 Back
BackAmino Acids and Protein Structure: Foundations for Biochemistry
Study Guide - Smart Notes

Proteins: Amino Acids and Structure
Definitions and Hierarchy of Protein Components
Proteins are large biological polymers composed of one or more chains of amino acids. The basic building blocks of proteins are amino acids, which are linked together by peptide bonds to form peptides and polypeptides. Understanding the structure and classification of amino acids is fundamental to biochemistry.
Protein: Large biological molecule consisting of one or more polypeptide chains.
Peptide: Short chain of amino acids (typically < 25 residues) linked by peptide bonds.
Amino Acid: Organic compound containing an amine group, a carboxylic acid group, and a unique side chain (R group).
Protein Digestion in Humans
Proteins are digested in a two-step process: first, they are denatured in the acidic environment of the stomach and hydrolyzed by pepsin into peptides. These peptides are further broken down in the intestine by various proteases, ultimately yielding free amino acids for absorption.
Common Structure of Amino Acids
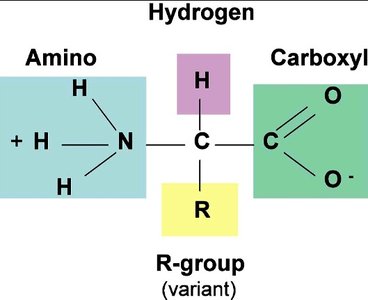
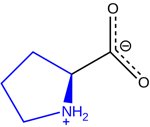
All amino acids share a common backbone structure, consisting of an alpha carbon bonded to an amino group, a carboxyl group, a hydrogen atom, and a variable R group.
Amine group: pKa ~9-10
Carboxyl group: pKa ~1.5-2.5
R group: Determines the unique properties of each amino acid
Properties and Classification of Amino Acids
Amino acids are classified based on their side chains (R groups), which influence their chemical properties and roles in proteins.
Acidic: Aspartate, Glutamate
Basic: Lysine, Arginine, Histidine
Hydroxyl: Serine, Threonine, Tyrosine (can be phosphorylated)
Sulfur: Cysteine, Methionine
Aliphatic: Glycine, Alanine, Valine, Leucine, Isoleucine
Aromatic: Phenylalanine, Tyrosine, Tryptophan
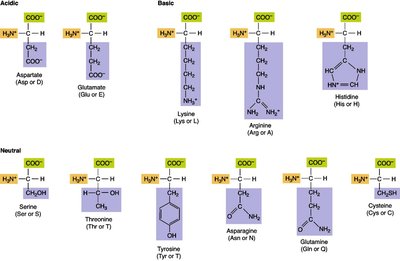
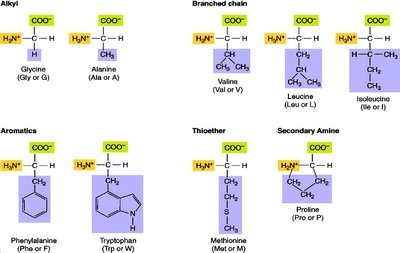
Table: Amino Acid Properties
The following table summarizes the pKa values and isoelectric points (pI) for common amino acids:
Amino Acid | Abbreviation | pK1 (-COOH) | pK2 (-NH3+) | pKR (R group) | pI |
|---|---|---|---|---|---|
Alanine | Ala (A) | 2.34 | 9.69 | - | 6.00 |
Arginine | Arg (R) | 2.17 | 9.04 | 12.48 | 10.76 |
