 Back
BackPancreatic Hormones and Glucose Homeostasis: Structure, Function, and Pathophysiology
Study Guide - Smart Notes

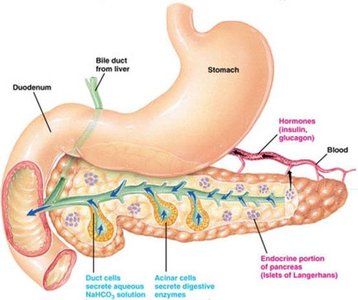
Pancreas: Structure and Dual Function
Overview of Pancreatic Anatomy and Physiology
The pancreas is a vital organ with both exocrine and endocrine functions. Its exocrine portion secretes digestive enzymes, while the endocrine portion, composed of the islets of Langerhans, regulates blood glucose through hormone secretion.
Exocrine function: Secretion of digestive enzymes and bicarbonate into the duodenum.
Endocrine function: Regulation of glucose homeostasis via hormone release into the bloodstream.
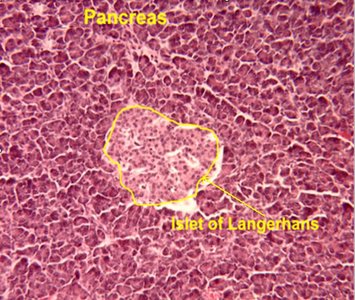
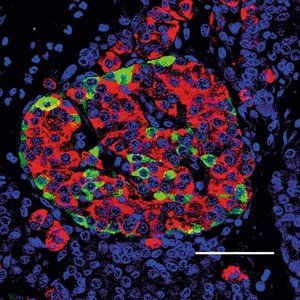
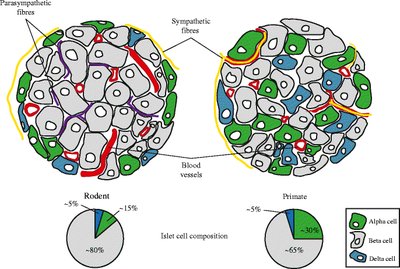
Islets of Langerhans: Structure and Cell Types
Organization and Cellular Composition
The islets of Langerhans are clusters of endocrine cells within the pancreas, comprising about 1-2% of pancreatic mass. They contain several distinct cell types, each producing specific hormones essential for metabolic regulation.
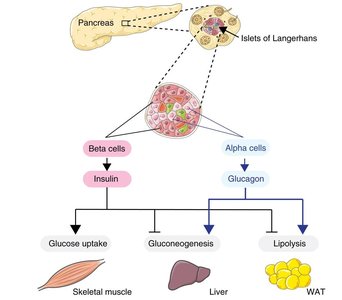
Alpha (α) cells: Secrete glucagon.
Beta (β) cells: Secrete insulin (most prevalent cell type).
Delta (D) cells: Secrete somatostatin (SST).
F (PP) cells: Secrete pancreatic polypeptide.
Insulin: Synthesis, Secretion, and Action
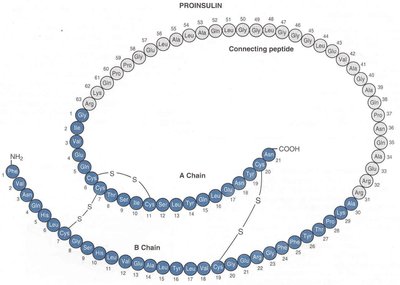
Insulin Biosynthesis
Insulin is a peptide hormone produced by β cells. Its synthesis involves several steps, beginning with the translation of preproinsulin and ending with the formation of active insulin and C-peptide.
Preproinsulin: Initial translation product; signal peptide is cleaved to form proinsulin.
Proinsulin: Processed in the Golgi apparatus; cleaved into insulin (51 amino acids, two chains) and C-peptide.
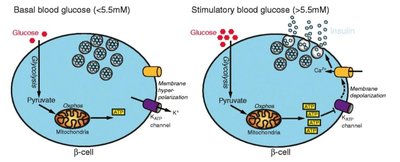
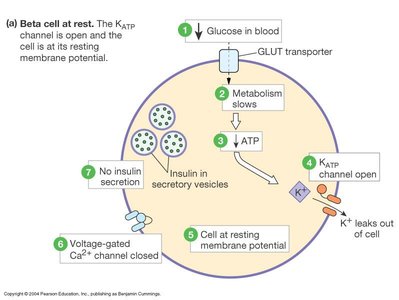
Regulation of Insulin Secretion
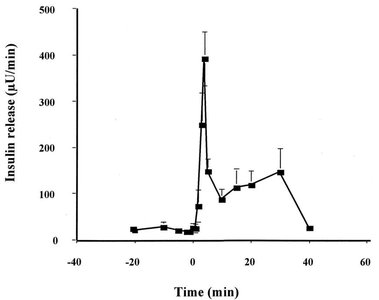
Insulin secretion is tightly regulated by blood glucose levels. Secretion is biphasic, with an initial rapid release followed by a sustained phase as long as glucose remains elevated.
Threshold for secretion: ~100 mg/dL plasma glucose.
Biphasic release: Initial burst (5-15 min), then gradual increase.
Stimuli: Glucose, amino acids, and certain hormones stimulate release; stress (epinephrine) inhibits it.
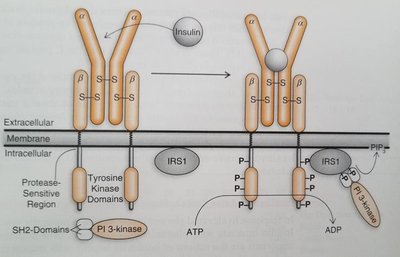
Insulin Receptor and Signal Transduction
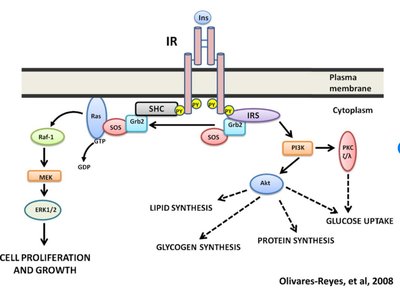
Insulin acts via the insulin receptor, a tyrosine kinase receptor composed of two alpha and two beta subunits. Binding of insulin triggers autophosphorylation and activation of downstream signaling pathways, including the PI3K/Akt pathway, leading to metabolic effects.
Key proteins: SHC, IRS (Insulin Receptor Substrate).
Major effects: Increased glucose uptake, glycogen synthesis, lipid synthesis, and protein synthesis.
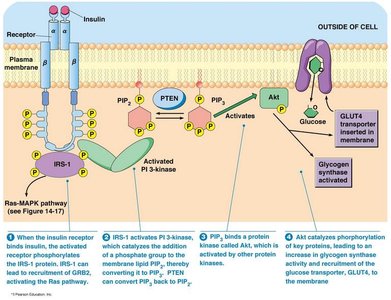
Glucose Transport and Metabolic Effects
Insulin promotes glucose uptake in muscle and adipose tissue by stimulating the translocation of GLUT4 transporters to the cell membrane. It also regulates carbohydrate, lipid, and protein metabolism in various tissues.
Liver: Stimulates glycogenesis, inhibits glycogenolysis and gluconeogenesis.
Muscle: Increases glucose uptake and glycogen synthesis.
Adipose tissue: Promotes glucose uptake, triglyceride synthesis, and inhibits lipolysis.
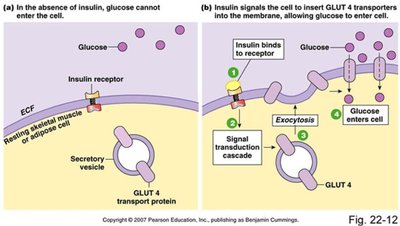
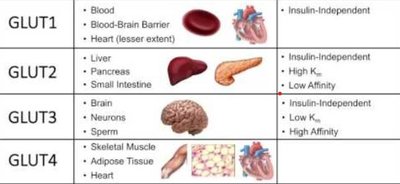
Summary of Insulin Action
Promotes storage of glucose as glycogen and fat.
Stimulates protein synthesis and inhibits protein degradation.
Maintains blood glucose within a narrow range.
Glucagon: Synthesis, Secretion, and Action
Glucagon Biosynthesis and Regulation
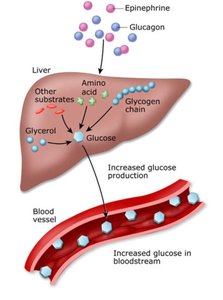
Glucagon is a 29-amino acid peptide hormone produced by α cells. Its secretion is stimulated by low blood glucose, amino acids, and stress (epinephrine), and inhibited by high glucose and insulin.
