 Back
BackChapter 39: Wave Functions and Uncertainty – Study Notes
Study Guide - Smart Notes

Wave Functions and Quantum Mechanics
Introduction to Quantum Mechanics
Quantum mechanics is the branch of physics that describes the behavior of light and matter at the atomic and subatomic scale. Unlike classical mechanics, quantum mechanics relies on probabilistic predictions and introduces concepts such as wave functions and uncertainty. - Quantum mechanics explains properties of atoms and nuclei. - Its predictions are experimentally verified with high precision.
The Wave Function
The wave function, denoted as ψ(x), is a fundamental concept in quantum mechanics. It is a mathematical function used to describe the probability of finding a particle at a particular position. - ψ(x) is an oscillatory function, but nothing is physically waving. - The square of the wave function, |ψ(x)|2, gives the probability density. - Particles are most likely found near the maxima of the probability density. 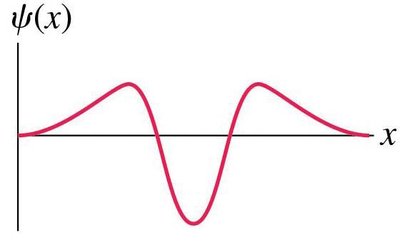
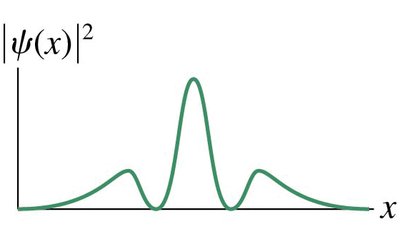
Probability in Quantum Mechanics
Quantum mechanics deals with probabilities rather than certainties. The probability of finding a particle in a region is determined by the wave function. - For N particles, if NA are detected in region A, then the probability is - The sum of probabilities for all possible outcomes must equal 1. - The expected value for N trials is 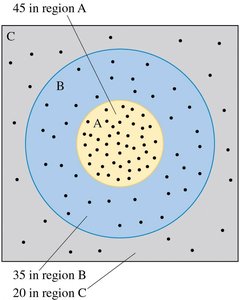
Wave-Particle Duality and the Double-Slit Experiment
Interference and Wave-Particle Duality
The double-slit experiment demonstrates the dual nature of light and matter. Both exhibit interference patterns, a wave-like property, but their detection is particle-like. - Photons, electrons, and neutrons all show interference fringes when passing through slits. - Arrival at the detector is a particle event, but the pattern is explained by wave interference. 

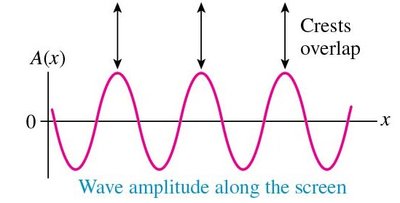
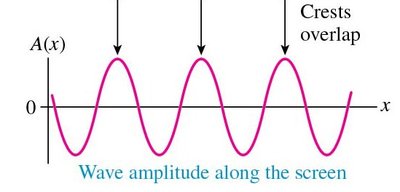
Mathematical Description of Double-Slit Interference
The amplitude and intensity of waves in the double-slit experiment are described mathematically: - Amplitude at the screen: - Intensity is proportional to the square of amplitude: 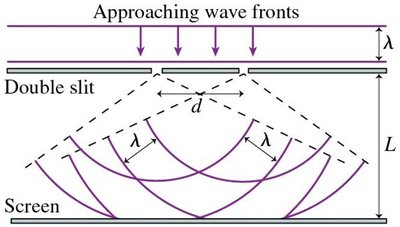
Probability Density and Normalization
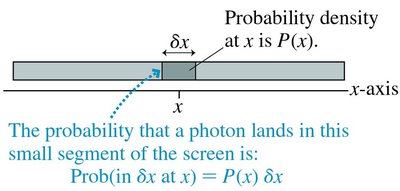
Probability Density
The probability density, P(x), is analogous to linear mass density in classical physics. It describes the likelihood of finding a particle at position x. - - The probability that a particle lands in a small segment δx at x is 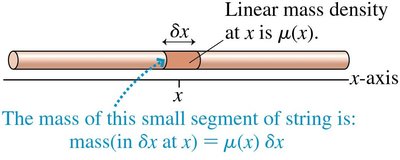
Normalization of the Wave Function
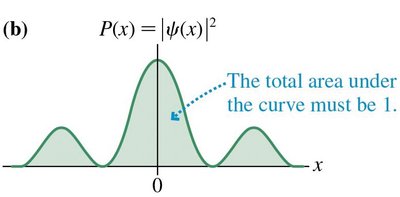
For the probability interpretation to be valid, the wave function must be normalized. - The total probability of finding the particle somewhere is 1: - The probability of finding the particle in a range is 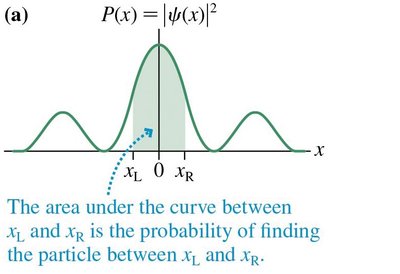
Wave Packets and Superposition
Wave Packets
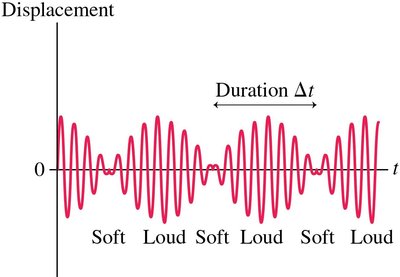
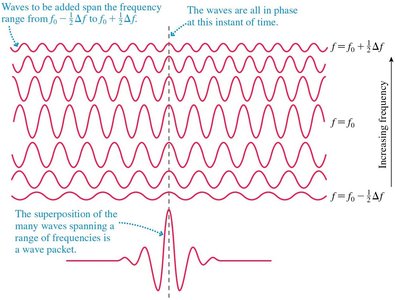
A wave packet is a localized wave formed by the superposition of many component waves. It exhibits both particle-like and wave-like characteristics. - Localization is particle-like; oscillations are wave-like. - Superposition of waves with similar frequencies creates a beat pattern. - The duration of a beat is , and the frequency range is - Fundamental relationship: 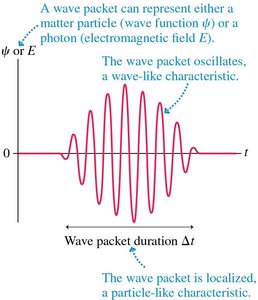
Heisenberg Uncertainty Principle
Statement and Implications
The Heisenberg uncertainty principle states that it is impossible to simultaneously know the exact position and momentum of a particle. - - The more precisely one quantity is known, the less precisely the other can be known. - This principle arises from the wave-like nature of matter.
Mathematical Derivation
- For a particle with velocity and de Broglie wavelength : - The spatial extent of the wave packet is , and the momentum uncertainty is . - The uncertainty principle is derived from the relationship between frequency and time: - For matter waves:
Examples and Applications
Example: Probability Density Calculation
In an experiment, 6000 out of 600,000 photons are detected in a 1.0-mm-wide strip at cm. - Probability: - Probability density:
Example: Uncertainty of an Electron
If an electron is confined to a 0.1-nm-wide region, the uncertainty in its velocity is: - - For nm, m/s
Summary Table: Key Quantum Concepts
Concept | Definition | Equation |
|---|---|---|
Wave Function | Describes probability amplitude | |
Probability Density | Probability per unit length | |
Normalization | Total probability is 1 | |
Uncertainty Principle | Limits precision of position and momentum | |
Wave Packet | Localized superposition of waves |
Conclusion
Quantum mechanics introduces a probabilistic framework for understanding the behavior of particles at the atomic scale. The wave function, probability density, and uncertainty principle are central to this framework, providing insight into the dual nature of matter and the fundamental limits of measurement.
