig reaction is a powerful method for converting aldehydes and ketones to alkenes using phosphorous ylides.
Ylide Formation: Prepared by SN2 reaction of a primary (or some secondary) alkyl halide with triphenylphosphine (PPh3), followed by deprotonation with base.
Mechanism: The ylide reacts with the carbonyl to form a betaine intermediate, which collapses to yield an alkene and triphenylphosphine oxide.
Scope: Mono-, di-, and trisubstituted alkenes can be synthesized; tetrasubstituted alkenes are generally inaccessible due to steric hindrance. Simple disubstituted alkenes are formed with cis selectivity.
Example: Reaction of benzaldehyde with a phosphonium ylide yields styrene.
Conjugate Addition of Nucleophiles
α,β-Unsaturated carbonyl compounds undergo two types of nucleophilic addition: direct (1,2-) and conjugate (1,4-).
1,2-Addition: Nucleophile adds directly to the carbonyl carbon.
1,4-Addition (Conjugate Addition): Nucleophile adds to the β-carbon, facilitated by polarization of the double bond by the carbonyl group.
Amines: Both primary and secondary amines add to α,β-unsaturated carbonyls via 1,4-addition, yielding β-amino aldehydes or ketones.
Cuprates (Gilman Reagents): Organocopper reagents add to α,β-unsaturated ketones (not aldehydes), introducing alkyl, vinyl, or aryl groups. Alkynes cannot be added this way.
Comparison: Cuprates favor 1,4-addition; Grignard and organolithium reagents favor 1,2-addition.
Spectroscopic Identification of Aldehydes and Ketones
Infrared (IR) Spectroscopy
IR spectroscopy is a key tool for identifying functional groups in organic molecules. Aldehydes and ketones display characteristic absorptions due to the C=O stretch.
Aldehyde C=O Stretch: 1660–1770 cm-1 (strong)
Aldehyde C–H Stretch: 2720–2820 cm-1 (two absorptions)
Ketone C=O Stretch: 1660–1770 cm-1 (strong)
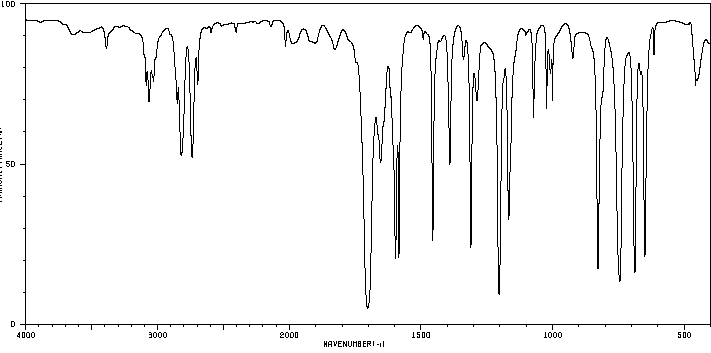
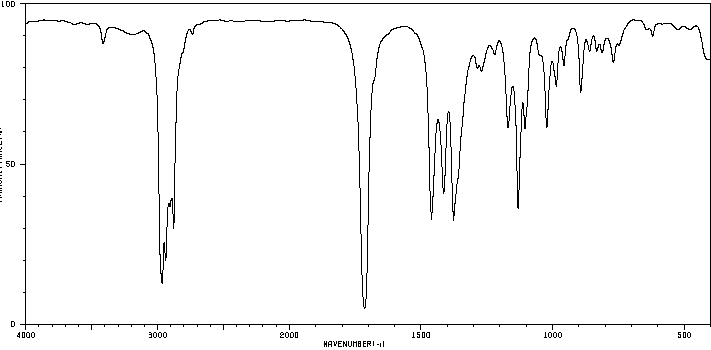
Carbonyl Type | Example | IR Absorption (cm-1) |
|---|---|---|
aliphatic aldehyde | acetaldehyde | 1730 |
aromatic aldehyde | benzaldehyde | 1705 |
α,β-unsaturated aldehyde | 2-propenal | 1705 |
aliphatic ketone | acetone | 1715 |
aromatic ketone | acetophenone | 1690 |
α,β-unsaturated ketone | 3-buten-2-one | 1685 |
6 membered ring ketone | cyclohexanone | 1715 |
5 membered ring ketone | cyclopentanone | 1750 |
4 membered ring ketone | cyclobutanone | 1785 |
Nuclear Magnetic Resonance (NMR) Spectroscopy
NMR spectroscopy provides detailed information about the hydrogen and carbon environments in aldehydes and ketones.
