 Back
BackSynthesis and Characterization of Esters: Fischer Esterification and Spectroscopic Analysis
Study Guide - Smart Notes

Synthesis and Characterization of Esters
Introduction to Esters
Esters are an important class of organic compounds characterized by the functional group -COOR. They are commonly found in natural products and are responsible for the pleasant aromas and flavors of many fruits and flowers. Esters are widely used in artificial flavorings and fragrances due to their distinctive odors.
Structure: Esters have the general formula RCOOR', where R and R' are alkyl or aryl groups.
Formation: Esters are typically synthesized by the reaction of a carboxylic acid with an alcohol, a process known as esterification.
Properties: Many esters are volatile and have strong, fruity odors.
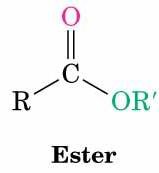
Artificial Flavorings and Common Esters
Many artificial flavors are created by synthesizing esters that mimic the natural flavors found in fruits and other foods. The table below lists some common esters and their associated flavors:
Name | Structure | Flavor |
|---|---|---|
1-propyl acetate | CH3CO2CH2CH2CH3 | Pear |
Octyl acetate | CH3CO2(CH2)7CH3 | Oranges |
Isopentyl acetate | CH3CO2CH2CH2CH(CH3)2 | Banana |
Isobutyl propionate | CH3CH2CO2CH2CH2CH(CH3)2 | Rum |
Ethyl butyrate | CH3CH2CH2CO2CH2CH3 | Pineapples |
Fischer Esterification: Synthesis of Esters
Overview of Fischer Esterification
The Fischer esterification is a classic method for synthesizing esters from carboxylic acids and alcohols in the presence of an acid catalyst. The reaction is an example of nucleophilic acyl substitution and is reversible, so conditions must be optimized to favor ester formation.
General Reaction:
Equilibrium: The reaction is driven to completion by using an excess of one reactant or by removing water as it forms (Le Châtelier’s Principle).
Catalyst: Concentrated sulfuric acid is commonly used as both a catalyst and a dehydrating agent.
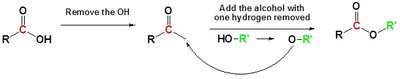
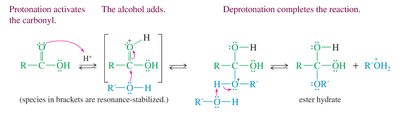
Mechanism of Fischer Esterification
The mechanism proceeds in two main stages: acid-catalyzed addition of the alcohol to the carbonyl group, followed by acid-catalyzed dehydration to form the ester.
Step 1: Protonation of the carbonyl oxygen increases the electrophilicity of the carbonyl carbon.
Step 2: Nucleophilic attack by the alcohol forms a tetrahedral intermediate.
Step 3: Proton transfers facilitate the departure of water and formation of the ester.
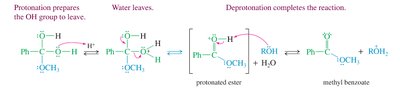
Other Methods for Ester Synthesis
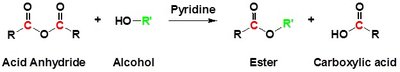
Besides Fischer esterification, esters can also be synthesized using acid chlorides or acid anhydrides with alcohols, or by transesterification.
Acid Chlorides: React with alcohols to form esters and HCl.
Acid Anhydrides: React with alcohols to form esters and carboxylic acids.
Transesterification: Exchange of the alkoxy group of an ester with another alcohol.
Experimental Techniques in Ester Synthesis
Reflux Setup and Laboratory Practice
Reflux is a common technique used to heat reaction mixtures for extended periods without loss of solvent. Proper assembly and use of the reflux apparatus are essential for safety and efficiency.
Lightly grease the ground glass joint of the condenser to ensure a good seal.
Connect water hoses correctly: water in at the lower nozzle, water out at the upper nozzle.
Clamp the round-bottom flask securely and ensure all connections are tight before heating.



Safe Laboratory Practices
Safety is paramount in the organic laboratory. Always wear appropriate PPE (goggles, gloves, lab apron), avoid direct inhalation of chemicals, and use wafting to detect odors.
Never eat, drink, or use cell phones in the lab.
Dispose of chemical waste properly as instructed.
Use the wafting technique to safely detect odors.

