 Back
BackAmino Acids and Fundamentals of Protein Structure: Mini-Textbook Study Notes
Study Guide - Smart Notes

Amino Acids
Definition and General Structure
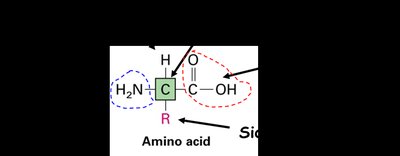
Amino acids are the building blocks of proteins, consisting of a central carbon atom (the α-carbon) bonded to an amino group (—NH2), a carboxyl group (—COOH), a hydrogen atom, and a unique side chain (R group) that determines the identity and properties of each amino acid.
Key Elements: Carbon, hydrogen, oxygen, nitrogen; some side chains contain sulfur or other elements.
General Formula:
Side Chain (R group): Distinguishes each amino acid and determines its chemical behavior.
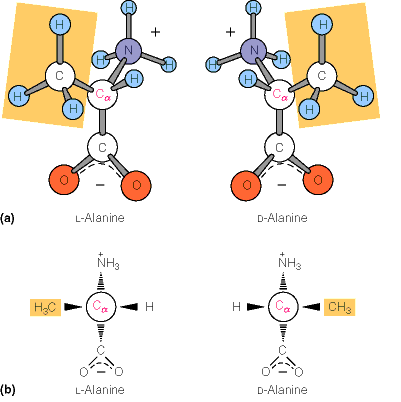
Chirality and Stereoisomers
Most amino acids (except glycine) are chiral, existing as L- and D- stereoisomers. Proteins in living organisms are composed exclusively of L-amino acids.
L-form: Used in protein synthesis.
D-form: Rare in nature, found in some bacterial cell walls.
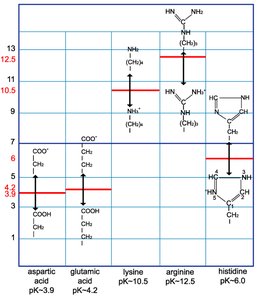
Zwitterions and Isoelectric Point (pI)
At physiological pH (~7), amino acids exist as zwitterions, carrying both positive and negative charges but having a net neutral charge at their isoelectric point (pI).
Zwitterion: Molecule with both a positive (amino group) and negative (carboxyl group) charge.
Isoelectric Point (pI): The pH at which the amino acid has no net charge.
pI Calculation: For neutral amino acids,
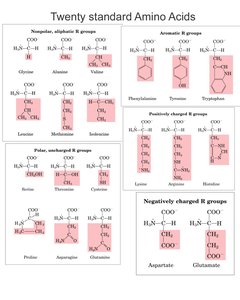
Classification and Properties of Amino Acids
Grouping by Side Chain Properties
Amino acids are classified based on the chemical nature of their side chains (R groups), which influence their role in protein structure and function.
Nonpolar, Aliphatic: Hydrophobic, found in protein interiors (e.g., alanine, valine, leucine).
Aromatic: Contain ring structures, absorb UV light (e.g., phenylalanine, tyrosine, tryptophan).
Polar, Uncharged: Hydrophilic, often on protein surfaces (e.g., serine, threonine, cysteine).
Positively Charged (Basic): Lysine, arginine, histidine; participate in salt bridges.
Negatively Charged (Acidic): Aspartate, glutamate; also form salt bridges.
Special Amino Acids
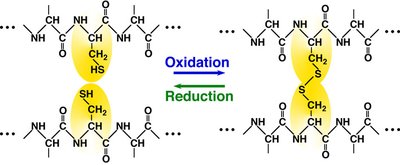
Cysteine: Can form disulfide bonds, stabilizing protein structure.
Proline: Cyclic structure, introduces rigidity and kinks in polypeptide chains.
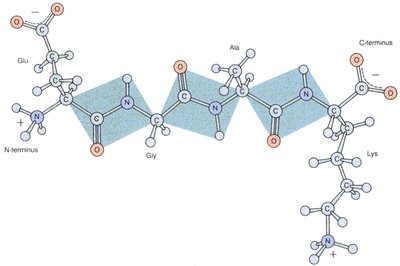
Peptide Bonds and Protein Structure
Formation of Peptide Bonds
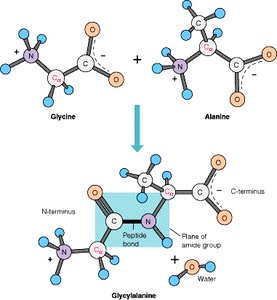
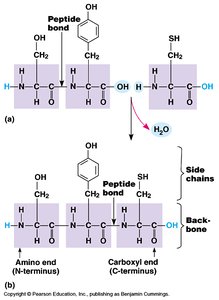
A peptide bond is a covalent bond formed between the carboxyl group of one amino acid and the amino group of another, releasing a molecule of water (condensation reaction).
Reaction:
Energy Requirement: Formation requires energy, typically supplied by ATP in living systems.
