 Back
BackStudy Guide: Chemistry – A Molecular Approach (Canadian Edition)
Study Guide - Smart Notes

Chemistry: A Molecular Approach – Overview
Introduction to Chemistry and Its Relevance
Chemistry is the study of matter, its properties, and the changes it undergoes. Understanding chemistry is essential for appreciating the molecular basis of the world and for engaging with scientific and societal issues. The textbook emphasizes the importance of chemistry in everyday life, biomedical contexts, and environmental issues.
Intrinsic Value: Chemistry reveals the extraordinary nature of the universe at the molecular level.
Extrinsic Value: Scientific literacy enables informed citizenship and engagement with contemporary issues.
Pedagogical Approach: The book uses clear language, visual aids, and problem-solving frameworks to support student learning.
Key Features of the Textbook
Visual and Conceptual Learning
The textbook employs pioneering artwork and multi-part images to connect macroscopic observations, molecular processes, and symbolic representations. This approach helps students visualize chemical phenomena and understand their underlying principles.
Macroscopic Images: Show observable phenomena.
Molecular Images: Illustrate atomic and molecular interactions.
Symbolic Representations: Use chemical equations and formulas.

Example: The cover image depicts the hexagonal lattice of water ice, highlighting hydrogen bonding between H2O molecules. This structure is fundamental to understanding physical properties of water and phase transitions.
Periodic Properties of the Elements
Nerve Signal Transmission and the Periodic Table
The periodic table organizes elements based on recurring chemical properties. Understanding periodic trends is crucial for explaining phenomena such as nerve signal transmission, which relies on the movement of sodium and potassium ions across cell membranes.
Periodic Trends: Atomic size, ionization energy, electron affinity, and metallic character.
Quantum Mechanics: Provides the theoretical basis for periodic properties.
Applications: Biological processes, such as nerve impulses, depend on ion transport.
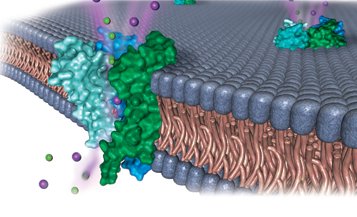
Example: Ion channels in nerve cells allow sodium and potassium ions to flow in opposite directions, enabling signal transmission.
Chemical Bonding: Models and Theories
Lewis Theory, Valence Bond Theory, and Molecular Orbital Theory
Chemical bonding is central to understanding molecular structure and reactivity. Three major theories are used:
Lewis Theory: Uses dots and dashes to represent valence electrons and predict stable molecules.
Valence Bond Theory: Treats electrons quantum-mechanically, focusing on orbital overlap.
Molecular Orbital Theory: Considers electrons as delocalized over the entire molecule, offering greater predictive power.
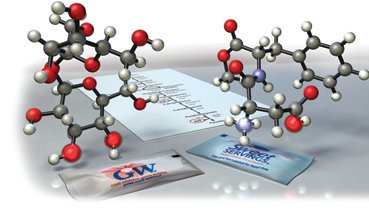
Example: The shape of molecules, such as artificial sweeteners, determines their ability to interact with biological receptors.
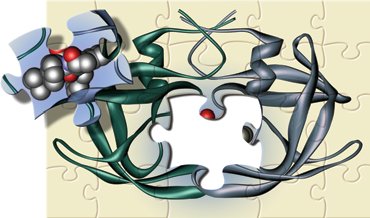
Example: Drug design, such as HIV-protease inhibitors, relies on understanding chemical bonding and molecular structure.
Chemical Reactions and Stoichiometry
Balancing Equations and Reaction Types
Chemical reactions involve the transformation of reactants into products. Stoichiometry quantifies the relationships between reactants and products.
Balanced Equations: Ensure conservation of mass and charge.
Types of Reactions: Precipitation, acid-base, oxidation-reduction.
Stoichiometry: Relates amounts of substances using mole ratios.

Example: Mixing baking soda (NaHCO3) and vinegar (acetic acid) produces carbon dioxide gas, water, and sodium ions.
Solutions and Solubility
Properties and Formation of Solutions
Solutions are homogeneous mixtures where atoms and molecules intermingle. Their properties differ from pure substances, and understanding solubility is key to many chemical and biological processes.
Types of Solutions: Aqueous, gaseous, solid.
Solubility Factors: Intermolecular forces, temperature, pressure.
Colligative Properties: Depend on the number of solute particles (e.g., boiling point elevation, freezing point depression).

Example: Drinking seawater causes dehydration because its high salt concentration draws water out of body tissues.
Environmental and Biomedical Contexts
Pharmaceuticals, Personal Care Products, and Blood Pressure
Chemistry is relevant to environmental and health issues, such as the impact of pharmaceuticals and personal care products (PPCPs) on water quality and the measurement of blood pressure in medicine.
PPCPs: Can bioaccumulate and affect aquatic life.
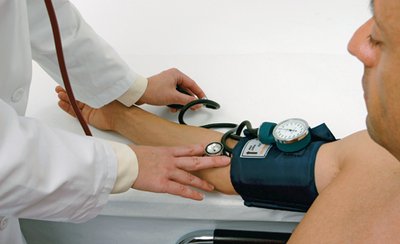
Blood Pressure: Measured using a sphygmomanometer; high blood pressure increases risk of cardiovascular disease.

Problem-Solving Framework
Conceptual Plans and Worked Examples
The textbook uses a consistent four-part structure for problem-solving: Sort, Strategize, Solve, and Check. Conceptual plans visually outline the steps from given information to the solution.
Sort: Identify what is given and what is required.
Strategize: Devise a solution strategy.
Solve: Implement the steps.
Check: Verify the answer for consistency.
Summary Table: Key Equations and Relationships
Equation | Description |
|---|---|
Pressure as force per unit area | |
Boyle's Law (Pressure-Volume relationship) | |
Charles's Law (Volume-Temperature relationship) | |
Avogadro's Law (Volume-Mole relationship) | |
Ideal Gas Law |
End-of-Chapter Review and Practice
Key Terms, Concepts, and Skills
Key Terms: Pressure, mole, stoichiometry, solubility, ionization energy, etc.
Key Concepts: Conservation of mass, periodic trends, chemical bonding, solution properties.
Key Skills: Balancing equations, calculating molar mass, predicting molecular geometry, solving stoichiometry problems.
Conclusion
This study guide summarizes the foundational concepts, visual learning strategies, and problem-solving approaches presented in "Chemistry: A Molecular Approach" (Canadian Edition). The textbook is designed to connect chemistry to real-world contexts, support student learning, and prepare students for advanced study in chemistry and related fields.
