 Back
BackQuantum Mechanics Foundations: Blackbody Radiation, Photoelectric Effect, and Wave-Particle Duality
Study Guide - Smart Notes

Origin of Quantum Theory
Introduction to Quantum Mechanics
Quantum mechanics forms the basis of modern physics, chemistry, and biology, explaining phenomena that classical physics cannot, such as blackbody radiation, the photoelectric effect, and Compton scattering. It introduces the concepts of wave-particle duality and quantization of energy.
Quantum mechanics applies to solid state, molecular, atomic, nuclear, and particle physics, as well as optics, thermodynamics, and statistical mechanics.
It is foundational for understanding chemical and biological systems.
Classical Concepts
Classical physics distinguishes between particles and waves, but fails to explain microscopic phenomena.
Particles: Characterized by energy and momentum .
Waves: Described by amplitude and wave vector .
Classical physics cannot explain phenomena like blackbody radiation and the photoelectric effect.
Blackbody Radiation
Definition and Experimental Observations
Blackbody radiation refers to the thermal radiation emitted by an idealized object that absorbs all incident radiation and emits energy when heated. The emitted radiation has a continuous frequency distribution, with a pronounced maximum that shifts with temperature.
Blackbody: An ideal absorber and emitter of radiation.
Spectral energy density depends on frequency and temperature .
Classical physics failed to explain the observed spectral distribution.
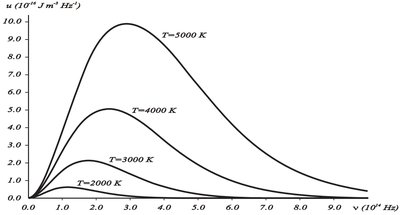
Classical Theories and Their Failures
Classical theories attempted to describe blackbody radiation but failed at certain frequency ranges.
Wien’s Law: fits high-frequency data but fails at low frequencies.
Rayleigh-Jeans Law: fits low-frequency data but diverges at high frequencies (ultraviolet catastrophe).
Both laws are based on classical physics and do not match experimental results across all frequencies.
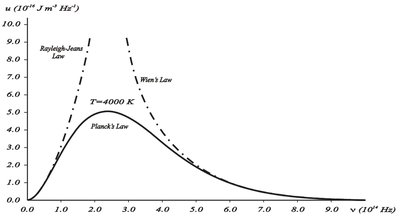
Planck’s Quantum Hypothesis
Max Planck resolved the discrepancies by proposing that energy exchange occurs in discrete units called quanta.
Quantum of energy: , where is Planck’s constant.
Planck’s formula:
This formula accurately describes the spectral energy density and avoids the ultraviolet catastrophe.
Planck’s Distribution and Limits
At low frequencies, Planck’s law reduces to Rayleigh-Jeans law.
At high frequencies, it approaches Wien’s law.
Total energy density is finite, resolving the ultraviolet catastrophe.
Photoelectric Effect
Definition and Experimental Setup
The photoelectric effect is the emission of electrons from a material when exposed to light. It was discovered by Heinrich Hertz and explained by Albert Einstein, marking a pivotal moment in quantum mechanics.
Electrons are ejected from a metal surface when illuminated by light of sufficient frequency.
The current generated by ejected electrons is measured.
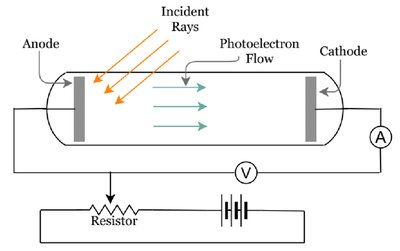
Key Observations
Threshold Frequency: No electrons are emitted if the light frequency is below a certain threshold .
Instantaneous Emission: Electrons are emitted instantly, even at low light intensities.
Kinetic Energy: Depends on light frequency, not intensity.
Current Proportionality: Number of emitted electrons is proportional to light intensity.
