 Back
BackPotential and Thermal Energy, Conservation of Energy – Study Notes
Study Guide - Smart Notes

Potential and Thermal Energy, Conservation of Energy
Overview of Energy in a System
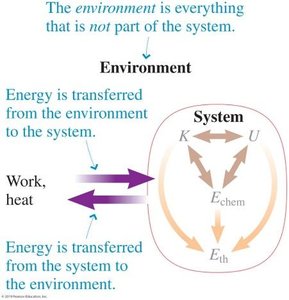
Energy in physics is a fundamental concept describing the capacity of a system to do work. A system can possess various forms of energy, and the total energy is the sum of all these forms. The boundary of a system defines what is included in energy calculations.
Kinetic Energy (K): Energy due to motion.
Gravitational Potential Energy (Ug): Energy stored due to position in a gravitational field.
Elastic Potential Energy (Us): Energy stored in stretched or compressed elastic objects.
Thermal Energy (Eth): Energy due to microscopic motion and interactions.
Chemical Energy (Echem): Energy stored in chemical bonds.
The total energy of a system is given by:

Energy Transfer Between System and Environment
The environment consists of everything outside the system. Energy can be transferred between the system and environment through work and heat.
Work: Energy transferred by forces acting over a distance.
Heat: Energy transferred due to temperature difference.
Potential Energy
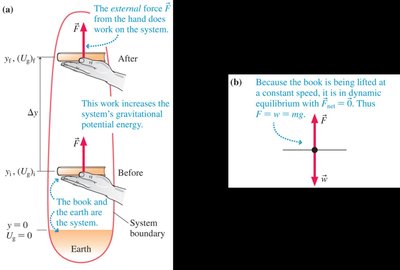
Gravitational Potential Energy
Gravitational potential energy is the energy stored due to an object's position in a gravitational field. It is relevant for systems involving the Earth and an object.
Expression:
Change in Potential Energy:
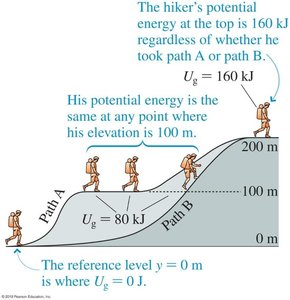
Path Independence: Only the change in height matters, not the path taken.
Reference Level: The origin for height can be chosen arbitrarily; only changes in are physically meaningful.
Units: Joules (J)
Example: The potential energy at the top of a hill is the same regardless of the path taken to reach it.
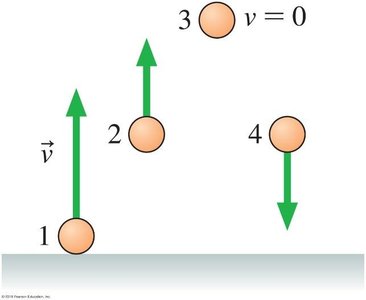
Example: Ranking gravitational potential energies of balls at different positions.
Elastic Potential Energy
Elastic potential energy is stored in objects that can be stretched or compressed, such as springs. The force exerted by a spring follows Hooke's Law:
Hooke's Law:
Spring Constant (k): Measures stiffness, units N/m.
Elastic Potential Energy:
Direction: The force and displacement are always in opposite directions.
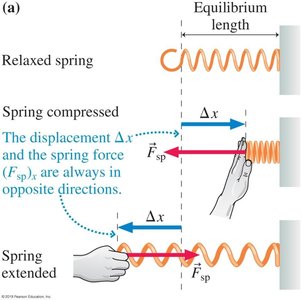
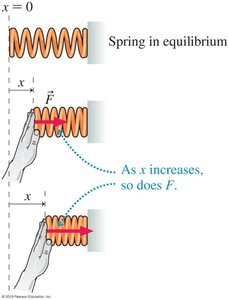
As the displacement increases, the force required to compress or stretch the spring increases.
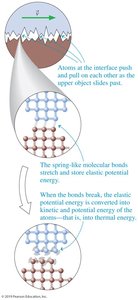
Thermal Energy
Microscopic Origin of Thermal Energy
Thermal energy is the sum of the microscopic kinetic and potential energies of all molecules in an object. It increases when friction acts on a system.
Hot Object: Fast-moving atoms have high kinetic and elastic potential energy.
Cold Object: Slow-moving atoms have low kinetic and elastic potential energy.

Friction increases thermal energy, as seen in thermographs showing temperature changes.

At the molecular level, friction causes bonds to stretch and break, converting elastic potential energy into thermal energy.
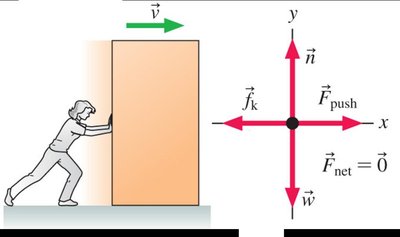
Change in Thermal Energy Due to Friction
When an object is pushed across a surface at constant speed, the work done against friction increases the system's thermal energy.
Expression:
Work-Energy Equation:
Conservation of Energy
Work-Energy Principle and Conservation Laws
The work-energy equation relates changes in kinetic, potential, and thermal energy to work done by external forces. In isolated systems, the law of conservation of energy states that the total energy remains constant.
Work-Energy Equation:
Law of Conservation of Energy: (no external work)
Mechanical Energy:
Conservation of Mechanical Energy: (if )
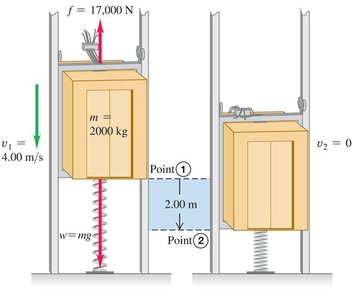
Example: Calculating the necessary spring constant to stop a falling elevator, considering friction and energy conservation.
Equation Summary
Concept | Equation |
|---|---|
Gravitational potential energy | |
Elastic potential energy | |
Change in thermal energy due to friction | |
Work-Energy Equation | |
Law of conservation of energy | |
Mechanical energy | |
Conservation of mechanical energy |
