All the chemical reactions that occur in the human body are collectively referred to as
(a) Anabolism
(b) Catabolism
(c) Metabolism
(d) Homeostasis

 Martini, Nath, Bartholomew 11th Edition
Martini, Nath, Bartholomew 11th Edition Ch. 2 The Chemical Level of Organization
Ch. 2 The Chemical Level of Organization Problem 5
Problem 5 Verified step by step guidance
Verified step by step guidance


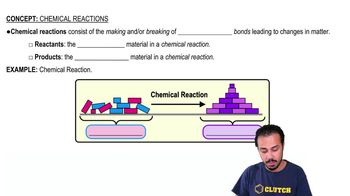
All the chemical reactions that occur in the human body are collectively referred to as
(a) Anabolism
(b) Catabolism
(c) Metabolism
(d) Homeostasis
A substance containing atoms of different elements that are bonded together is called a(n)
(a) Molecule
(b) Compound
(c) Mixture
(d) Isotope
(e) Solution
Isotopes of an element differ from each other in the number of
(a) Protons in the nucleus
(b) Neutrons in the nucleus
(c) Electrons in the outer shells
(d) a, b, and c are all correct
The subatomic particle with the least mass
(a) Carries a negative charge
(b) Carries a positive charge
(c) Plays no part in the atom's chemical reactions
(d) Is found only in the nucleus
All organic compounds in the human body contain all of the following elements except
(a) Hydrogen
(b) Oxygen
(c) Carbon
(d) Calcium
(e) Both a and d
What is the following type of decomposition reaction called?
ABCD + H₂O → ABCH + DOH