Asparagine | Asn (N) | 2.02 | 8.80 | - | 5.41 |
... | ... | ... | ... | ... | ... |
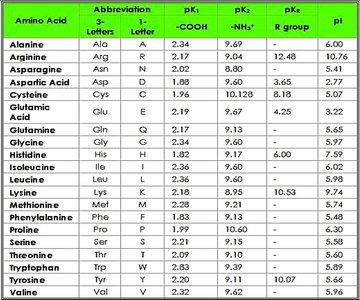
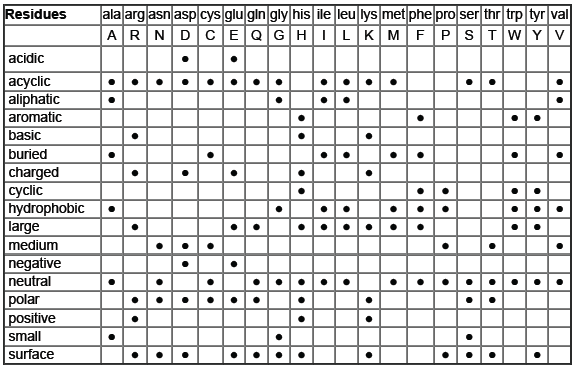
Table: Amino Acid Residue Properties
This table classifies amino acids by their chemical and physical properties:
Residues | ala | arg | asn | asp | cys | glu | gln | gly | his | ile | leu | lys | met | phe | pro | ser | thr | trp | tyr | val |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
acidic | • | • | ||||||||||||||||||
basic | • | • | • | |||||||||||||||||
... | ... | ... | ... | ... | ... | ... | ... | ... | ... | ... | ... | ... | ... | ... | ... | ... | ... | ... | ... |
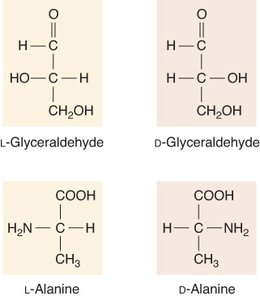
Stereochemistry of Amino Acids
The alpha carbon of amino acids is chiral (except for glycine), giving rise to L- and D- forms. All natural amino acids in proteins are L-form (levo).
Essential vs. Nonessential Amino Acids
Amino acids are classified based on their ability to be synthesized in the human body:
Essential: Must be obtained from diet (e.g., Histidine, Isoleucine, Leucine, Lysine, Methionine, Phenylalanine, Threonine, Tryptophan, Valine)
Nonessential: Can be synthesized in the body (e.g., Alanine, Arginine, Aspartic acid, Asparagine, Cysteine, Glutamic acid, Glutamine, Glycine, Proline, Serine, Tyrosine)
Ketogenic vs. Glucogenic Amino Acids
Amino acids are also classified based on their catabolic products:
Ketogenic: Leucine, Lysine (breakdown into Acetyl CoA or Acetoacetyl CoA)
Glucogenic: Most other amino acids (breakdown into intermediates of sugar metabolism)
Both: Isoleucine, Threonine, Phenylalanine, Tyrosine, Tryptophan
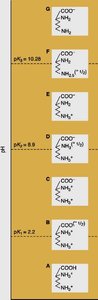
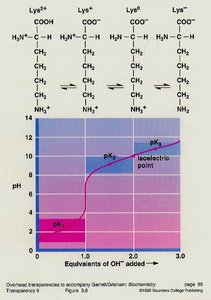
Acid-Base Properties and Isoelectric Point
Amino acids possess multiple dissociable groups, each with its own pKa. The isoelectric point (pI) is the pH at which the amino acid exists as a zwitterion (net charge zero).
Zwitterion: Molecule with both positive and negative charges, but overall neutral.
pI calculation: For amino acids without dissociable R groups:
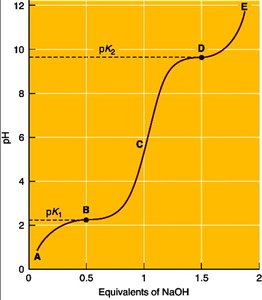
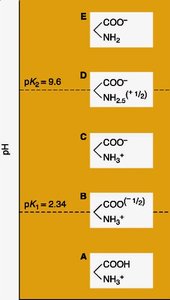
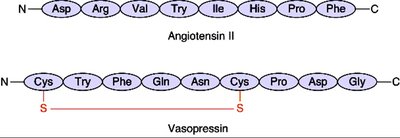
Examples of Peptides
Short peptides such as angiotensin II and vasopressin demonstrate the diversity and biological activity of peptide chains.
