 Back
BackSystematic Naming and Identification of Organic Compounds
Study Guide - Smart Notes

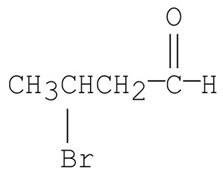
Q1. What is the following compound’s systematic name?
Background
Topic: IUPAC Nomenclature of Aldehydes and Substituted Alkanes
This question tests your ability to apply IUPAC rules to name an aldehyde with a halogen substituent. You need to identify the parent chain, locate the substituents, and assign the correct locants and prefixes.
Key Terms and Concepts:
Aldehyde: A compound containing a carbonyl group bonded to at least one hydrogen atom (–CHO).
Substituent: An atom or group of atoms attached to the main carbon chain.
Locant: The number assigned to a carbon atom in the chain to indicate the position of a substituent.
Parent Chain: The longest continuous chain containing the functional group (aldehyde in this case).
Step-by-Step Guidance
Identify the longest carbon chain containing the aldehyde group. Remember, the aldehyde carbon is always carbon 1.
Number the chain starting from the aldehyde carbon (C=O), so the functional group gets the lowest possible number.
Locate the position of the bromine substituent and assign its locant based on the numbering from step 2.
Combine the substituent name and locant with the parent chain name, ending with "al" for an aldehyde.
Try solving on your own before revealing the answer!
Final Answer: 3-bromobutanal
The parent chain is butanal (4 carbons, aldehyde at C1). Bromine is on carbon 3, so the name is 3-bromobutanal.
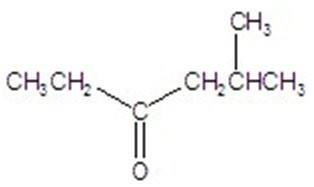
Q2. What is the following compound’s systematic name?
Background
Topic: IUPAC Nomenclature of Ketones and Branched Alkanes
This question tests your ability to name a ketone with branching. You must identify the parent chain, the position of the carbonyl group, and the position and identity of any alkyl substituents.
Key Terms and Concepts:
Ketone: A compound with a carbonyl group (C=O) bonded to two carbon atoms.
Parent Chain: The longest chain containing the carbonyl group.
Numbering: Number the chain so the carbonyl carbon gets the lowest possible number.
Substituent: An alkyl group attached to the main chain.
Step-by-Step Guidance
Identify the longest carbon chain containing the carbonyl group (ketone).
Number the chain from the end nearest the carbonyl group to give it the lowest possible number.
Identify and locate the position of the methyl substituent.
Combine the substituent name and locant with the parent chain name, ending with "one" for a ketone.
Try solving on your own before revealing the answer!
Final Answer: 5-methyl-3-hexanone
The parent chain is hexanone (6 carbons, ketone at C3). The methyl group is at C5, so the name is 5-methyl-3-hexanone.
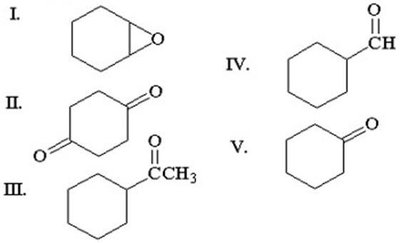
Q3. Which of the following compounds is cyclohexanone?
Background
Topic: Identification of Cyclic Ketones
This question tests your ability to recognize the structure of cyclohexanone among several cyclic compounds. Cyclohexanone is a six-membered ring with a single ketone group.
Key Terms and Concepts:
Cyclohexanone: A six-membered ring (cyclohexane) with a ketone group (C=O) at one position.
Ketone: A carbonyl group bonded to two carbons.
Ring Size: Count the number of carbons in the ring to confirm it is a cyclohexane derivative.
Step-by-Step Guidance
Examine each structure and count the number of carbons in the ring.
Identify which structure has a six-membered ring.
Check for the presence of a single ketone group (C=O) attached to the ring.
Eliminate any structures with additional substituents or different ring sizes.
Try solving on your own before revealing the answer!
Final Answer: Structure II
Structure II is a six-membered ring with a single ketone group, which matches cyclohexanone.
