Back
BackStereoisomerism and Chirality: Foundations and Applications
Study Guide - Smart Notes

Chapter 3: Stereoisomerism and Chirality
Chirality: The Handedness of Molecules
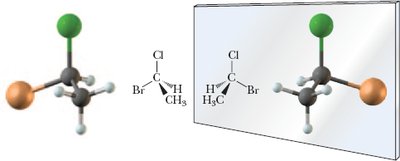
Chirality is a fundamental concept in organic chemistry, describing objects or molecules that are not superposable on their mirror images. This property is crucial for understanding the behavior of many organic compounds, especially in biological systems.
Chiral: Objects or molecules that are not superposable on their mirror images.
Achiral: Objects or molecules that are superposable on their mirror images.
Plane of symmetry: An imaginary plane dividing an object so that one half is the mirror image of the other.
Center of symmetry: A point in a molecule where identical components are located equidistant and opposite along any axis through that point.
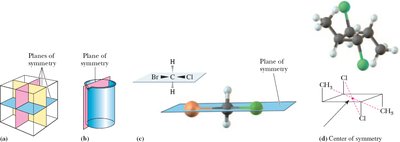
Key Point: The absence of symmetry elements (plane or center of symmetry) often indicates chirality in a molecule.
Stereoisomerism
Stereoisomers are compounds with the same molecular formula and connectivity but different spatial arrangements of atoms. Stereoisomerism is a central theme in organic chemistry, affecting physical and chemical properties.
Constitutional isomers: Same molecular formula, different connectivity.
Stereoisomers: Same molecular formula and connectivity, different spatial arrangement.
Configurational isomers: Stereoisomers that cannot be interconverted by rotation around single bonds.
Cis-trans isomerism: A type of configurational isomerism, especially in alkenes and cyclic compounds.
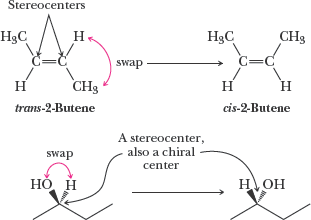
Example: Cis- and trans-2-butene are configurational isomers but not chiral; they are diastereomers, not enantiomers.
Enantiomers and Diastereomers
Enantiomers are stereoisomers that are nonsuperposable mirror images of each other, while diastereomers are stereoisomers that are not mirror images.
Enantiomers: Always come in pairs; have identical physical and chemical properties in achiral environments.
Diastereomers: Stereoisomers that are not mirror images; can have different physical and chemical properties.
Chiral center: Typically a carbon atom bonded to four different groups.
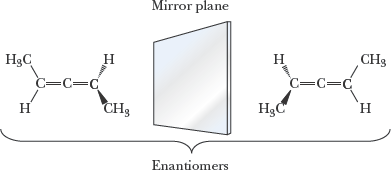
Example: Swapping two groups at a chiral center generates the enantiomer.
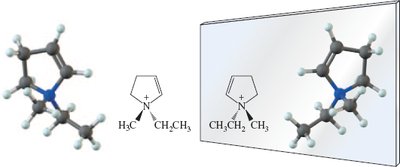
Additional info: Chiral centers can also be found in atoms other than carbon, such as silicon, phosphorus, and germanium.
Conformational Isomerism
Conformational isomers arise from rotation around single bonds. Unlike configurational isomers, they can interconvert rapidly at room temperature.
Gauche and anti forms: Different spatial arrangements in butane due to rotation around the C–C bond.
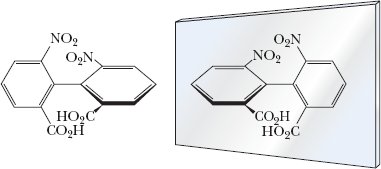
Atropisomers: Stereoisomers that lack a chiral center but are chiral due to restricted rotation.
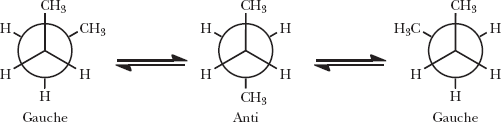
Key Point: Conformational isomers are not true stereoisomers unless the barrier to rotation is high enough to prevent interconversion at room temperature.
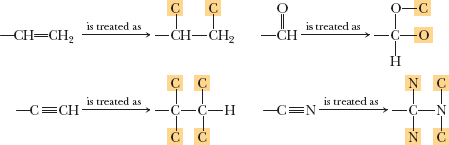
Naming Chiral Centers: The R, S System
The Cahn-Ingold-Prelog (CIP) system assigns absolute configuration (R or S) to chiral centers based on a set of priority rules.
R (rectus): Clockwise arrangement of priorities.
S (sinister): Counterclockwise arrangement of priorities.
Priority rules:
Assign priority based on atomic number of atoms directly attached to the chiral center.
If a tie, move outward atom by atom until a difference is found.