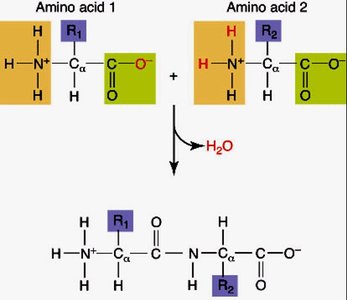
The Peptide Bond
The peptide bond is a covalent bond formed between the amino group of one amino acid and the carboxyl group of another via a dehydration reaction. Peptide bonds exhibit partial double bond character due to resonance, restricting rotation and stabilizing protein structure.
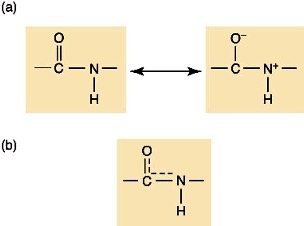
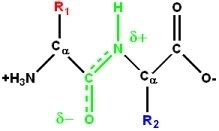
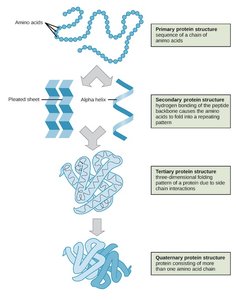
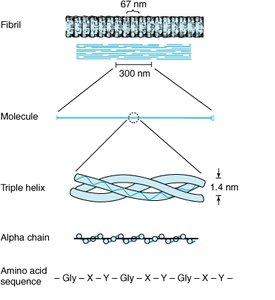
Protein Structure: Levels of Organization
Protein structure is organized into four hierarchical levels:
Primary: Sequence of amino acids
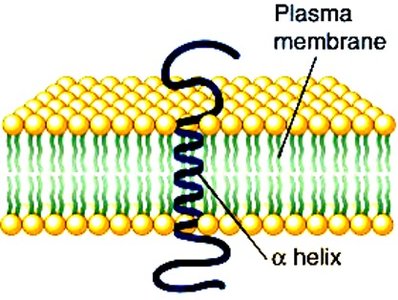
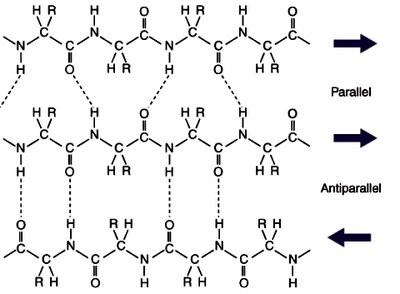
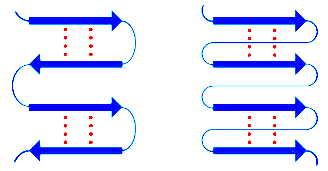
Secondary: Local 3D structures (alpha helix, beta sheet, coil/turn)
Tertiary: Overall 3D structure of a single polypeptide
Quaternary: Arrangement of multiple polypeptide chains
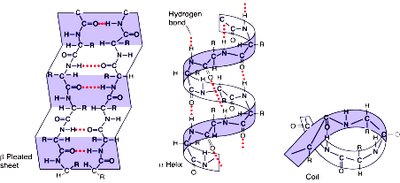
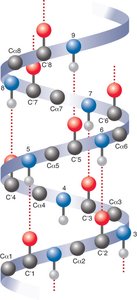
Secondary Protein Structure
Secondary structures are stabilized by hydrogen bonds and include alpha helices, beta sheets, and loops/turns. Certain amino acids, such as glycine and proline, influence the formation and stability of these structures.