Stimuli for release: Hypoglycemia, high protein meals, sympathetic stimulation.
Inhibition: Hyperglycemia, insulin, somatostatin.

Mechanism of Action and Metabolic Effects
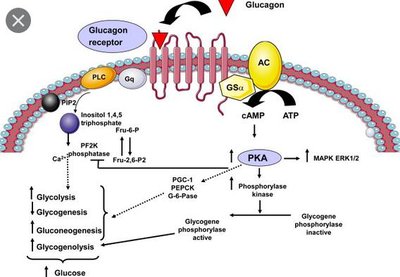
Glucagon acts primarily on the liver via G protein-coupled receptors, activating adenylate cyclase and increasing cAMP. This leads to the activation of protein kinase A (PKA) and subsequent metabolic effects.
Stimulates: Glycogenolysis (breakdown of glycogen), gluconeogenesis (formation of glucose from non-carbohydrate sources), and lipolysis (release of fatty acids from adipose tissue).
Inhibits: Glycolysis in the liver.
Main effect: Increases blood glucose during fasting or stress.
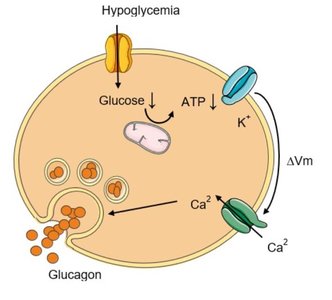
Integration of Insulin and Glucagon in Glucose Homeostasis
Feedback Regulation and Homeostatic Balance
Insulin and glucagon have opposing actions that maintain blood glucose within a physiological range. Their secretion is regulated by negative feedback mechanisms in response to plasma glucose levels.
High glucose: Stimulates insulin, inhibits glucagon.
Low glucose: Stimulates glucagon, inhibits insulin.
Other regulators: Amino acids, stress hormones, somatostatin.
Other Pancreatic Hormones
Somatostatin (SST) and Pancreatic Polypeptide (PP)
Other hormones produced by the islets of Langerhans include somatostatin (from D cells) and pancreatic polypeptide (from F cells). These hormones modulate the secretion of insulin and glucagon and have roles in gastrointestinal function.
Somatostatin: Inhibits insulin and glucagon secretion; acts as a paracrine regulator.
Pancreatic polypeptide: Role not fully defined; increased by protein-rich meals and hypoglycemia.
Pathophysiology: Diabetes Mellitus
Types and Mechanisms
Diabetes mellitus is a disorder of carbohydrate metabolism resulting from insufficient insulin secretion or action. It is characterized by hyperglycemia and the classic symptoms of polyuria, polydipsia, and polyphagia.
Type 1 Diabetes Mellitus (IDDM): Autoimmune destruction of β cells; absolute insulin deficiency; requires insulin therapy.
Type 2 Diabetes Mellitus (NIDDM): Insulin resistance and/or impaired insulin secretion; often associated with obesity; may progress to β cell dysfunction.
Key differences: Type 1 is due to β cell failure, while Type 2 involves insulin resistance and eventual β cell exhaustion.
Clinical Features and Consequences
Polyuria: Excessive urination due to osmotic diuresis from high glucose.
Polydipsia: Excessive thirst due to dehydration.
Polyphagia: Excessive hunger due to inability to utilize glucose.
Summary Table: Insulin vs. Glucagon Actions
Hormone | Main Source | Main Target | Primary Actions |
|---|---|---|---|
Insulin | β cells (Islets of Langerhans) | Liver, muscle, adipose tissue | ↓ Blood glucose, ↑ Glycogenesis, ↑ Lipogenesis, ↑ Protein synthesis |
Glucagon | α cells (Islets of Langerhans) | Liver, adipose tissue | ↑ Blood glucose, ↑ Glycogenolysis, ↑ Gluconeogenesis, ↑ Lipolysis |
Additional info: The balance between insulin and glucagon is essential for metabolic homeostasis. Disruption leads to metabolic diseases such as diabetes mellitus, with significant clinical consequences if untreated.
