 Back
BackEnergy Transformations, Thermal Processes, and the Ideal Gas Law: Study Notes for College Physics
Study Guide - Smart Notes

Energy Transformations and Energy Transfer
Forms of Energy and Energy Transformation
Energy exists in various forms and can be transformed from one type to another. Understanding these transformations is fundamental in physics, chemistry, and biology.
Mechanical Energy: Associated with motion and position of objects.
Electrical Energy: Related to the movement of electric charges.
Thermal Energy: Due to the random motion of particles; proportional to temperature.
Light (Radiant) Energy: Energy carried by electromagnetic waves.
Chemical Energy: Stored in chemical bonds.
Examples of Energy Transformation Devices:
Solar panels: Transform light energy into electrical energy.
Wind turbines: Convert kinetic energy of wind into electrical energy.
Energy Transformation in Systems
Energy transformations occur within defined systems, and energy can be transferred between the system and its environment.
System: The part of the universe under study (e.g., kettle, wheel, wire, light bulb).
Environment: Everything outside the system.
Energy Transfer: Occurs when energy moves from the system to the environment or vice versa.
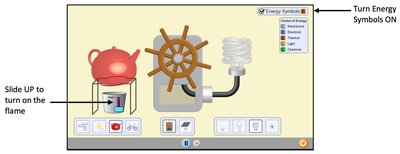
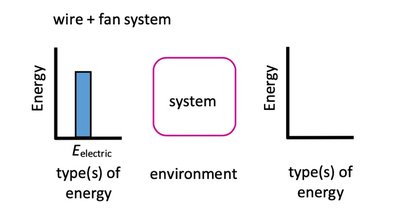
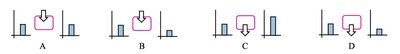
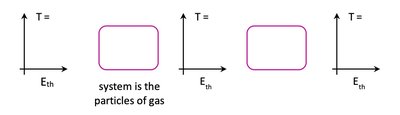
Energy Flow Diagrams
Energy flow diagrams visually represent energy conservation and transformation. They show the initial and final states of energy in a system, the process of energy transfer, and the direction of energy flow.
Bar charts indicate the types and amounts of energy before and after a process.
The rectangle represents the system; arrows show energy transfer direction and magnitude.
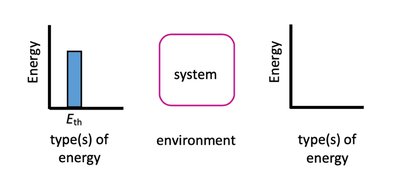
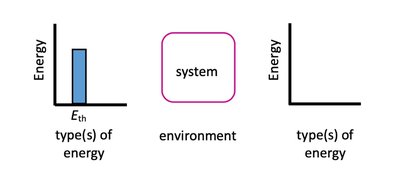
Thermal Energy, Temperature, and Heat Transfer
Heating and Cooling Processes
Thermal energy is the total kinetic energy of particles in a substance. Heating increases thermal energy and temperature, while cooling decreases them.
Heat Transfer (Q): The process of energy moving due to temperature difference.
Temperature: A measure of the average kinetic energy of particles.
Thermal Energy (Eth): The total energy due to particle motion.
Relationship Between Temperature and Thermal Energy
Higher temperature corresponds to higher thermal energy.
Heat transfer is observed as a change in temperature, measured by a thermometer.
Energy Efficiency and the Human Body
Defining Energy Efficiency
Energy efficiency is the ratio of useful energy output to energy input:
Efficiency Equation:
Energy in Biological Systems
Chemical Energy (Echem): Stored in food, released during metabolism.
Thermal Energy (Eth): Maintains body temperature.
Food Calories: 1 food Cal = 1000 cal = 1 kcal = 4190 J.

