 Back
BackChapter 2: Chemistry Comes Alive – ANP Study Guidance
Study Guide - Smart Notes

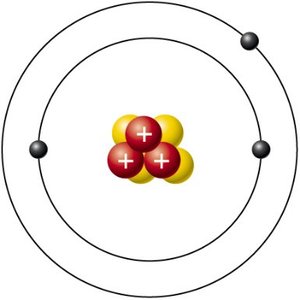
Q6. The atom shown below has _______ electrons, _________ protons, and ________ neutrons.
Background
Topic: Atomic Structure
This question tests your understanding of the basic structure of an atom, including the arrangement and number of subatomic particles: electrons, protons, and neutrons.
Key Terms and Formulas
Electron: Negatively charged particle found in electron shells surrounding the nucleus.
Proton: Positively charged particle found in the nucleus.
Neutron: Neutral particle found in the nucleus.
Atomic Number: Number of protons in the nucleus.
Mass Number: Sum of protons and neutrons in the nucleus.
Step-by-Step Guidance
Examine the diagram carefully. Count the number of black dots (electrons) in the electron shells.
Count the number of red spheres with plus signs (protons) in the nucleus.
Count the number of yellow spheres (neutrons) in the nucleus.
Recall that the atomic number equals the number of protons, and for a neutral atom, the number of electrons equals the number of protons.
Try solving on your own before revealing the answer!
Final Answer: 2 electrons, 2 protons, 2 neutrons
The diagram shows two electrons in the shells, two protons (red spheres with plus signs), and two neutrons (yellow spheres) in the nucleus. This represents a helium atom.
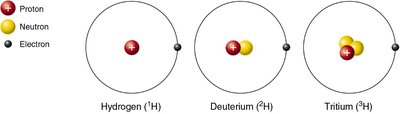
Q13. These structural variations of hydrogen are called _____________________. Which subatomic particle can vary in number among these three structural variations?
Background
Topic: Isotopes
This question is about isotopes, which are atoms of the same element with different numbers of neutrons.
Key Terms and Formulas
Isotope: Atoms of the same element with different numbers of neutrons.
Proton: Determines the element; does not change among isotopes.
Neutron: Varies among isotopes, affecting mass number.
Step-by-Step Guidance
Observe the three diagrams: each has one proton (red sphere with plus sign) and one electron (black dot).
Count the number of neutrons (yellow spheres) in each diagram: hydrogen has none, deuterium has one, tritium has two.
Recall that isotopes are defined by their differing numbers of neutrons, while the number of protons remains the same.
Identify the term for these variations and the subatomic particle that changes.
Try solving on your own before revealing the answer!
Final Answer: Isotopes; Neutron
These variations are called isotopes. The number of neutrons varies among them, which changes their mass number but not their chemical properties.
Q5. Explain why the overall reaction for cellular respiration (shown below) is never reversed in body cells.
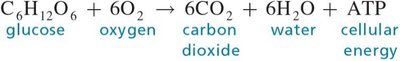
Background
Topic: Cellular Respiration and Metabolic Reactions
This question tests your understanding of the directionality of metabolic reactions, specifically why cellular respiration is considered irreversible in living cells.
Key Terms and Formulas
Cellular Respiration: The process by which cells break down glucose and oxygen to produce carbon dioxide, water, and ATP (energy).
Irreversible Reaction: A reaction that cannot easily be reversed under normal physiological conditions.
ATP: Adenosine triphosphate, the energy currency of the cell.
Step-by-Step Guidance
Examine the chemical equation: .
Consider the energy released during the breakdown of glucose; this energy is captured in ATP and lost as heat.
Think about the physiological conditions in cells: the products (CO2 and H2O) are removed from the cell, making reversal difficult.
Reflect on why the cell cannot easily convert CO2 and H2O back into glucose under normal conditions.
Try solving on your own before revealing the answer!
Final Answer: Cellular respiration is irreversible because the products are removed and energy is lost as heat.
The reaction cannot be reversed in body cells because the products are continually removed and the energy released is not recoverable.
Q11. The bicarbonate buffer system shown below resists changes in blood pH by shifting to the right or left in response to a rise or drop in pH.

Background
Topic: Acid-Base Balance and Buffer Systems
This question tests your understanding of how the bicarbonate buffer system maintains blood pH by shifting equilibrium in response to changes in acidity.
Key Terms and Formulas
Buffer: A system that resists changes in pH by absorbing or releasing H+ ions.
Bicarbonate Buffer System:
Equilibrium Shift: Movement to the right or left depending on pH changes.
Step-by-Step Guidance
Examine the equation: .
Consider what happens when blood becomes more acidic (increase in H+ ions).
Think about which direction the equilibrium will shift to remove excess H+ ions.
Determine whether this shift results in the addition or removal of H+ ions from the blood.
Try solving on your own before revealing the answer!
Final Answer: The equilibrium shifts to the left, removing H+ ions from the blood.
When blood becomes more acidic, the buffer system shifts to the left, converting H+ and HCO3- into H2CO3, thus reducing free H+ ions.
Q7. Why do phospholipids form a bilayer (shown below) when placed in a watery environment? (In your answer be sure to indicate which parts of a phospholipid are polar or nonpolar.)

Background
Topic: Cell Membrane Structure
This question tests your understanding of the amphipathic nature of phospholipids and how this leads to the formation of a bilayer in aqueous environments.
Key Terms and Formulas
Phospholipid: A molecule with a polar (hydrophilic) head and nonpolar (hydrophobic) tails.
Bilayer: Two layers of phospholipids with heads facing outward toward water and tails facing inward away from water.
Hydrophilic: Water-loving, polar.
Hydrophobic: Water-fearing, nonpolar.
Step-by-Step Guidance
Identify the structure of a phospholipid: a polar head and two nonpolar tails.
Recall that polar heads interact with water, while nonpolar tails avoid water.
Explain how phospholipids arrange themselves in a bilayer: heads face outward toward water, tails face inward away from water.
Consider why this arrangement is stable in a watery environment.
Try solving on your own before revealing the answer!
Final Answer: Phospholipids form a bilayer because their polar heads face water and nonpolar tails avoid water.
The bilayer is stable because the hydrophilic heads interact with water, while the hydrophobic tails are shielded from water inside the membrane.
Q5. When cooking an egg (shown below) what is the term that describes what happens to the protein albumin to cause the coagulation of egg white?

Background
Topic: Protein Structure and Denaturation
This question tests your understanding of how heat affects protein structure, specifically the process that causes proteins to lose their native shape and coagulate.
Key Terms and Formulas
Denaturation: The process by which proteins lose their three-dimensional structure due to external stress (such as heat).
Albumin: The main protein in egg white.
Coagulation: The process of proteins clumping together after denaturation.
Step-by-Step Guidance
Consider what happens to proteins when exposed to heat: their structure unfolds.
Recall the term for this process: denaturation.
Understand that denatured proteins can aggregate, leading to coagulation.
Apply this concept to the cooking of egg whites, where albumin proteins denature and coagulate.
Try solving on your own before revealing the answer!
Final Answer: Denaturation
When egg whites are cooked, the albumin proteins denature, losing their native structure and coagulating to form a solid mass.
