 Back
BackAmino Acids: Structure, Classification, and Biochemical Roles
Study Guide - Smart Notes

Amino Acids: Structure and Function
Introduction to Amino Acids
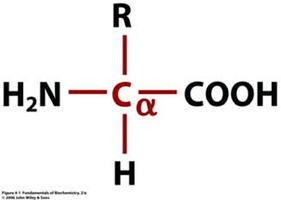
Amino acids are the fundamental building blocks of proteins, playing critical roles in nearly all biological processes. Each amino acid contains a central alpha carbon (Cα) bonded to an amino group, a carboxyl group, a hydrogen atom, and a unique side chain (R group) that determines its properties.
Definition: Organic compounds containing both amino (-NH2) and carboxyl (-COOH) functional groups attached to the same carbon atom.
General Structure: The alpha carbon is the central atom to which the amino group, carboxyl group, hydrogen, and R group are attached.
Biological Importance: Amino acids are precursors for proteins, neurotransmitters, hormones, and other biomolecules.
Stereochemistry of Amino Acids
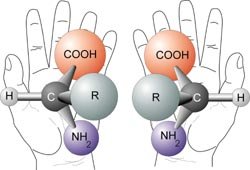
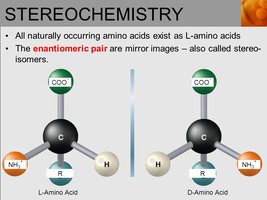
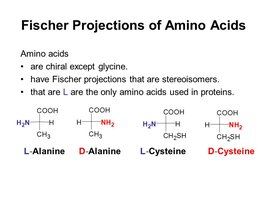
Most amino acids (except glycine) are chiral, existing as L- and D-enantiomers. In biological systems, only L-amino acids are incorporated into proteins.
Chirality: The presence of four different groups attached to the alpha carbon makes amino acids (except glycine) chiral.
Enantiomers: L- and D-forms are mirror images; only L-forms are used in ribosomal protein synthesis.
Classification of Amino Acids
Amino acids are classified based on the properties of their side chains (R groups), which influence their chemical behavior and role in proteins.
Nonpolar (Hydrophobic): R groups are alkyl or aromatic (e.g., glycine, alanine, valine, leucine, isoleucine, phenylalanine, tryptophan, methionine, proline).
Polar (Uncharged): R groups contain alcohol, thiol, or amide (e.g., serine, threonine, cysteine, tyrosine, asparagine, glutamine).
Acidic: R group contains a carboxylic acid (e.g., aspartic acid, glutamic acid).
Basic: R group contains an amine (e.g., lysine, arginine, histidine).
Special Classes: Hydroxy, sulfur-containing, aromatic, heterocyclic, branched-chain, and imino acids (proline).
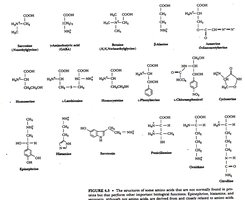
Structures of the 20 Standard Amino Acids
The 20 standard amino acids are encoded by the genetic code and are the primary building blocks of proteins.
Alpha-amino acids: All proteinogenic amino acids are alpha-amino acids.
Structural Diversity: The side chains (R groups) confer unique chemical properties to each amino acid.
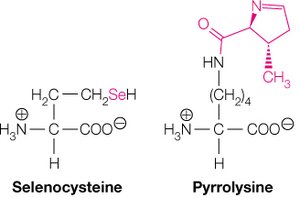
Non-Standard and Derived Amino Acids
Selenocysteine and Pyrrolysine
In addition to the 20 standard amino acids, selenocysteine and pyrrolysine are incorporated into proteins in certain organisms, known as the 21st and 22nd amino acids.
Selenocysteine (Sec): Analog of cysteine with selenium replacing sulfur; incorporated at UGA codons.
Pyrrolysine (Pyl): Derivative of lysine with a pyrroline ring; found in some archaea and bacteria.
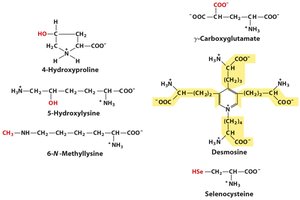
Post-Translationally Modified Amino Acids
Some amino acids are modified after translation, resulting in non-standard amino acids with specialized functions.
Examples: 4-hydroxyproline, 5-hydroxylysine, 6-N-methyllysine, γ-carboxyglutamate, desmosine.
Biological Roles: These modifications are critical for protein function, stability, and regulation.
Non-Protein Amino Acids
Some amino acids are not incorporated into proteins but have important metabolic or signaling roles (e.g., homoserine, homocysteine, β-alanine, γ-aminobutyric acid/GABA).
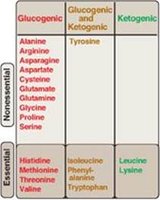
Essential and Nonessential Amino Acids
Essential Amino Acids
Essential amino acids cannot be synthesized by the human body and must be obtained from the diet. Nonessential amino acids can be synthesized endogenously.
Essential: Histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, valine, and (conditionally) arginine.
Nonessential: Alanine, asparagine, aspartate, cysteine, glutamate, glutamine, glycine, proline, serine, tyrosine.
Conditionally Essential: Arginine (in children), tyrosine (if phenylalanine is deficient).

