 Back
BackStudy Guide: The Study of Chemical Reactions (Organic Chemistry)
Study Guide - Smart Notes

CHAPTER 04: The Study of Chemical Reactions
Overview of Chemical Reactions
Chemical reactions in organic chemistry involve the transformation of molecules through the breaking and forming of chemical bonds. Understanding these reactions requires knowledge of their mechanisms, thermodynamics, and kinetics.
Mechanism: The step-by-step sequence of events by which reactants are converted into products, detailing which bonds break and form.
Thermodynamics: The study of energy changes during reactions, predicting the stability and equilibrium of reactants and products.
Kinetics: The study of reaction rates and how they change with varying conditions.
Bond-Dissociation Enthalpy (BDE)
Bond-dissociation enthalpy (BDE) is the energy required to break a specific bond in a molecule, often measured in kJ/mol or kcal/mol. It is a key parameter in understanding reaction energetics.
Homolytic cleavage: Each atom retains one electron from the bond, forming free radicals.
Heterolytic cleavage: Both electrons go to one atom, forming ions.

Types of Bond Cleavage
Bond cleavage can occur in two main ways, each leading to different intermediates:
Homolytic cleavage: Produces free radicals, important in radical chain reactions.
Heterolytic cleavage: Produces ions, relevant in ionic mechanisms.
Free Radical Chain Reactions
Free radical chain reactions are a fundamental class of organic reactions, exemplified by the chlorination of methane. These reactions proceed via a chain mechanism with distinct steps:
Initiation: Formation of reactive intermediates (radicals).
Propagation: Radicals react with stable molecules to form products and new radicals, sustaining the chain.
Termination: Side reactions that consume radicals, ending the chain.


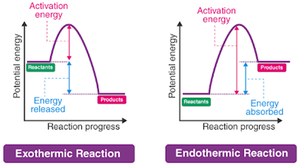
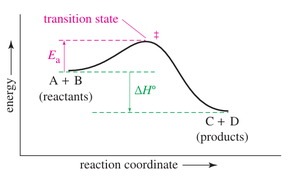
Reaction-Energy Diagrams
Reaction-energy diagrams graphically represent the energy changes during a reaction, showing the energy of reactants, products, and the transition state. The difference between reactant and product energy is the enthalpy change (), and the energy required to reach the transition state is the activation energy ().
Exothermic reactions: Release energy; products are lower in energy than reactants.
Endothermic reactions: Absorb energy; products are higher in energy than reactants.
The Rate-Limiting Step
In multistep reactions, the rate-limiting step is the slowest step, which determines the overall reaction rate. It is associated with the highest energy barrier in the reaction pathway.
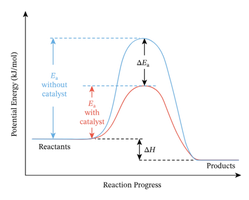
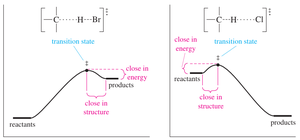
The Hammond Postulate
The Hammond Postulate states that the structure of a transition state resembles the structure of the closest stable species in energy. For exothermic steps, the transition state is reactant-like; for endothermic steps, it is product-like.
Exothermic step: Transition state resembles reactants.
Endothermic step: Transition state resembles products.


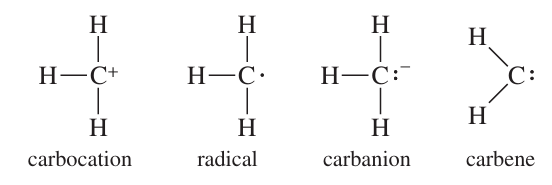
Reactive Intermediates
Reactive intermediates are short-lived species formed during reactions, including free radicals, carbocations, carbanions, and carbenes. They are highly reactive and rarely accumulate in high concentrations.
Free radicals: Species with an unpaired electron.
Carbocations: Positively charged carbon species.
Carbanions: Negatively charged carbon species.
Carbenes: Neutral species with two nonbonding electrons on carbon.
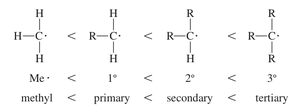
Stability of Free Radicals
The stability of free radicals depends on their structure. Tertiary radicals are more stable than secondary, primary, or methyl radicals due to greater alkyl group substitution, which provides stabilization via hyperconjugation and inductive effects.
Radical Type | Structure | Relative Stability |
|---|---|---|
Methyl | H3C• | Least stable |
Primary | RCH2• | More stable |
Secondary | R2CH• | Even more stable |
Tertiary | R3C• | Most stable |
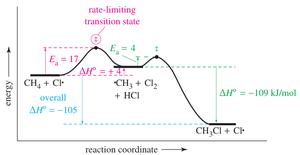
Example: Reaction-Energy Diagram for a Given Reaction
Consider a reaction with an activation energy () of +17 kJ/mol and a of +4 kJ/mol. The reaction-energy diagram would show the reactants, a transition state at +17 kJ/mol above the reactants, and products at +4 kJ/mol above the reactants.
Activation energy (): Energy required to reach the transition state.
Enthalpy change (): Energy difference between reactants and products.
Equation:
Diagram:

