 Back
BackCoulomb’s Law and Electric Charge: Study Notes for Physics II
Study Guide - Smart Notes

Coulomb’s Law and Electric Charge
Introduction to Electric Charge and Forces
Electric charge is a fundamental property of matter that gives rise to electric forces and interactions. There are two types of electric charge: positive and negative. The study of electric forces, especially those between stationary charges, is known as electrostatics. Devices and phenomena in everyday life, such as computers and telecommunications, rely on the principles of electromagnetism.
Electric charge can be transferred between objects.
Objects can be electrically neutral, positively charged, or negatively charged.
Like charges repel; opposite charges attract.
Classification of Materials by Electrical Properties
Materials are classified based on their ability to allow the movement of electric charge:
Conductors: Materials (e.g., metals) with free electrons that can move easily.
Insulators: Materials (e.g., rubber, glass) where electrons are not free to move.
Semiconductors: Intermediate materials (e.g., silicon, germanium) used in electronics.
Superconductors: Materials (e.g., mercury) that allow charge to move without resistance.
Atomic Structure and Electric Charge
The electrical properties of materials are determined by the structure of atoms. Atoms consist of a nucleus (protons and neutrons) surrounded by electrons. In conductors, some electrons become conduction electrons and are free to move.
Protons: Positive charge
Electrons: Negative charge
Neutrons: Electrically neutral

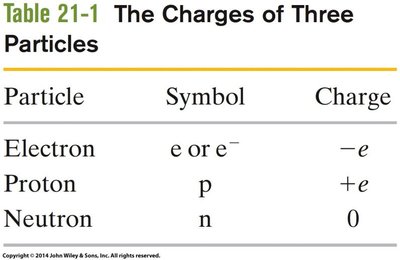
Particle | Symbol | Charge |
|---|---|---|
Electron | e or e- | -e |
Proton | p | +e |
Neutron | n | 0 |
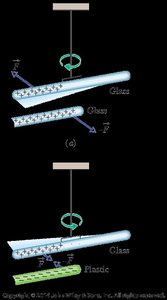
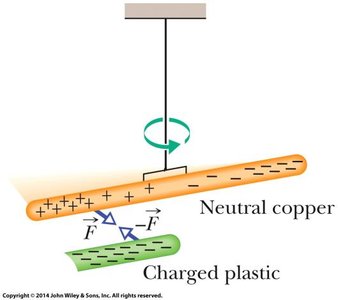
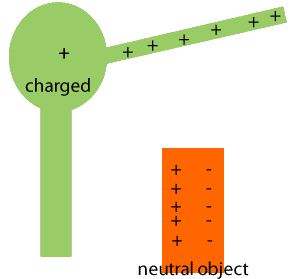
Induced Charge and Electrical Isolation
When a neutral conductor is brought near a charged object, charges within the conductor redistribute, creating an induced charge. Electrical isolation prevents charge transfer between the object and its surroundings.
Induced charge: Redistribution of charges in a neutral object due to a nearby charged object.
Grounding: Connecting an object to the earth to allow charge transfer.
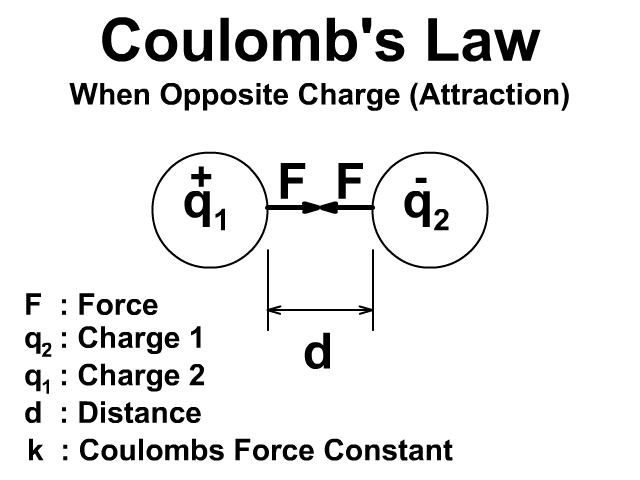
Coulomb’s Law
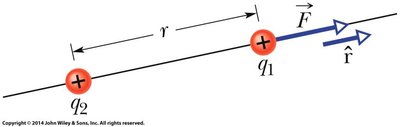
Statement and Formula
Coulomb’s Law describes the electrostatic force between two point charges. The force is proportional to the product of the charges and inversely proportional to the square of the distance between them.
Electrostatic force: The force between two stationary charges.
Direction: Repulsive for like charges, attractive for opposite charges.
Mathematical Expression:
where is the permittivity of free space, and is the Coulomb constant.
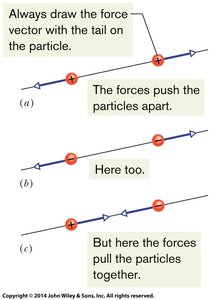
Vector Nature of Electrostatic Force
The electrostatic force is a vector quantity. If multiple forces act on a particle, the net force is the vector sum of individual forces.
Draw force vectors with the tail anchored on the particle.
For two charges, the force acts along the line joining them.
Worked Example: Two Charged Particles
Given two charges C and C separated by m, the magnitude of the force is:
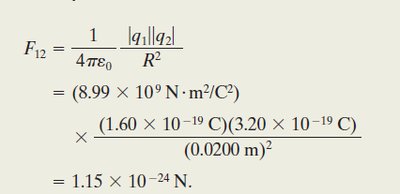
Worked Example: Three Charged Particles
When a third particle with charge C is placed between and , the net force on $q_1$ is:
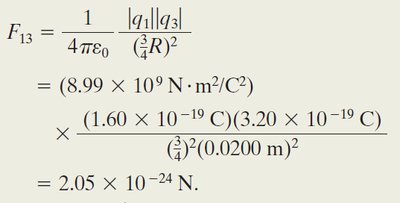
The net force is:
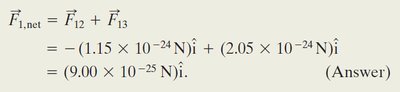
Quantization and Conservation of Charge
Quantization of Charge
Electric charge is quantized, meaning it exists in discrete units. The elementary charge is the smallest unit, and any charge can be written as , where is an integer.
Elementary charge: C
All observable charges are integer multiples of .
Example: Mutual Repulsion in a Nucleus
Calculate the repulsive force between two protons separated by m:
Conservation of Charge
The net electric charge of any isolated system is always conserved. Charge can be transferred but not created or destroyed.
Rubbing objects transfers charge, but the total charge remains constant.
Pair production and annihilation processes always produce equal and opposite charges.
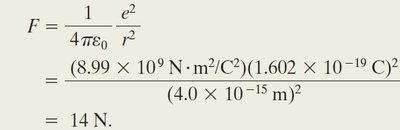
Summary Table: Charges of Fundamental Particles
Particle | Symbol | Charge |
|---|---|---|
Electron | e or e- | -e |
Proton | p | +e |
Neutron | n | 0 |

Key Points and Summary
Particles with the same sign of charge repel; opposite signs attract.
Electrostatic force is described by Coulomb’s Law.
Conductors allow free movement of electrons; insulators do not.
Electric charge is quantized and conserved.