Hydrolysis: Peptide bonds can be broken by adding water (hydrolysis), releasing energy.
Properties of the Peptide Bond
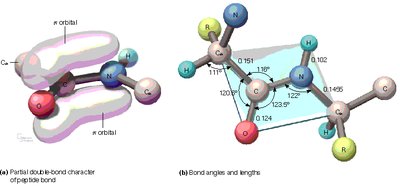
The peptide bond has partial double-bond character due to resonance, restricting rotation and resulting in a planar structure.
Partial Double Bond: Resonance between the carbonyl oxygen and amide nitrogen.
Planarity: Peptide bonds are planar, limiting flexibility.
Bond Angles: Typical bond angles are ~120° due to sp2 hybridization.
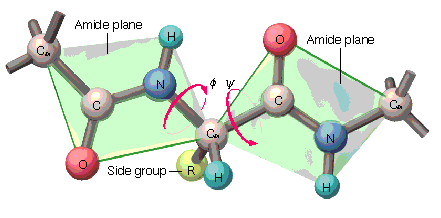
Flexibility in Polypeptide Chains
Rotation in polypeptide chains occurs around the bonds adjacent to the α-carbon, defined by the φ (phi) and ψ (psi) torsional angles.
φ (Phi) Angle: Rotation around the N—Cα bond.
ψ (Psi) Angle: Rotation around the Cα—C bond.
Importance: These angles determine the possible conformations of the polypeptide chain.
Effects of Amino Acids on Protein Structure
Role of R Groups in Folding
The chemical properties of amino acid side chains drive the folding of proteins into their functional three-dimensional structures.
Hydrophobic Amino Acids: Congregate in the protein core, away from water.
Hydrophilic Amino Acids: Located on the surface, interact with water.
Acidic and Basic Amino Acids: Form ionic interactions (salt bridges).
Cysteine: Forms covalent disulfide bonds, stabilizing structure.
Hydrogen Bonds: Many amino acids can form hydrogen bonds, contributing to secondary and tertiary structure.

Hydrogen Bonding in Proteins
Definition and Importance
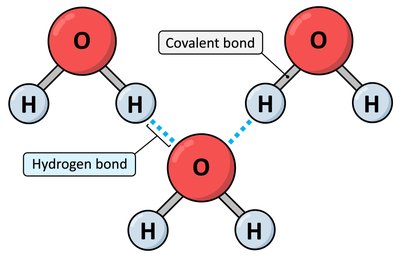
Hydrogen bonds are weak electrostatic attractions between a hydrogen atom covalently bonded to an electronegative atom (donor) and another electronegative atom with a lone pair (acceptor). They are crucial for stabilizing protein and nucleic acid structures.
Donor: Typically N—H or O—H groups.
Acceptor: O or N atoms with lone pairs.
Role: Stabilize α-helices, β-sheets, and tertiary structure.
Hydrogen Bonds in Proteins
Hydrogen bonds form between backbone atoms and side chains, contributing to secondary and tertiary structure.
Backbone Hydrogen Bonds: Between carbonyl oxygen and amide hydrogen in α-helices and β-sheets.
Side Chain Hydrogen Bonds: Between polar side chains, stabilizing tertiary structure.
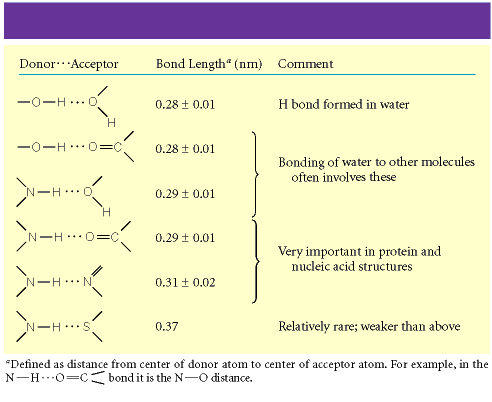
Bond Lengths and Types
Donor···Acceptor | Bond Length (nm) | Comment |
|---|---|---|
O—H···O | 0.28 ± 0.01 | H bond formed in water |
O—H···O=C | 0.28 ± 0.01 | Bonding of water to other molecules |
N—H···O | 0.29 ± 0.01 | Very important in protein and nucleic acid structures |
N—H···O=C | 0.29 ± 0.01 | Very important in protein and nucleic acid structures |
N—H···N | 0.31 ± 0.02 | Relatively rare; weaker than above |
N—H···S | 0.37 | Relatively rare; weaker than above |
Summary of Key Concepts
Amino acids are the monomers of proteins, each with unique side chains that determine their properties.
Peptide bonds link amino acids into polypeptide chains, with partial double-bond character restricting rotation.
Protein folding is driven by hydrophobic, hydrophilic, ionic, covalent, and hydrogen bonding interactions among amino acid side chains.
Hydrogen bonds are essential for stabilizing protein secondary and tertiary structures.