Thermal Properties of Gases
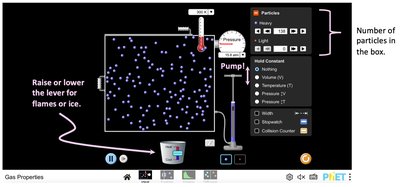
Ideal Gas Model and Kinetic Theory
An ideal gas consists of small, non-interacting particles. The temperature of a gas is proportional to the average kinetic energy of its particles:
Average Kinetic Energy:
Total Thermal Energy:
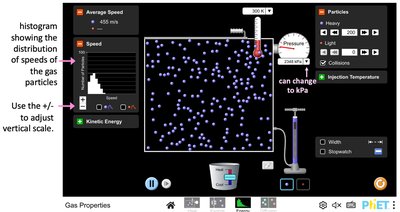
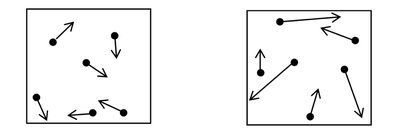
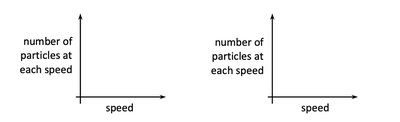
Speed Distribution of Gas Particles
The speed distribution of gas particles broadens and shifts to higher speeds as temperature increases.
At higher temperatures, particles move faster and have higher kinetic energy.
The area under the speed distribution curve remains constant (total number of particles).
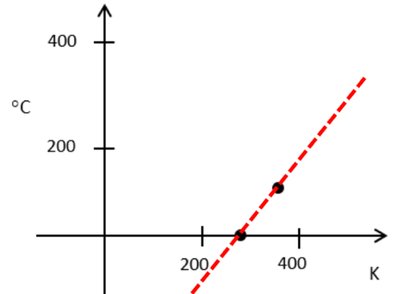
Temperature Scales
Celsius (°C), Kelvin (K), Fahrenheit (°F): Common temperature units.
Conversion Equations:
The First Law of Thermodynamics
Statement and Application
The First Law of Thermodynamics relates changes in thermal energy to heat transfer and work:
Equation:
For an ideal gas,
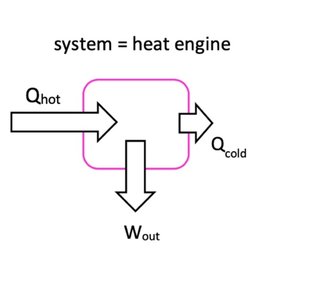
Heat Engines and Efficiency
Heat engines convert thermal energy into work, operating between hot and cold reservoirs.
Efficiency of a Heat Engine:
Maximum Efficiency: (temperatures in Kelvin)
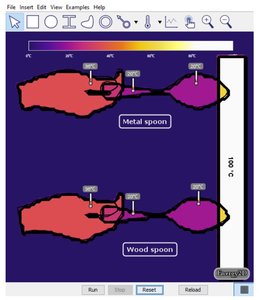
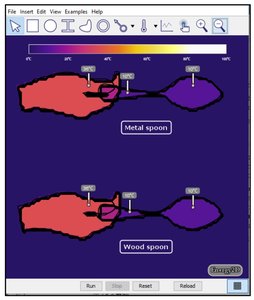
Heat Transfer Between Objects and Sensing Temperature
Thermal Contact and Heat Transfer
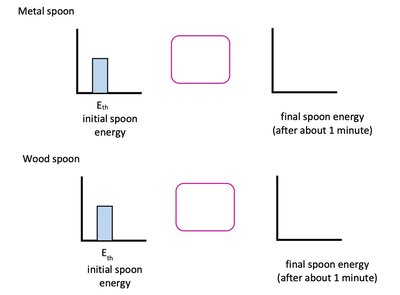
When two objects at different temperatures are in contact, heat flows from the hotter to the colder object until thermal equilibrium is reached.
Metal conducts heat quickly; wood conducts heat slowly.
Human touch senses the rate of heat transfer, not the actual temperature.
Ideal Gas Law and Gas Processes
The Ideal Gas Law
The ideal gas law relates pressure, volume, temperature, and number of particles:
Equation: or
Gas Constant:
Boltzmann's Constant:

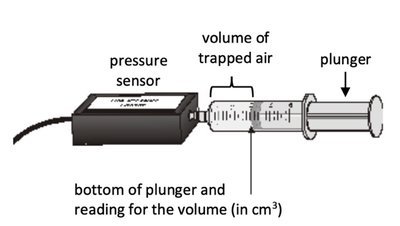
pV Diagrams and Gas Processes
pV diagrams graphically represent the states and processes of an ideal gas.
States are points defined by p, V, n (or N), T, and Eth.
Processes are lines connecting states, defined by Q and W.
Work and Heat in Gas Processes
Isovolumetric Process: Volume is constant; .
Isothermal Process: Temperature is constant; .
Adiabatic Process: No heat transfer; .
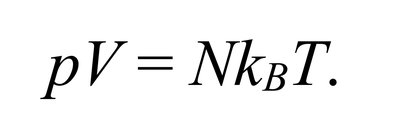
States of Matter
Microscopic and Macroscopic Properties
Matter exists in three primary states: solid, liquid, and gas. Each state has distinct microscopic and macroscopic properties.
Solid: Rigid, definite shape, closely packed particles.
Liquid: Flows, takes shape of container, particles less tightly packed.
Gas: Fills container, particles far apart, high kinetic energy.
Phase Changes and Temperature
Melting point: Temperature at which solid becomes liquid.
Boiling point: Temperature at which liquid becomes gas.
Phase changes involve changes in order and kinetic energy of particles.
Summary Table: Key Equations and Concepts
Concept | Equation | Description |
|---|---|---|
Efficiency | Ratio of useful energy output to input | |
First Law of Thermodynamics | Change in thermal energy equals work plus heat transfer | |
Ideal Gas Law | or | Relates pressure, volume, temperature, and number of particles |
Average Kinetic Energy | For monatomic ideal gas | |
Thermal Energy | Total kinetic energy of gas particles | |
Maximum Heat Engine Efficiency | Determined by reservoir temperatures (Kelvin) |
Additional info:
Energy flow diagrams and pV diagrams are essential tools for visualizing energy conservation and thermodynamic processes.
Temperature is a macroscopic property, but it reflects the microscopic kinetic energy of particles.
Phase changes involve energy transfer and changes in particle arrangement.