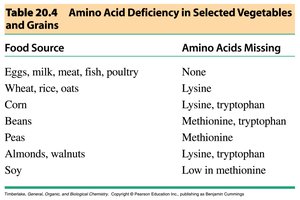
Dietary Sources and Protein Quality
Complete proteins contain all essential amino acids and are typically found in animal products. Plant proteins may lack one or more essential amino acids, requiring dietary variety for vegetarians.
Food Source | Amino Acids Missing |
|---|---|
Eggs, milk, meat, fish, poultry | None |
Wheat, rice, oats | Lysine |
Corn | Lysine, tryptophan |
Beans | Methionine, tryptophan |
Peas | Methionine |
Almonds, walnuts | Lysine, tryptophan |
Soy | Low in methionine |
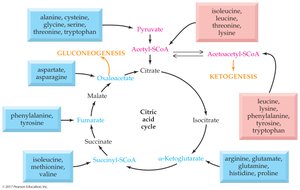
Metabolic Classification of Amino Acids
Glucogenic and Ketogenic Amino Acids
Amino acids are classified based on their catabolic end products: glucogenic amino acids yield glucose precursors, while ketogenic amino acids yield ketone bodies or fatty acids.
Glucogenic: Yield pyruvate or TCA cycle intermediates (e.g., alanine, aspartate, glutamine).
Ketogenic: Yield acetyl-CoA or acetoacetyl-CoA (e.g., leucine, lysine).
Both: Some amino acids are both glucogenic and ketogenic (e.g., isoleucine, phenylalanine, tryptophan, tyrosine).
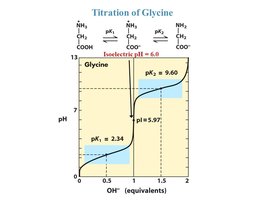
Acid-Base Properties of Amino Acids
Zwitterions and Isoelectric Point (pI)
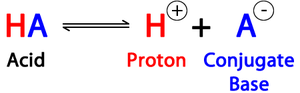
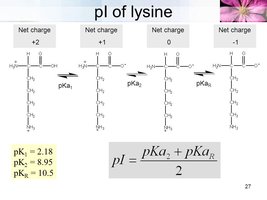
Amino acids can exist as zwitterions, molecules with both positive and negative charges but a net charge of zero. The isoelectric point (pI) is the pH at which the amino acid has no net charge.
Zwitterion: At physiological pH, the amino group is protonated and the carboxyl group is deprotonated.
Isoelectric Point (pI): The pH at which the amino acid does not migrate in an electric field.
Henderson-Hasselbalch Equation
The Henderson-Hasselbalch equation describes the relationship between pH, pKa, and the ratio of protonated to deprotonated forms of an acid.
Equation:
Application: Used to calculate the pH at which an amino acid exists in a particular ionic form.

Acid-Base Behavior of Amino Acids
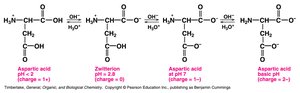
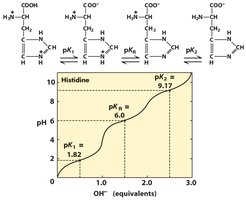
The ionization state of amino acids depends on the pH of the solution relative to their pKa values. Acidic and basic amino acids have additional ionizable groups, resulting in more complex titration curves and multiple pKa values.
Acidic Amino Acids: Aspartic acid and glutamic acid have low pI values due to extra carboxyl groups.
Basic Amino Acids: Lysine, arginine, and histidine have high pI values due to extra amino groups.
Separation and Reactions of Amino Acids
Electrophoresis
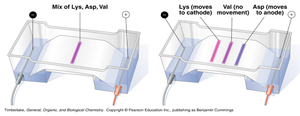
Electrophoresis separates amino acids based on their isoelectric points. Amino acids with different charges migrate toward opposite electrodes in an electric field.
Principle: At pH below pI, amino acids are positively charged and move toward the cathode; above pI, they are negatively charged and move toward the anode.
Application: Used for analytical separation and identification of amino acids.
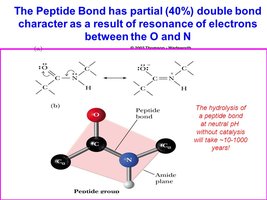
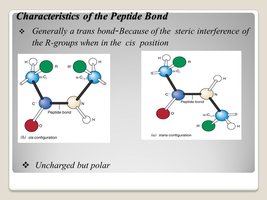
Peptide Bond Formation
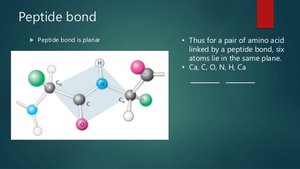
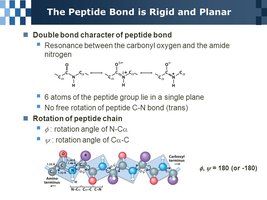
A peptide bond is a covalent amide bond formed between the carboxyl group of one amino acid and the amino group of another, releasing water. Peptide bonds are planar, rigid, and have partial double-bond character due to resonance.
Formation:
Properties: Planar, limited rotation, usually trans configuration.

