 Back
BackStudy Notes: Alkanes, Cycloalkanes, and Functional Groups in Organic Chemistry
Study Guide - Smart Notes

Alkanes and Cycloalkanes
Introduction to Alkanes
Alkanes are a fundamental class of organic compounds known as saturated hydrocarbons. They consist only of carbon and hydrogen atoms, with all carbon-carbon bonds being single bonds. Their general formula is .
Definition: Alkanes are saturated hydrocarbons, meaning each carbon atom is bonded to as many hydrogen atoms as possible.
Types: Alkanes can be straight-chain, branched-chain, or cyclic (cycloalkanes).
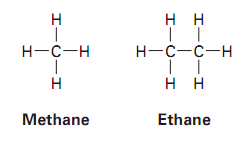
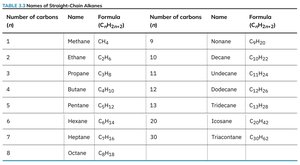
Example: Methane (), ethane (), propane (), and butane () are simple straight-chain alkanes.
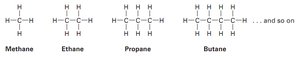
Types of Alkanes
Alkanes are classified based on their structure:
Straight-chain alkanes: Carbon atoms are connected in a continuous chain.
Branched-chain alkanes: Carbon chains branch off from the main chain.
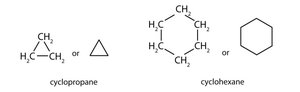
Cycloalkanes: Carbon atoms form a ring structure.
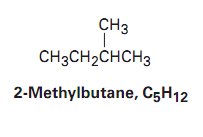
Isomers and Constitutional Isomers
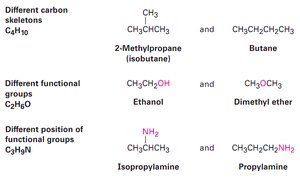
Isomers are compounds with the same molecular formula but different arrangements of atoms.
Constitutional isomers: Isomers with different connectivity of atoms.
Example: Butane and isobutane () are constitutional isomers.
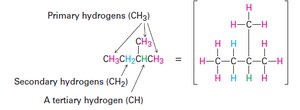
Alkyl Groups
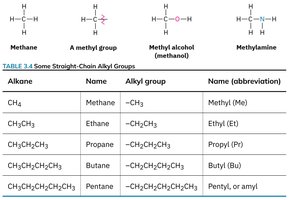
An alkyl group is formed by removing a hydrogen atom from an alkane.
Definition: The partial structure that remains is called an alkyl group.
Examples: Methyl (), ethyl (), propyl ().

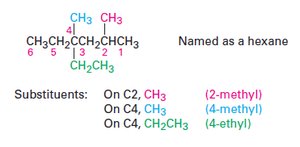
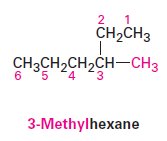
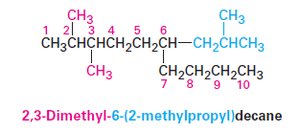
Naming Alkanes: IUPAC Nomenclature
The IUPAC system provides a systematic method for naming organic compounds.
Prefix: Identifies substituent groups.
Parent: Indicates the main carbon chain.
Locants: Specify positions of substituents.
Suffix: Identifies the primary functional group.

Steps in IUPAC Nomenclature
Identify the parent hydrocarbon.
Number the atoms in the longest chain.
Identify and number the substituents.
Write the name as a single word.
Name complex substituents as compounds.

Properties and Reactions of Alkanes
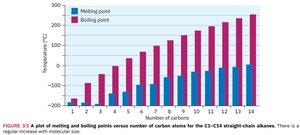
Physical properties: Alkanes show regular increases in boiling and melting points with increasing molecular weight due to dispersion forces.
Chemical properties: Alkanes are generally unreactive but can undergo combustion and halogenation.
Combustion equation:

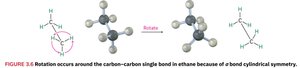
Conformations of Ethane
Stereochemistry examines the three-dimensional aspects of molecules. Ethane can rotate around its C–C bond due to the cylindrical symmetry of the sigma bond.
Conformations: Different spatial arrangements due to bond rotation.
Conformers: Molecules with different conformations.
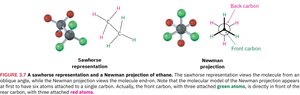
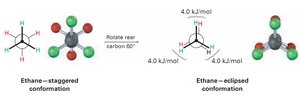
Staggered and Eclipsed Conformations
Staggered: All C–H bonds are as far apart as possible; lowest energy.
Eclipsed: C–H bonds are as close as possible; highest energy.