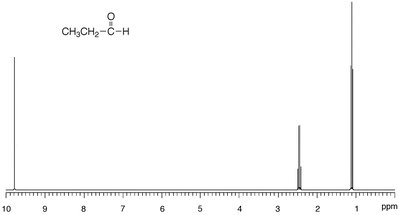
1H NMR: Protons adjacent to the carbonyl are slightly deshielded (δ 2.0–2.3 ppm). Aldehyde protons appear at δ ~10.0 ppm and often show fine splitting.
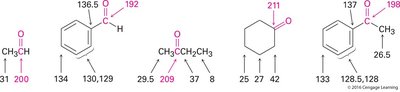
13C NMR: Carbonyl carbons of aldehydes and ketones resonate at δ 190–215 ppm. Saturated ketones: δ 200–215 ppm; α,β-unsaturated: δ 190–200 ppm.
Mass Spectrometry
Mass spectrometry is used to determine molecular weight and fragmentation patterns. Aldehydes and ketones show two characteristic fragmentation modes:
Alpha Cleavage: Cleavage of the bond adjacent to the carbonyl group, generating a resonance-stabilized acylium ion.
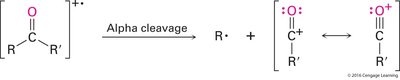
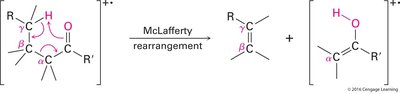
McLafferty Rearrangement: Occurs in compounds with γ-hydrogens, resulting in transfer of a hydrogen and cleavage to form an alkene and an enol or carbonyl compound.
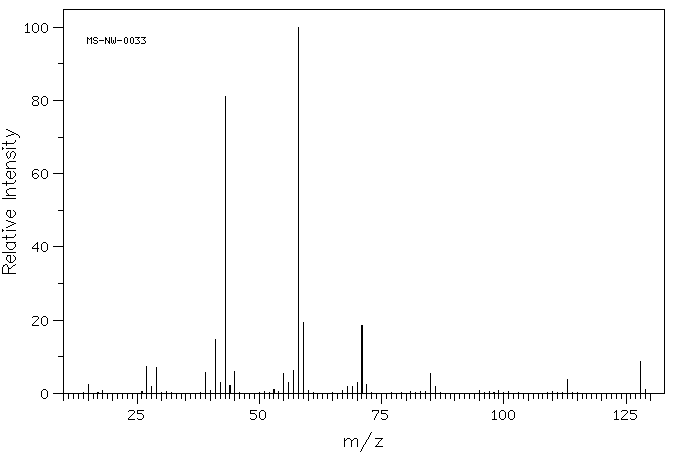
Example: Mass spectrum of 2-octanone shows characteristic peaks due to these fragmentations.
Ultraviolet (UV) Spectroscopy
UV spectroscopy is useful for detecting conjugated systems. Saturated aldehydes and ketones are not UV active, but α,β-unsaturated aldehydes and ketones absorb in the UV region due to extended conjugation.
Application: Useful for distinguishing between saturated and conjugated carbonyl compounds.
Additional info: The above notes integrate mechanistic, synthetic, and spectroscopic aspects of aldehydes and ketones, providing a comprehensive overview suitable for exam preparation in a college-level organic chemistry course.
