 Back
BackElectrostatics: Fundamentals of Electric Charge and Fields
Study Guide - Smart Notes

Electrostatics
Introduction to Electrostatics
Electrostatics is the branch of physics that studies electric charges at rest, the forces between them, and the fields and potentials they produce. It is foundational for understanding electricity and magnetism, and underpins many phenomena in both natural and technological contexts.
Electricity refers to a wide range of electrical phenomena, such as lightning, sparks, and the forces that hold atoms together.
Electrostatics specifically involves stationary electric charges, the forces between them, and their behavior in materials.
Examples include static cling, lightning, and the operation of devices like the Van de Graaff generator.
Electric Charge
Electric charge is a fundamental property of matter that causes it to experience a force when placed in an electric and magnetic field. There are two types of charge: positive and negative.
Charge is carried by protons (+) and electrons (−).
Central rule: Opposite charges attract; like charges repel.
Charge can be neutralized by combining equal amounts of positive and negative charge.
Charge is conserved: it cannot be created or destroyed, only transferred.
Atomic Structure and Charge Quantization
Atoms are composed of a nucleus (protons and neutrons) surrounded by electrons. The properties of these subatomic particles determine the electrical behavior of matter.
Protons: positive charge (+e), located in the nucleus.
Neutrons: no charge, slightly more massive than protons, located in the nucleus.
Electrons: negative charge (−e), much less massive than protons, orbit the nucleus.
Atoms are usually electrically neutral (equal numbers of protons and electrons).
Charge is quantized: the smallest unit is the elementary charge, C.
Unit of charge: Coulomb (C).
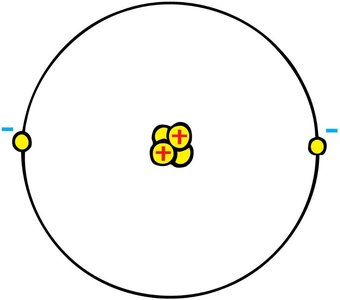
Coulomb's Law
Coulomb's Law describes the force between two point charges. It is analogous to Newton's law of gravitation but applies to electric charges.
The force is proportional to the product of the charges and inversely proportional to the square of the distance between them.
Like charges repel; opposite charges attract.
where N·m2/C2, and are the charges, and is the distance between them.
Conductors, Insulators, Semiconductors, and Superconductors
Materials can be classified by how easily they allow electric charges to move.
Conductors: Materials (e.g., metals) where electrons are free to move, allowing electric current to flow easily.
Insulators: Materials (e.g., rubber, glass) where electrons are tightly bound and do not move freely.
Semiconductors: Materials (e.g., silicon, germanium) that can behave as insulators or conductors depending on impurities (doping) or external conditions.
Superconductors: Materials that, at very low temperatures, have zero electrical resistance and allow current to flow indefinitely without energy loss.




Charging Methods
Objects can be charged by different methods, including friction, contact, and induction.
Friction: Rubbing two materials together transfers electrons from one to the other (e.g., combing hair, rubbing a balloon).
Contact: Touching a charged object to a neutral one transfers charge.
Induction: Bringing a charged object near a conductor causes a redistribution of charges without direct contact.


Polarization and Electric Dipoles
When a charged object is brought near a neutral object, it can cause the charges within the neutral object to rearrange, a process called polarization. Some molecules, like water, are naturally polarized and are called electric dipoles.
Polarization results in one side of the molecule or atom being slightly more positive or negative than the other.
Electric dipoles have a permanent separation of charge.
Electric Field
An electric field is a region around a charged object where other charges experience a force. The field is a vector field, meaning it has both magnitude and direction.
The electric field () at a point is defined as the force () per unit charge ():
Units: Newtons per Coulomb (N/C).
Field lines point away from positive charges and toward negative charges.

Electric Potential and Voltage
Electric potential energy is the energy a charge has due to its position in an electric field. Electric potential (commonly called voltage) is the potential energy per unit charge.
Unit: Volt (V), where 1 V = 1 Joule/Coulomb.
Electric potential difference between two points is what causes current to flow in a circuit.
A battery provides a constant potential difference between its terminals.
Capacitors
A capacitor is a device used to store electric charge and energy. It consists of two conducting plates separated by an insulator (dielectric).
When connected to a battery, one plate accumulates positive charge and the other negative charge.
The amount of charge stored depends on the voltage and the physical characteristics of the plates (area, distance apart).
Energy stored in a capacitor can be released quickly, making them useful in applications like camera flashes and defibrillators.

Van de Graaff Generator
The Van de Graaff generator is a laboratory device that produces high voltages and static electricity. It is commonly used to demonstrate electrostatic phenomena and the effects of large electric potentials.
It works by transferring charge to a large metal dome, building up a high voltage.
It can make hair stand on end due to repulsion of like charges.

Summary Table: Types of Materials and Their Electrical Properties
Material Type | Charge Mobility | Examples | Special Properties |
|---|---|---|---|
Conductor | High (free electrons) | Copper, Aluminum | Allows current flow easily |
Insulator | Low (bound electrons) | Rubber, Glass | Prevents current flow |
Semiconductor | Intermediate (can be controlled) | Silicon, Germanium | Conductivity can be modified by doping |
Superconductor | Zero resistance at low temperatures | Lead, Niobium alloys | Current flows indefinitely without energy loss |