Spectroscopic Characterization of Esters
Infrared (IR) Spectroscopy
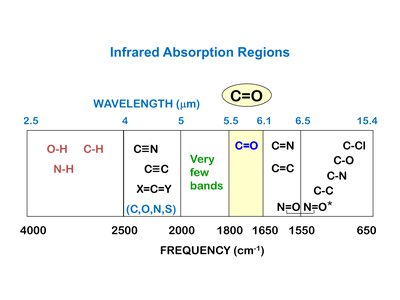
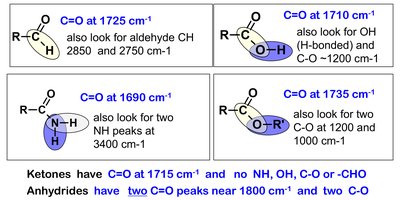
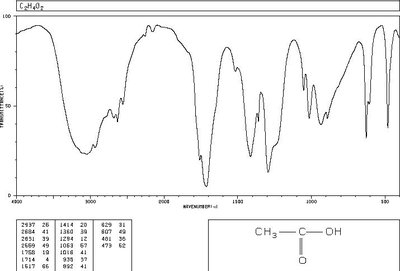
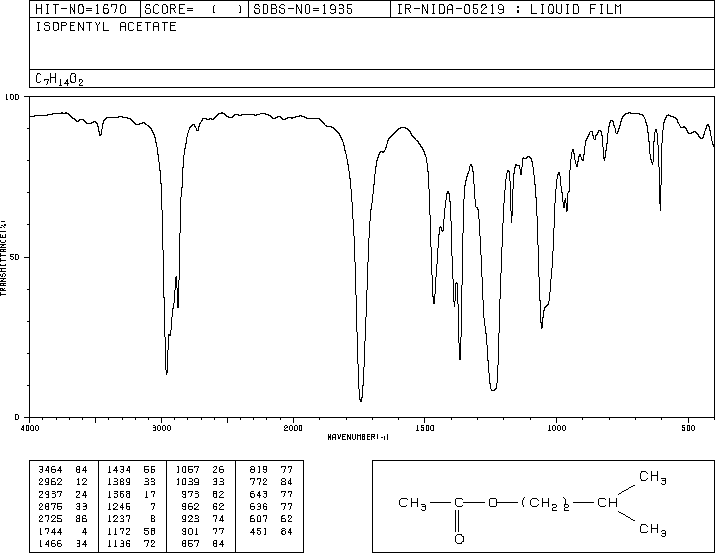
IR spectroscopy is used to identify functional groups in organic molecules. The carbonyl (C=O) stretch is a strong, characteristic absorption in the IR spectrum, typically found between 1800 and 1650 cm-1.
C=O Stretch: Strong absorption, base value for ketones is 1715 cm-1; for esters, typically around 1735 cm-1.
Other Functional Groups: O-H, N-H, and C-O stretches can help confirm the presence of specific functional groups.


Example IR Spectra
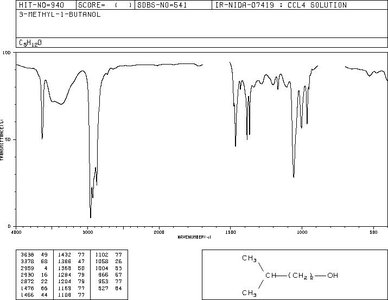
3-Methyl-1-butanol: Shows O-H and C-O stretches.
Acetic Acid: Shows broad O-H and strong C=O stretches.
Isopentyl Acetate (Ester): Shows strong C=O and C-O stretches, absence of O-H stretch.
Nuclear Magnetic Resonance (NMR) Spectroscopy
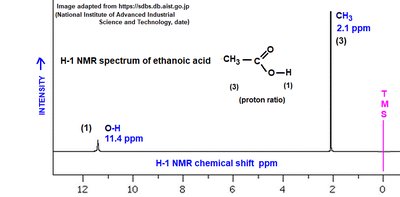
1H NMR spectroscopy provides information about the hydrogen environments in a molecule. Chemical shifts, splitting patterns, and integration help identify the structure of the compound.
Alcohols: O-H proton appears as a broad singlet, alkyl protons show characteristic splitting.
Carboxylic Acids: O-H proton appears downfield (10-12 ppm), methyl protons upfield (2-3 ppm).
Esters: Alkoxy and alkyl protons show distinct chemical shifts and splitting patterns.
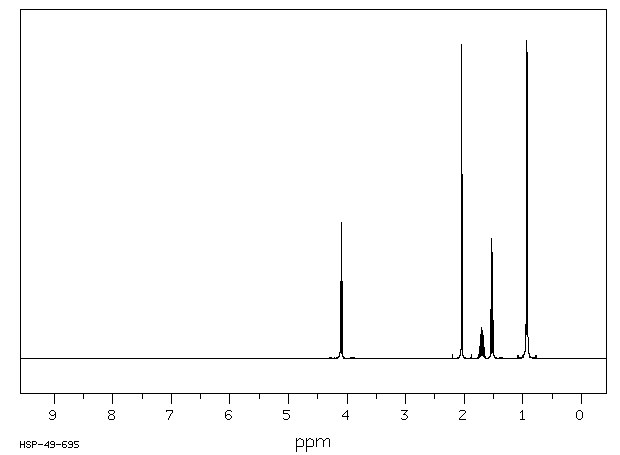
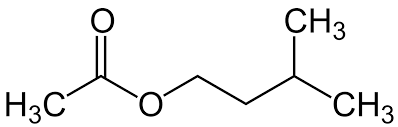
Example: Synthesis of Isopentyl Acetate (Banana Oil)
Reaction Equation
Isopentyl acetate is synthesized from acetic acid and 3-methyl-1-butanol via Fischer esterification:
Procedure Summary
Mix acetic acid and 3-methyl-1-butanol in a round-bottom flask with boiling stones.
Add concentrated sulfuric acid as catalyst.
Assemble reflux apparatus and heat for 25 minutes.
Cool, separate layers, wash, and dry the organic layer.
Characterize the product by IR and NMR spectroscopy.
Key Laboratory Skills and Calculations
Determine the limiting reagent and calculate theoretical and percent yield.
Practice safe handling and disposal of chemicals.
Maintain a detailed scientific notebook and communicate results effectively.
Summary Table: Spectroscopic Features of Reactants and Product
Compound | Key IR Peaks (cm-1) | Key 1H NMR Shifts (ppm) |
|---|---|---|
3-Methyl-1-butanol | O-H (broad, ~3300), C-O (~1050) | 0.9-3.5 (alkyl, O-H) |
Acetic Acid | O-H (broad, ~3000), C=O (~1710) | 11.4 (O-H), 2.1 (CH3) |
Isopentyl Acetate | C=O (~1740), C-O (~1200, 1050) | 0.9-4.2 (alkyl, O-CH2) |
Additional info: This guide covers the synthesis, mechanism, and spectroscopic identification of esters, focusing on isopentyl acetate as a representative example. It integrates laboratory safety, experimental setup, and data analysis for a comprehensive understanding of ester chemistry in the undergraduate organic laboratory.
