 Back
BackKinetic Theory and Thermodynamics of Ideal Gases
Study Guide - Smart Notes

Kinetic Theory of Ideal Gases
Macroscopic and Microscopic Descriptions
The kinetic theory of gases provides a microscopic explanation for macroscopic properties such as pressure and temperature. It uses statistical averages over the motions of individual gas molecules to derive these properties, complementing the thermodynamic approach.
Mole: A mole is the amount of substance containing Avogadro's number () of particles.
Number of moles: , where is the number of molecules.
Ideal Gas Law: or , where J/mol·K and J/K.
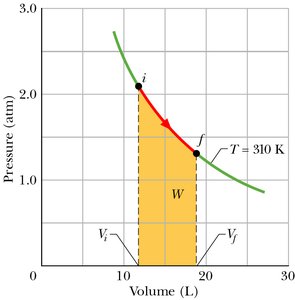
Work Done by an Ideal Gas (Isothermal Process)
During an isothermal expansion, the temperature remains constant and the work done by the gas is:
Work is positive if .
Example: The area under the curve in a p-V diagram represents the work done during an isothermal process.
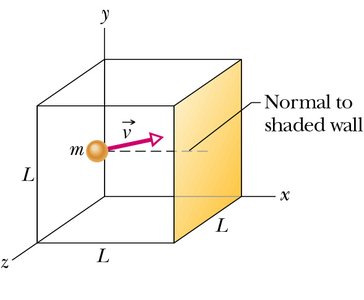
Kinetic Theory Calculation of Pressure
Microscopic Basis of Pressure
Pressure arises from the collisions of gas molecules with the walls of their container. The change in momentum for a molecule colliding with a wall is .
The rate of momentum transfer to the wall is , where is the length of the box.
For molecules, the pressure is .
Root-mean-square speed: .
Pressure in terms of : .
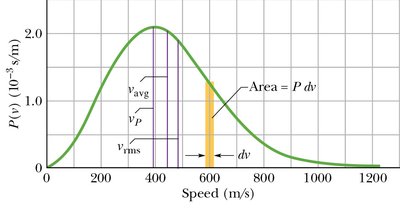
Distribution of Molecular Speeds
Maxwell Distribution Function
The speeds of molecules in an ideal gas follow the Maxwell distribution:
The fraction of molecules with speeds between and is .
Average speed:
Root-mean-square speed:
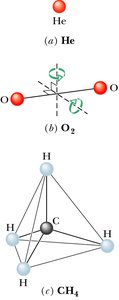
Thermal Energy and Equipartition Theorem
Average Kinetic Energy and Degrees of Freedom
The average kinetic energy of a molecule in an ideal gas is:
Total thermal energy:
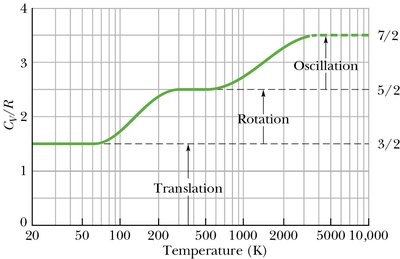
For diatomic and polyatomic gases, rotational and vibrational degrees of freedom must be considered.
Equipartition theorem: Each degree of freedom contributes per molecule.
Specific Heats of Ideal Gases
Molar Specific Heat Capacity
The molar specific heat at constant volume () is:
, where is the number of degrees of freedom (3 for monatomic, 5 for diatomic, 6 for polyatomic).
At constant volume:
At constant pressure:
Rotational and vibrational motions affect at different temperatures.
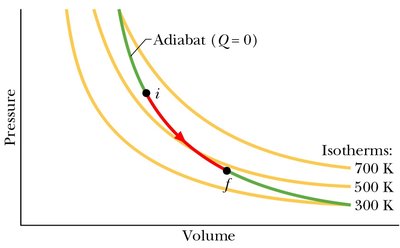
Adiabatic Expansion of an Ideal Gas
Adiabatic Processes
In an adiabatic process, no heat is transferred (). The relationship between pressure and volume is:
, where
For monatomic gases,
Both pressure and temperature drop during adiabatic expansion.
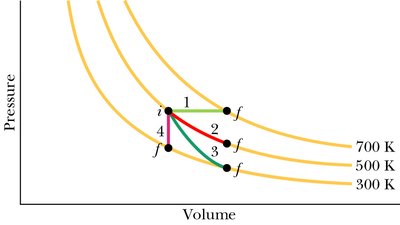
Summary of Thermodynamic Processes for an Ideal Gas
Types of Processes
Several thermodynamic processes can be represented on a p-V diagram:
Isobaric: Constant pressure
Isothermal: Constant temperature
Adiabatic: No heat transfer ()
Isochoric: Constant volume
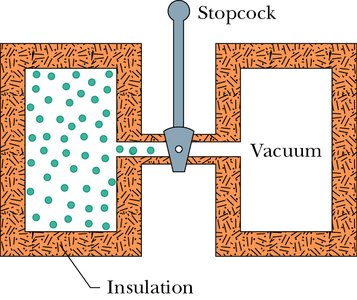
Adiabatic Free Expansion
Free Expansion Process
In an adiabatic free expansion, the gas expands into a vacuum without doing work and without heat transfer. The thermal energy remains unchanged, and the temperature does not change.
Examples and Applications
Calculations for Isothermal and Isobaric Processes
Example calculations show how to determine final pressure, temperature, and work done for isothermal and isobaric expansions using the ideal gas law and thermodynamic equations.
Isothermal:
Isobaric:
Adiabatic: ,
Table: Degrees of Freedom and Specific Heat Capacities
Type of Molecule | Degrees of Freedom (f) | C_V (per mole) | C_p (per mole) |
|---|---|---|---|
Monatomic | 3 | ||
Diatomic | 5 | ||
Polyatomic | 6 |
Additional info: Vibrational degrees of freedom become important at high temperatures, increasing the specific heat capacity.