Example Calculation
Problem: Calculate the maximum kinetic energy of electrons emitted from a metal with work function eV when illuminated with light of wavelength nm.
Convert wavelength to frequency: Hz
Photon energy: J eV
Maximum kinetic energy: eV eV eV
Einstein’s Explanation
Einstein proposed that light consists of discrete packets called photons.
Photon energy:
Electrons are ejected when a photon transfers its energy to an electron.
Maximum kinetic energy:
Threshold frequency:
Wave-Particle Duality
Classical vs. Quantum Views
Classical physics treats particles and waves as distinct entities, but quantum mechanics describes particles by wave functions, providing probabilistic information about their position and momentum.
Wave function : Describes the quantum state of a particle.
Probability density: gives the likelihood of finding a particle at position at time .
Particles exhibit both wave-like and particle-like properties.
Double-Slit Experiment
The double-slit experiment demonstrates wave-particle duality: particles create an interference pattern (wave-like behavior) but are detected as discrete entities (particle-like behavior). 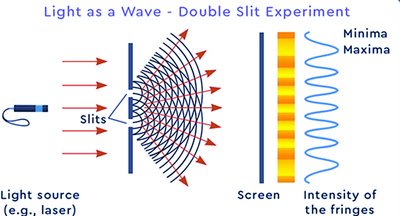
De Broglie Hypothesis
Louis de Broglie proposed that particles have an associated wavelength:
De Broglie wavelength: , where is momentum.
Experimental evidence: Electron diffraction confirms the wave nature of electrons.
Heisenberg’s Uncertainty Principle
It is impossible to simultaneously know the exact position and momentum of a particle.
Uncertainty relation: , where
This principle arises from the wave nature of particles.
Complementarity Principle
Proposed by Niels Bohr, this principle states that particles and waves are complementary aspects of the same entity, and the observed nature depends on the experimental setup.
Observing which slit a particle passes through destroys the interference pattern.
Wave Function and Probability
Definition and Properties
The wave function is a complex-valued function containing information about the quantum state of a particle.
Solution to the Schrödinger equation:
Properties: Complex-valued, continuous, single-valued, normalizable.
Probability density:
Normalization:
Probability Current Density
The probability current density describes the flow of probability in space and time, ensuring conservation of probability.
Definition:
Continuity equation:
Total probability is conserved over time.
Example: Free Particle Wave Function
For a free particle described by , the probability current density is:
Example: Gaussian Wave Packet
For a Gaussian wave packet, the probability current density is proportional to the group velocity .
Mapping of Course Outcomes (COs) with Program Outcomes (POs) and Program-Specific Outcomes (PSOs)
Purpose of Mapping
Mapping COs with POs and PSOs ensures that course content aligns with program goals and academic standards.
1 = weakly mapped
2 = moderately mapped
3 = strongly mapped
PO1 | PO2 | PO3 | PO4 | PO5 | PO6 | PO7 | PO8 | PO9 | PO10 | PO11 | PO12 | PSO1 | PSO2 | PSO3 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
COPHY1008.1 | 2 | 2 | 3 | 3 | 3 | 1 | 1 | 3 | 3 | 3 | 1 | ||||
COPHY1008.2 | 2 | 2 | 3 | 3 | 3 | 1 | 1 | 3 | 3 | 3 | 1 | ||||
COPHY1008.3 | 2 | 2 | 2 | 3 | 3 | 1 | 1 | 3 | 3 | 3 | 1 | ||||
COPHY1008.4 | 2 | 2 | 3 | 3 | 3 | 1 | 1 | 3 | 3 | 3 | 1 | ||||
COPHY1008.5 | 3 | 1 | 2 | 3 | 3 | 1 | 1 | 3 | 3 | 3 | 1 |
Additional info: The notes cover foundational quantum mechanics topics relevant to college-level physics, including blackbody radiation, the photoelectric effect, wave-particle duality, and the mathematical framework of quantum theory.
