 Back
BackChapter 1: Foundations of Physics – Scientific Method, Symmetry, Matter, Time, Representations, and Quantitative Tools
Study Guide - Smart Notes

Chapter 1: Foundations
1.1 The Scientific Method
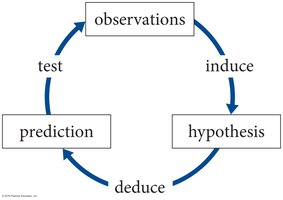
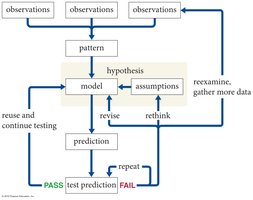
The scientific method is a systematic, iterative process used to discover and validate the fundamental laws governing physical phenomena. It emphasizes observation, hypothesis formation, prediction, and experimental testing, forming the basis for scientific inquiry in physics.
Observation: Careful examination of phenomena (e.g., a rock falling).
Hypothesis: Proposed explanation for the observed phenomena.
Prediction: Using the hypothesis to forecast outcomes in similar situations.
Experiment/Test: Conducting experiments to validate or refute the hypothesis.
Iterative Process: The method repeats, refining hypotheses and models based on results.
Laws vs. Theories: Law describes what happens; Theory explains why, based on underlying principles.
Models: Simplified conceptual representations used to understand and predict phenomena.
Science is Tentative: All theories and laws are subject to revision with new evidence.
Example: If a music player fails to play, hypothesize the battery is dead. Predict replacing the battery will restore function. Test and observe outcomes to confirm or refute the hypothesis.
Additional info: The scientific method is foundational for all physics topics, from classical mechanics to quantum physics.
1.1 Reasoning in Physics
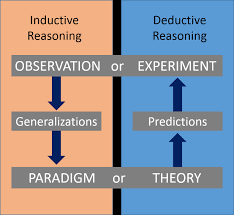
Physics relies on both deductive and inductive reasoning to advance understanding. Deductive reasoning applies general principles to specific cases, while inductive reasoning derives general rules from specific observations.
Deductive Reasoning: From general principles to specific predictions.
Inductive Reasoning: From specific observations to general principles.
Both are essential: Scientists use both approaches to develop and refine theories.
1.2 Symmetry
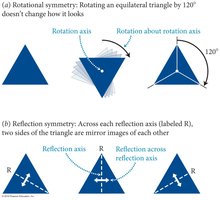
Symmetry is a fundamental concept in physics, describing situations where certain operations leave an object or phenomenon unchanged. Symmetry applies to both physical objects and the laws governing them, providing order, beauty, and harmony.
Definition: An object exhibits symmetry if certain operations (e.g., rotation, translation, reflection) do not change its appearance.
Types of Symmetry:
Translational Symmetry: Moving an object or experiment yields the same results.
Rotational Symmetry: Rotating an object by a certain angle leaves it unchanged.
Reflection Symmetry: Mirroring an object across an axis leaves it unchanged.
Physical Laws: Laws must possess the same symmetries as the phenomena they describe.
Abstract Symmetries: Charge symmetry, parity symmetry, unitary transformations.

Example: A chessboard pattern looks the same from any square, demonstrating translational symmetry.
Additional info: Symmetry principles are central to modern physics, including conservation laws and quantum mechanics.
1.3 Matter and the Universe
The universe consists of matter and energy, combined with space and time. Physicists use physical quantities and units to describe and measure properties of the universe.
Matter: Composed of atoms, which consist of protons, neutrons, and electrons.
Energy: The capacity to do work or cause change.
Space and Time: The framework in which all events occur.
Physical Quantities: Measurable properties such as length, mass, and time.
Scale of Length: From atomic diameters ( m) to cosmic distances.
Example: Atoms have diameters of about m; their nuclei are about m.
1.4 Time and Change
Time is a fundamental quantity in physics, flowing in one direction and establishing causality between events. The standard unit of time is the second.
Arrow of Time: Time progresses from past to future, dictating the sequence of events.
Principle of Causality: Event A causes event B; all observers see A happen first.
Standard Unit: The second (s), defined by the frequency of radiation from cesium atoms.
Example: Aging, motion, and collisions are all affected by the passage of time.
1.5 Representations
Visual representations are essential tools in physics for organizing information, understanding relationships, and solving problems.
Types: Pictures, sketches, diagrams, graphs.
Purpose: Clarify complex phenomena, aid in problem-solving, and communicate scientific models.
Example: Diagram of a person pulling a cart, elevator motion, or swings in a playground.
1.6 Physical Quantities and Units
Physical quantities are expressed as a number multiplied by a unit. The SI system defines seven base units, and powers of ten notation and metric prefixes are used for convenience.
SI Base Units: Meter (m), kilogram (kg), second (s), ampere (A), kelvin (K), mole (mol), candela (cd).
Metric Prefixes: Used to express large and small numbers (e.g., kilo-, milli-, micro-).
Density: Number density (), mass density ().
Unit Conversion: Use ratios equal to one for converting between units.
Example: 1 mol = atoms (Avogadro's number).
1.7 Significant Digits
Significant digits indicate the precision of a measurement. Rules for counting significant digits help ensure accuracy in calculations.
Precision: Consistency of repeated measurements.
Accuracy: Closeness to the true value.
Rules:
All nonzero digits are significant.
Zeros between nonzero digits are significant.
Leading zeros are not significant.
Trailing zeros after a decimal point are significant.
Trailing zeros without a decimal point are ambiguous; use scientific notation.
Example: 21.3 has three significant digits; 0.037 has two.
Summary Table: SI Base Units
Quantity | Unit | Symbol |
|---|---|---|
Length | meter | m |
Mass | kilogram | kg |
Time | second | s |
Electric current | ampere | A |
Temperature | kelvin | K |
Amount of substance | mole | mol |
Luminous intensity | candela | cd |
Key Equations
Number Density:
Mass Density:
Unit Conversion Example:
Take Home Message
Physics is the study of the fundamental laws governing the universe, using the scientific method, symmetry, precise measurement, and visual representations to build models and theories. Mastery of these foundational concepts is essential for success in all areas of physics.
