 Back
BackThermodynamics and the Kinetic Theory of Gases: Foundations and Applications
Study Guide - Smart Notes

Thermodynamics: Introduction and Scope
Overview of Thermodynamics
Thermodynamics is the branch of physics that deals with the relationships between heat, work, temperature, and energy. It provides a macroscopic description of physical systems and is fundamental to understanding energy transformations in physical, chemical, and biological processes.
Thermodynamics studies the collective behavior of large numbers of particles, using statistical methods to relate microscopic properties to macroscopic observables.
It is essential for understanding engines, refrigerators, biological systems, and atmospheric phenomena.
Microscopic and Macroscopic Descriptions
From Newtonian Mechanics to Statistical Physics
While Newton's laws provide exact solutions for two-body problems, systems with more than two interacting particles (such as gases) require statistical approaches due to the complexity and number of particles involved.
Two-body problem: Can be solved exactly using Newton's laws.
Three-body problem: Becomes analytically unsolvable in general; requires numerical or statistical methods.
Many-body systems: Statistical mechanics is used to describe the average behavior of large numbers of particles.
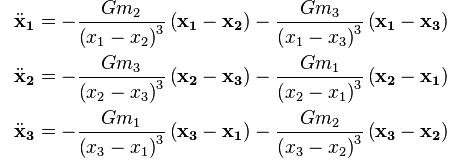
Atomic and Molecular Quantities
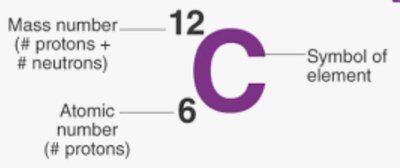
The Mole, Avogadro's Number, and Atomic Mass Unit
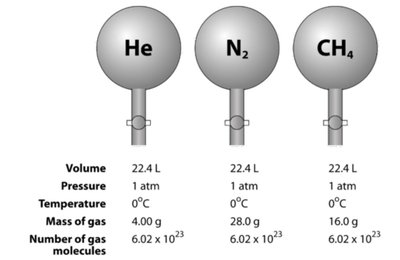
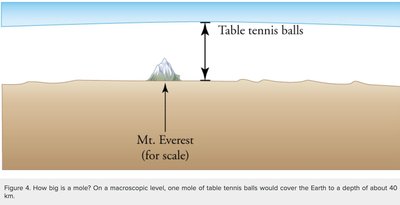
To handle the vast number of particles in macroscopic samples, physicists use the concepts of the mole, Avogadro's number, and the atomic mass unit (amu).
Mole (mol): The amount of substance containing exactly elementary entities (Avogadro's number, ).
Atomic Mass Unit (amu or u): Defined as the mass of a carbon-12 atom; .
Molar Mass: The mass of one mole of a substance (e.g., : 32 g/mol, : 44 g/mol).
Thermodynamic Variables and State Functions
State Functions vs. Path Functions
Thermodynamic systems are described by variables such as pressure, volume, temperature, and entropy. These variables are classified as state functions or path functions.
State Functions: Quantities that depend only on the current state of the system, not on how it was reached (e.g., Internal energy , Pressure , Volume , Temperature , Entropy ).
Derived State Functions: Enthalpy , Helmholtz free energy , Gibbs free energy .
Path Functions: Quantities that depend on the process or path taken (e.g., Work , Heat ).
Gas Laws and the Ideal Gas Model
Empirical Gas Laws
The behavior of gases is described by several empirical laws, which are unified in the ideal gas law.
Boyle’s Law: At constant temperature, .
Charles’ Law: At constant pressure, .
Gay-Lussac’s Law: At constant volume, .
Combined Gas Law: (for a fixed amount of gas).
The Ideal Gas Law combines these relationships:
= pressure (Pa)
= volume (m3)
= number of moles
= universal gas constant ()
= temperature (K)
On a per-molecule basis, the ideal gas law is:
= number of molecules
= Boltzmann constant ()


Conditions for Ideal Gas Behavior
Low density (molecules far apart)
High temperature (well above condensation point)
No significant intermolecular forces or chemical reactions
Kinetic Theory of Gases
Microscopic Interpretation of Temperature and Pressure
The kinetic theory explains macroscopic properties of gases in terms of the motion of their molecules.
Average kinetic energy per molecule:
Root-mean-square (rms) speed:
where is the mass of a molecule.
Mean free path (λ): The average distance a molecule travels between collisions:
where is the molecular diameter, is pressure.

Worked Example: Ideal Gas Law Application
Calculating Pressure of a Gas
Problem: 100 g of pure oxygen gas is in a 600 cm3 container at 150°C. What is the gas pressure?
Convert mass to moles:
Convert volume:
Convert temperature:
Apply ideal gas law:
Note: Temperatures must always be in kelvins when using the ideal gas law.
Conceptual Questions and Applications
Understanding Molecular Speeds and Temperature
If the temperature of an ideal gas increases, the speed of the molecules increases, and so does the pressure (if volume is constant) or the volume (if pressure is constant).
If the root-mean-square speed of molecules doubles, the temperature increases by a factor of 4 (since ).
Summary Table: Key Thermodynamic Quantities
Quantity | Symbol | Definition | SI Unit |
|---|---|---|---|
Pressure | P | Force per unit area | Pa (N/m2) |
Volume | V | Space occupied by gas | m3 |
Temperature | T | Measure of average kinetic energy | K |
Number of moles | n | Amount of substance | mol |
Boltzmann constant | kB | Relates temperature to energy | J/K |
Universal gas constant | R | Proportionality constant in ideal gas law | J/(mol·K) |
Additional info:
Thermodynamics bridges the gap between microscopic particle behavior and macroscopic observables, providing a foundation for advanced topics such as entropy, free energy, and statistical mechanics.
Understanding the distinction between state and path functions is crucial for solving thermodynamic problems and analyzing energy transformations.
