 Back
BackElectric Charges and Forces: Foundations of Electrostatics
Study Guide - Smart Notes

Electric Charge and Atomic Structure
Subatomic Particles and Charge
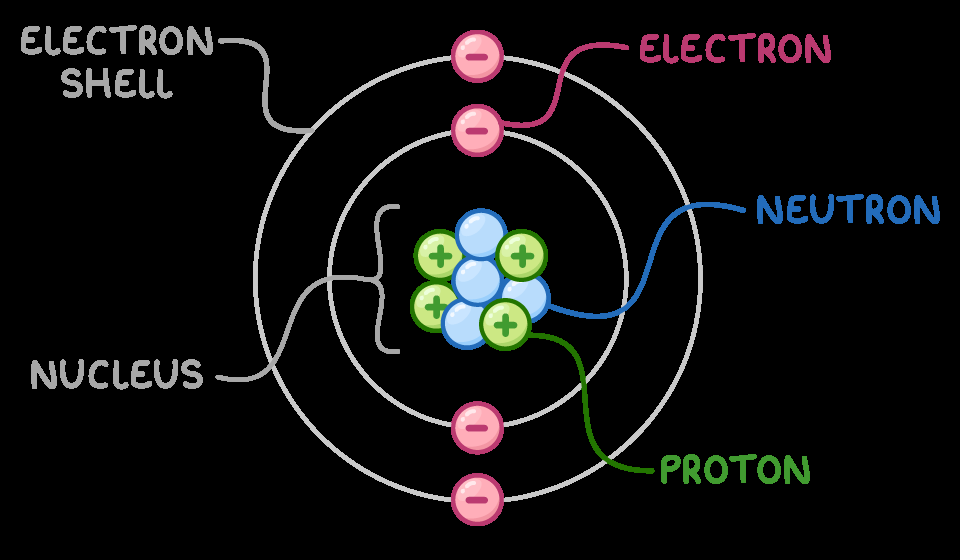
Electric charge is a fundamental property of matter, carried by subatomic particles. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. Protons carry a positive charge, electrons carry a negative charge, and neutrons are neutral. The arrangement and movement of these particles determine the electrical properties of matter.
Protons: Positively charged, located in the nucleus.
Electrons: Negatively charged, orbit the nucleus and are easily transferred.
Neutrons: Electrically neutral, found in the nucleus.
Charge Quantization: Electric charge exists in discrete units, with the elementary charge C.
Conservation of Charge: The total electric charge in an isolated system remains constant.
Types of Materials: Conductors and Insulators
Electrical Properties of Materials
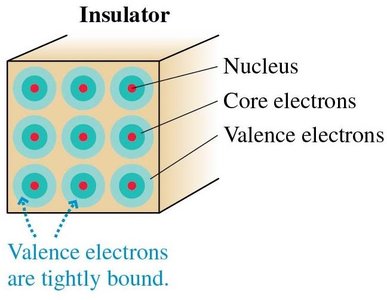
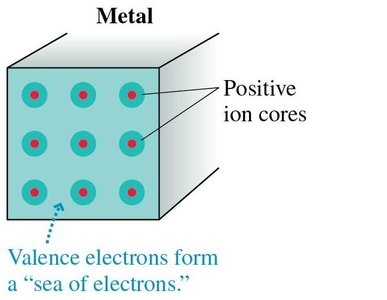
Materials are classified based on how easily charges move within them. This distinction is crucial for understanding how objects become charged and how electric forces act in different environments.
Conductors: Materials (like metals) where electrons are free to move throughout the material, allowing charge to flow easily.
Insulators: Materials where electrons are tightly bound to atoms and do not move freely, so charge does not flow easily.
Charging Methods: Friction, Contact, and Induction
How Objects Acquire Charge
Objects can become electrically charged through three main processes: friction, contact, and induction. Each method involves the transfer or redistribution of electrons.
Friction: Rubbing two different materials can transfer electrons from one to the other, leaving one object negatively charged and the other positively charged.
Contact: Touching a charged object to a neutral one can transfer charge directly.
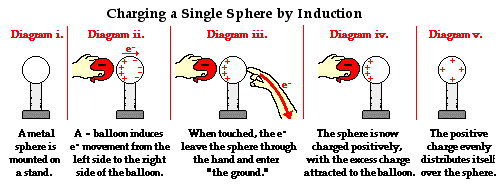
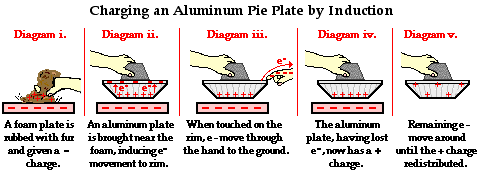
Induction: Bringing a charged object near a conductor can cause a redistribution of charges within the conductor, even without direct contact.


Coulomb’s Law: The Force Between Charges
Electrostatic Force and Its Calculation
Coulomb’s Law quantifies the electrostatic force between two point charges. The force is directly proportional to the product of the charges and inversely proportional to the square of the distance between them.
Formula: where is the force (N), and are the charges (C), is the separation (m), and N·m2/C2 is Coulomb’s constant.
Direction: Like charges repel, unlike charges attract. The force acts along the line joining the charges.
Vector Nature: The force has both magnitude and direction. For multiple charges, use vector addition (superposition principle).
Example: If the distance between two charges is doubled, the force becomes one-fourth as large. If the magnitude of one charge is doubled, the force doubles.
The Electric Field
Definition and Properties
The electric field is a region of space around a charged object where other charges experience a force. It is defined as the force per unit positive test charge.
Formula: where is the electric field (N/C), is the force on a test charge .
Point Charge Field: The direction is away from positive charges and toward negative charges.
Units: N/C or V/m.
Example: The electric field at a distance from a proton is extremely strong at atomic scales, influencing atomic structure and chemical behavior.
Motion of Charged Particles in Electric Fields
Kinematics in a Uniform Electric Field
A charged particle in a uniform electric field experiences a constant force, resulting in constant acceleration. The equations of motion are analogous to those for objects under gravity.
Force:
Acceleration:
Kinematic Equations:
Example: An electron entering a region with a uniform electric field perpendicular to its velocity will follow a parabolic path, similar to projectile motion under gravity.
Electric Dipoles
Definition, Torque, and Potential Energy
An electric dipole consists of two equal and opposite charges separated by a fixed distance. Dipoles experience torque and potential energy changes in external electric fields.
Dipole Moment: , points from negative to positive charge.
Torque: or
Potential Energy:
Work Done:
Example: Water molecules are natural dipoles, which explains their strong interactions with electric fields and their effectiveness as solvents for ionic compounds.
Summary Table: Key Concepts in Electrostatics
Concept | Definition/Formula | Units |
|---|---|---|
Elementary Charge | Coulombs (C) | |
Coulomb’s Law | Newtons (N) | |
Electric Field | N/C or V/m | |
Dipole Moment | C·m | |
Torque on Dipole | N·m | |
Potential Energy (Dipole) | Joules (J) |