Multiple bonds are treated as an equivalent number of single-bonded "phantom" atoms.
First point of difference determines priority.
Example: Assigning R or S configuration to chiral centers in various molecules.
Molecules with Multiple Stereocenters
Molecules with two or more stereocenters can have multiple stereoisomers. The maximum number is given by the formula $2^n$, where n is the number of chiral centers.
Enantiomers: Each chiral center can be R or S, leading to pairs of enantiomers.
Diastereomers: Stereoisomers that are not mirror images.
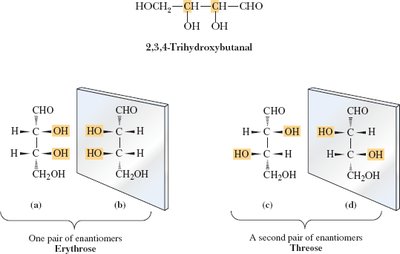
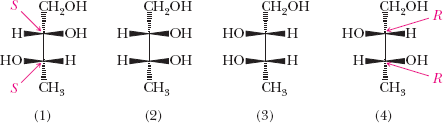
Example: 2,3,4-Trihydroxybutanal and tartaric acid each have multiple stereoisomers, including pairs of enantiomers and meso compounds.
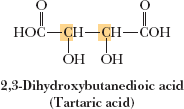
Meso Compounds
Meso compounds are achiral molecules that contain two or more chiral centers but have an internal plane of symmetry, making them superposable on their mirror images.
Key property: Meso compounds reduce the number of possible stereoisomers below $2^n$.
Example: Tartaric acid has three stereoisomers: a pair of enantiomers and one meso compound.
Property | (R,R)-Tartaric Acid | (S,S)-Tartaric Acid | Meso Tartaric Acid |
|---|---|---|---|
Specific rotation | +12.7 | -12.7 | 0 |
Melting point (°C) | 171-174 | 171-174 | 146-148 |
Density at 20°C (g/cm³) | 1.7598 | 1.7598 | 1.660 |
Solubility in water at 20°C (g/100 mL) | 139 | 139 | 125 |
pKa1 (25°C) | 2.98 | 2.98 | 3.22 |
pKa2 (25°C) | 4.34 | 4.34 | 4.82 |
Key Point: Enantiomers have identical physical and chemical properties in achiral environments, while meso compounds differ.
Fischer Projection Formulas
Fischer projections are two-dimensional representations of molecules, especially useful for carbohydrates and molecules with multiple chiral centers.
Horizontal lines represent bonds projecting out of the plane (toward the viewer).
Vertical lines represent bonds projecting behind the plane (away from the viewer).
Example: Fischer projection of (R)-glyceraldehyde.
Cyclic Molecules with Two or More Chiral Centers
Cyclic compounds such as cyclopentane and cyclohexane derivatives can have multiple stereocenters, leading to various stereoisomers, including enantiomers, diastereomers, and meso compounds.
Disubstituted cyclopentanes: Can have cis and trans isomers, each existing as a pair of enantiomers or as meso compounds.
Disubstituted cyclohexanes: Chair conformations are important for analyzing symmetry and chirality.
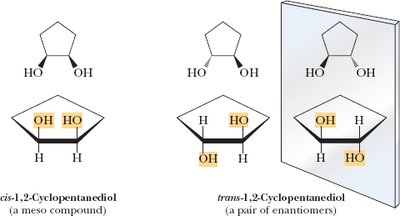
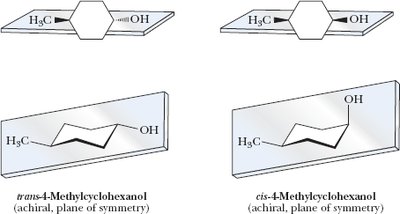
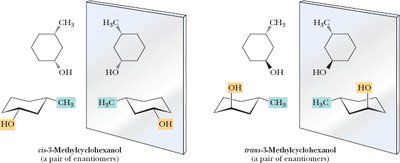
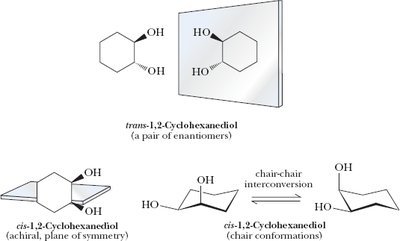
Key Point: The presence of a plane of symmetry in a cyclic molecule often indicates achirality or a meso compound.
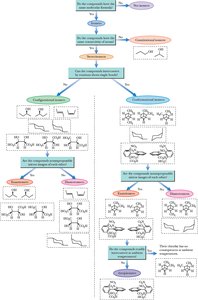
Summary Flowchart: Classification of Isomers
A comprehensive flowchart can help classify isomers as constitutional, configurational, conformational, enantiomers, diastereomers, or meso compounds based on their structural features and symmetry.