Functional Groups in Organic Chemistry
Definition and Importance
Functional groups are specific groups of atoms within molecules that determine the chemical behavior of those molecules.
Consistency: Functional groups react similarly in different compounds.
Example: Both ethylene and menthene contain a carbon–carbon double bond and react with bromine in the same way.
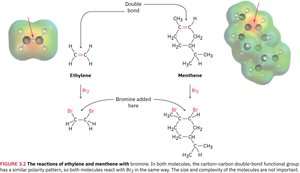
Structures of Common Functional Groups
Functional groups include alkenes, alkynes, aromatic rings, alcohols, ethers, amines, thiols, and more.
Examples: Alkene (C=C), Alkyne (C≡C), Alcohol (–OH), Ether (–O–), Amine (–NH2).
Functional Group | Structure | Example |
|---|---|---|
Alkene | C=C | Ethylene |
Alkyne | C≡C | Acetylene |
Aromatic ring | C6H6 | Benzene |
Alcohol | –OH | Ethanol |
Ether | –O– | Dimethyl ether |
Amine | –NH2 | Methylamine |
Thiols | –SH | Methanethiol |


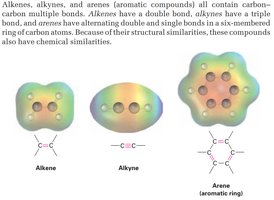
Functional Groups with Carbon-Carbon Multiple Bonds
Alkenes: Double bond (C=C)
Alkynes: Triple bond (C≡C)
Arenes: Alternating double and single bonds in a ring
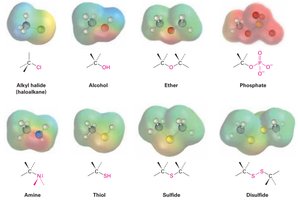
Functional Groups with Carbon Singly Bonded to Electronegative Atoms
Examples: Alkyl halides, alcohols, ethers, amines, thiols, sulfides, disulfides
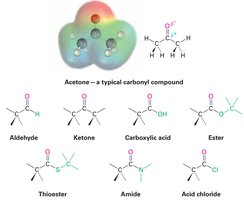
Functional Groups with Carbon-Oxygen Double Bond (Carbonyl Groups)
Carbonyl groups are present in many organic and biological molecules.
Examples: Aldehydes, ketones, carboxylic acids, esters, amides, acid chlorides
Polarity: Carbonyl carbon is partially positive (), oxygen is partially negative ()
Cycloalkanes
Definition and Structure
Cycloalkanes are saturated cyclic hydrocarbons with the general formula .
Less flexibility: Cycloalkanes have restricted rotation compared to open-chain alkanes.
Example: Cyclopropane is rigid and planar; cyclohexane adopts a chair conformation.

Chair Conformation of Cyclohexane
Cyclohexane adopts a strain-free, three-dimensional chair conformation.
Stability: Chair conformation minimizes torsional and angle strain.
Steps to draw: Draw two parallel lines, add topmost and bottommost carbons, connect bonds.

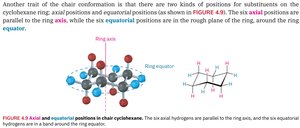
Axial and Equatorial Substituents
In the chair conformation, substituents occupy either axial (parallel to ring axis) or equatorial (around ring equator) positions.
Axial: Parallel to ring axis
Equatorial: Around ring equator

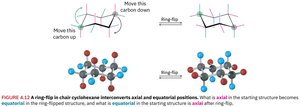
Ring-Flip of Chair Conformation
Cyclohexane can undergo a ring-flip, interconverting axial and equatorial positions.
Mobility: Chair conformations rapidly interconvert at room temperature.
Effect: Substituents switch between axial and equatorial positions.
Summary Table: Functional Groups and Their Properties
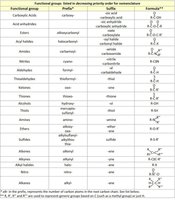
Functional Group | Prefix | Suffix | Formula |
|---|---|---|---|
Carboxylic Acid | carboxy | oic acid | COOH |
Acid Anhydride | alkoxycarbonyl | anhydride | RCOOCOOR |
Ester | alkoxycarbonyl | oate | COOR |
Amide | aminocarbonyl | amide | CONH2 |
Aldehyde | formyl | al | CHO |
Ketone | oxo | one | CO |
Alcohol | hydroxy | ol | OH |
Amine | amino | amine | NH2 |
Alkene | alkenyl | ene | C=C |
Alkyne | alkynyl | yne | C≡C |
Alkane | alkyl | ane | C–C |
Conclusion
These notes provide a comprehensive overview of alkanes, cycloalkanes, and functional groups, including their definitions, structures, nomenclature, properties, and conformational analysis. Understanding these foundational concepts is essential for further study in organic chemistry. Additional info: Academic context was added to clarify definitions, examples, and the importance of functional groups and conformational analysis.