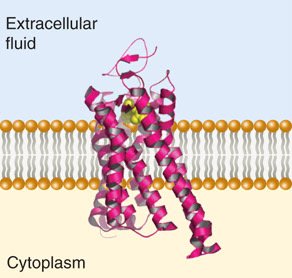
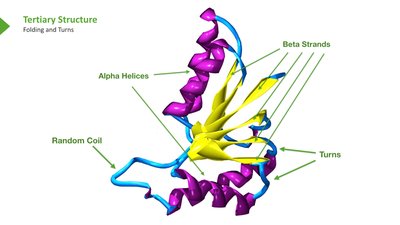
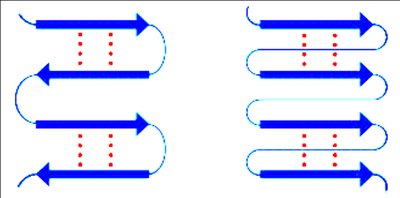
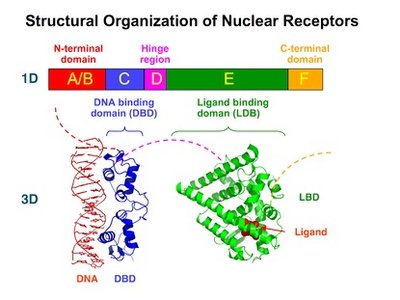
Protein Domains and Motifs
Protein domains are compact structural units within proteins, often responsible for specific functions such as ligand binding, DNA interaction, or dimerization. Domains are fundamental units of tertiary structure and drive protein folding.
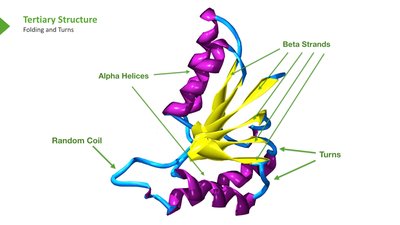
Tertiary and Quaternary Structure
Tertiary structure refers to the overall 3D shape of a single polypeptide, determined by interactions among side chains. Quaternary structure describes the arrangement of multiple polypeptide chains in a protein complex (e.g., hemoglobin).
Protein Folding and Chaperones
Proper protein folding is essential for function. Chaperone proteins assist in folding and refolding, preventing aggregation and misfolding. Examples include heat shock proteins and GroEL/GroES complexes.
Ligand Binding and Protein Configuration
Binding of ligands such as oxygen to myoglobin and hemoglobin induces conformational changes, affecting protein function and oxygen affinity. Hemoglobin exhibits cooperative binding (Hill kinetics), while myoglobin follows Michaelis-Menten kinetics.
Bohr Effect and Oxygen Transport
The Bohr effect describes the inverse relationship between hemoglobin's oxygen affinity and acidity/CO2 concentration. In active tissues, increased CO2 and lower pH promote oxygen release from hemoglobin, facilitating delivery to tissues.
Methemoglobinemia
Methemoglobinemia is a condition where hemoglobin's iron is oxidized to Fe3+, reducing its oxygen-carrying capacity and shifting the dissociation curve, impairing oxygen delivery to tissues.
Summary Table: Factors Affecting Oxygen Binding
Factor | Effect on O2 Binding |
|---|---|
CO2 (high) | Decreases O2 affinity (promotes release) |
pH (low) | Decreases O2 affinity (promotes release) |
2,3-BPG | Decreases O2 affinity |
Temperature (high) | Decreases O2 affinity |
CO | Increases O2 affinity (inhibits release) |
Key Equations
Isoelectric Point:
Michaelis-Menten Equation:
Hill Equation:
Coulomb's Law:
Additional info:
Protein structure and function are tightly linked; changes in structure (denaturation, ligand binding, post-translational modification) can activate or inhibit function.
Chaperones are essential for maintaining protein homeostasis, especially under stress conditions.
Understanding amino acid properties is foundational for predicting protein structure, function, and interactions.
