 Back
BackWeek 1 Lec. 2
Study Guide - Smart Notes

Electric Charge and Atomic Structure
Definition and Properties of Charge
Electric charge is a fundamental property of matter that causes it to experience a force when placed in an electric or magnetic field. Charges are either positive or negative, and like charges repel while unlike charges attract. Atomic structure consists of protons (positive), electrons (negative), and neutrons (neutral), with charge quantization in units of the elementary charge e.
Key Point: The unit of charge is the coulomb (C).
Key Point: Protons carry +e, electrons carry -e.
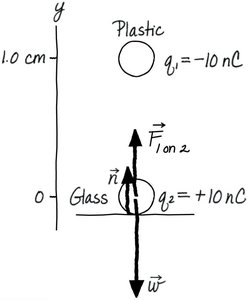
Example: A glass bead with +10 nC and a plastic sphere with -10 nC.
Polarization and Charging Methods
Charging by Contact and Induction
Objects can be charged by direct contact (transfer of electrons) or by induction (redistribution of charges without direct contact). Polarization occurs when charges within an object rearrange due to an external electric field.
Key Point: Contact transfers charge; induction redistributes charge.
Example: Bringing a charged rod near a neutral object causes polarization.
Coulomb’s Law
Fundamental Law of Electrostatics
Coulomb’s Law quantifies the force between two point charges. The force is proportional to the product of the charges and inversely proportional to the square of the distance between them. The direction of the force depends on the sign of the charges.
Key Point: The force is attractive for opposite charges and repulsive for like charges.
Equation: where
Example: Calculating the force between a -10 nC plastic sphere and a +10 nC glass bead separated by 1.0 cm.
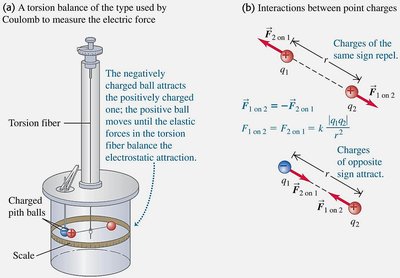
Experimental Basis of Coulomb’s Law
Coulomb used a torsion balance to measure the electric force between charged spheres, confirming the inverse-square law.
Key Point: The torsion balance measures the force by the rotation caused by charge interaction.

Worked Example: Forces Between Point Charges
Free-Body Diagram and Force Comparison
Consider a plastic sphere (q1 = -10 nC) held above a glass bead (q2 = +10 nC, mass = 15 mg) at rest on a table. The forces acting on the bead are the electric force, its weight, and the normal force from the table.
Key Point: The bead will rise if the electric force exceeds its weight.
Equation:
Example: Since is much greater than , the bead leaps upward.
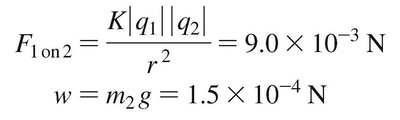
Superposition Principle
Vector Addition of Forces
When multiple charges are present, the net force on any charge is the vector sum of the forces exerted by each individual charge. This is known as the superposition principle.
Key Point: Forces from each charge are calculated using Coulomb’s Law and then added vectorially.
Equation:
Example: Three point charges arranged in space; calculate the resultant force on one using vector addition.
Electric Field Concept
Definition and Use
The electric field is a region of space around a charged particle where other charges experience a force. The field is defined as the force per unit charge and is a vector quantity.
Key Point: The electric field emanates from charges and is sensed by other charges.
Equation:
Example: A positive test charge placed near a negative charge experiences an attractive force.
Dipole Forces
Dipole Interaction with External Charges
A dipole consists of two equal and opposite charges connected by a rigid rod. When placed near another charge, the net force depends on the relative positions and magnitudes.
Key Point: The closer charge exerts a stronger force, resulting in net attraction or repulsion.
Equation:
Example: A dipole near a large negative charge experiences a net force to the left if the positive charge is closer.
Summary Table: Coulomb’s Law and Superposition
Concept | Equation | Key Feature |
|---|---|---|
Coulomb’s Law | Force between two point charges | |
Superposition Principle | Vector sum of all forces | |
Electric Field | Force per unit charge |
Additional info: The notes cover topics from Ch 21: Electric Charge and Electric Field, and Ch 22: Gauss' Law (introductory concepts), as well as foundational vector concepts from Ch 01.