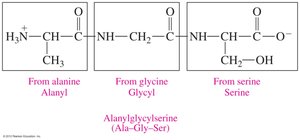
Naming Peptides
Peptides are named from the N-terminus to the C-terminus, with -yl endings for all but the last amino acid.
Biochemical Reactions of Amino Acids
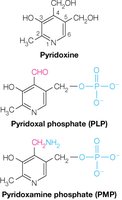
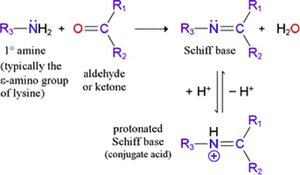
Transamination-requires PLP
Transamination is the reversible transfer of an amino group from an amino acid to an α-keto acid, catalyzed by aminotransferases with pyridoxal phosphate (PLP) as a coenzyme.
Importance: Central to amino acid metabolism and synthesis of nonessential amino acids.
Coenzyme: PLP forms a Schiff base with the amino acid, facilitating the transfer.

Oxidative Deamination
Oxidative deamination removes the amino group from amino acids as ammonia (NH4+), primarily via glutamate dehydrogenase. The ammonia enters the urea cycle for excretion.
Post-Translational Modifications
Methylation and Acetylation
Protein methylation (on lysine or arginine) and acetylation (on N-terminal residues or lysine side chains) are common modifications affecting protein function, stability, and gene regulation.
Methylation: Catalyzed by methyltransferases using S-adenosyl methionine (SAM) as the methyl donor.
Acetylation: Catalyzed by acetyltransferases, important in histone modification and gene expression.
Summary Table: Properties of Common Amino Acids
Amino Acid | Abbreviation | pKa (COOH) | pKa (NH2) | pI |
|---|---|---|---|---|
Alanine | Ala, A | 2.3 | 9.7 | 6.0 |
Arginine | Arg, R | 2.2 | 9.0 | 10.8 |
Asparagine | Asn, N | 2.1 | 8.8 | 5.4 |
Aspartic acid | Asp, D | 2.1 | 9.8 | 2.8 |
Glutamic acid | Glu, E | 2.2 | 9.7 | 3.2 |
Glutamine | Gln, Q | 2.2 | 9.1 | 5.7 |
Glycine | Gly, G | 2.3 | 9.6 | 6.0 |
Histidine | His, H | 1.8 | 9.2 | 7.6 |
Isoleucine | Ile, I | 2.4 | 9.7 | 6.0 |
Leucine | Leu, L | 2.4 | 9.6 | 6.0 |
Lysine | Lys, K | 2.2 | 9.2 | 9.7 |
Methionine | Met, M | 2.3 | 9.2 | 5.7 |
Phenylalanine | Phe, F | 2.2 | 9.2 | 5.5 |
Proline | Pro, P | 2.0 | 10.6 | 6.3 |
Serine | Ser, S | 2.2 | 9.2 | 5.7 |
Threonine | Thr, T | 2.1 | 9.1 | 5.6 |
Tryptophan | Trp, W | 2.4 | 9.4 | 5.9 |
Tyrosine | Tyr, Y | 2.2 | 9.1 | 5.7 |
Valine | Val, V | 2.3 | 9.6 | 6.0 |
Clinical Correlations
Phenylketonuria (PKU)
PKU is a metabolic disorder caused by a deficiency in phenylalanine hydroxylase, leading to accumulation of phenylalanine and its metabolites, which can cause intellectual disability if untreated. Management involves dietary restriction of phenylalanine.
Maple Syrup Urine Disease (MSUD)
MSUD is caused by a deficiency in the branched-chain α-keto acid dehydrogenase complex, resulting in accumulation of leucine, isoleucine, and valine. Symptoms include sweet-smelling urine, neurological damage, and can be fatal without dietary management.
