 Back
BackElectric Charge, Coulomb’s Law, and Vector Quantities: Study Notes for College Physics
Study Guide - Smart Notes

Four Fundamental Forces
Overview of Fundamental Forces
The universe is governed by four fundamental forces, each responsible for different interactions between particles and objects. Understanding these forces is essential for studying physics at all scales.
Gravity: The force of attraction between masses. Responsible for planetary motion and structure of the universe.
Electromagnetism: The force between charged particles. Governs electricity, magnetism, and light.
Strong Force: The force that binds protons and neutrons in the nucleus. Strongest force at subatomic scales.
Weak Force: Responsible for radioactive decay and certain nuclear reactions.



Electric Charge
Properties and Types of Electric Charge
Electric charge is a fundamental property of matter, determining how particles interact via electromagnetic forces. Charges can be positive or negative, and their interactions are governed by specific rules.
Positive Charge: Typically carried by protons.
Negative Charge: Typically carried by electrons.
Same sign charges repel; opposite sign charges attract.
Electrically neutral objects contain equal amounts of positive and negative charges.
Net charge of a system: The algebraic sum of all charges present.
Conservation of charge: The net charge of a closed system never changes.

Coulomb’s Law
Definition and Experimental Basis
Coulomb’s Law describes the force between two point charges. Charles Coulomb established this relationship in 1785 using a torsion balance, showing that the force depends on the magnitude of the charges and the distance between them.
Force is proportional to the product of the charges and inversely proportional to the square of the distance.
Experimental apparatus: Torsion balance with metal spheres.

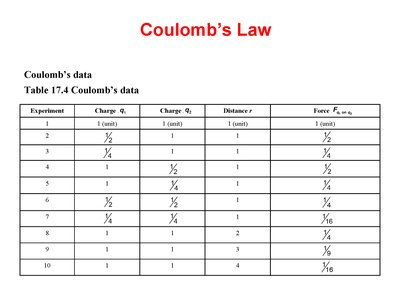
Coulomb’s Law Data Table
The table below summarizes experimental data showing how force varies with charge and distance.
Experiment | Charge Q1 | Charge Q2 | Distance r | Force Fcoul |
|---|---|---|---|---|
1 | 1 | 1 | 1 | 1 |
2 | 1/2 | 1 | 1 | 1/2 |
3 | 1/4 | 1 | 1 | 1/4 |
4 | 1 | 1/2 | 1 | 1/2 |
5 | 1 | 1/4 | 1 | 1/4 |
6 | 1 | 1 | 2 | 1/4 |
7 | 1 | 1 | 3 | 1/9 |
8 | 1 | 1 | 4 | 1/16 |
9 | 1 | 1 | 1 | 1 |
10 | 1 | 1 | 4 | 1/16 |
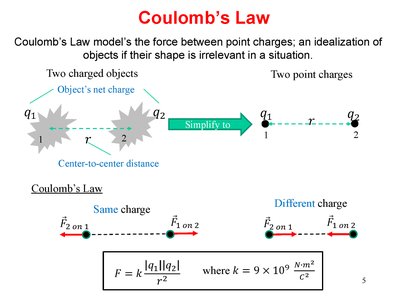
Mathematical Formulation
Coulomb’s Law models the force between point charges as:
Formula: , where
Direction: Repulsive for like charges, attractive for opposite charges.
Application: Force on Electron in Hydrogen Atom
Coulomb’s Law can be used to calculate the force between a proton and an electron in a hydrogen atom:
Formula:
Permittivity of free space:
Example calculation: (to the left)

Comparison: Coulomb Force vs. Gravitational Force
The electrostatic force between two charges is much stronger than the gravitational force between two masses at atomic scales.
Electrostatic force:
Gravitational force:
Ratio:

Worked Example: Electrostatic Force Calculation
Calculate the force between two positive charges (2 μC and 7 μC) separated by 20 cm:
Formula:
Calculation:

Vector and Scalar Quantities
Definitions and Examples
Physical quantities can be classified as scalars or vectors. Scalars have only magnitude, while vectors have both magnitude and direction.
Scalar: Example: temperature, mass, energy.
Vector: Example: wind velocity, force, displacement.

Quiz: Force and Charge Classification
Are force and charge scalars or vectors?
Force: Vector (has magnitude and direction)
Charge: Scalar (has magnitude only)

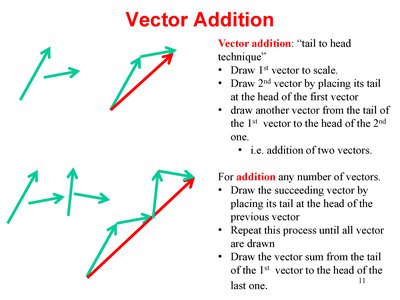
Vector Addition and Subtraction
Vector Addition: Tail-to-Head Technique
Vectors are added using the tail-to-head method. This technique is essential for combining forces and other vector quantities.
Draw the first vector to scale.
Draw the second vector by placing its tail at the head of the first vector.
Continue for additional vectors.
The resultant vector is drawn from the tail of the first to the head of the last.

Vector Subtraction
Vector subtraction uses the same technique, but the subtracted vector is reversed in direction.
Formula:

Principle of Superposition
Superposition of Forces
When multiple point charges are present, the total force on any one charge is the vector sum of the individual forces exerted by each charge.
Formula:
Forces are added using vector addition techniques.
Worked Example: Net Force on a Charge
Three positive charges are located along a line. Calculate the net force on the 0.02-C charge by the other two charges:
Formula:
Calculation: (to the left)
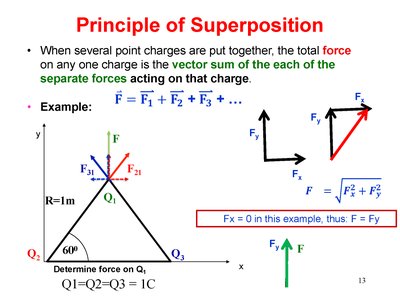
Worked Example: Triangle Arrangement of Charges
Calculate the force on a +2 μC charge due to two other charges (+7 μC and -3.5 μC) arranged in a triangle. Forces must be added as vectors.
Calculate force from each charge using Coulomb’s Law.
Add forces using vector addition.


Adding Vectors: Components
Decompose forces into x and y components for addition:
Component formulas: ,
Add x and y components separately.
Resultant force:


Problem Solving Strategies
Approach for Physics Problems
Effective problem solving in physics requires a systematic approach:
Draw a clear force diagram.
Use consistent units (meter, Coulomb, Newton).
Remember that force is a vector.
Look for symmetry in the arrangement of charges.

Summary
This study guide covers the fundamental concepts of electric charge, Coulomb’s Law, and vector quantities, providing definitions, formulas, examples, and problem-solving strategies essential for college-level physics.
