 Back
BackAmino Acids: Structure, Properties, and Biological Roles
Study Guide - Smart Notes

Chapter 4: Amino Acids – Building Blocks of Proteins
Structure and Properties of Amino Acids
Amino acids are the fundamental units of proteins, sharing a common backbone structure but differing in their side chains (R groups). Their chemical diversity underlies the vast array of protein functions in biology.
General Structure: Each amino acid contains a central, tetrahedral α-carbon bonded to an amino group (–NH2), a carboxyl group (–COOH), a hydrogen atom, and a variable side chain (R group).
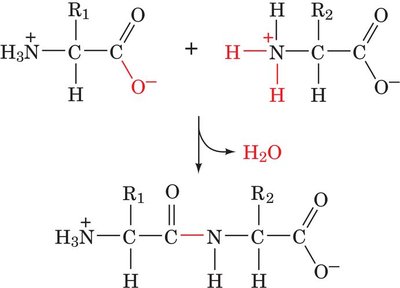
Peptide Bonds: Amino acids are linked by peptide bonds to form polypeptides and proteins.
Ionizable Groups: Some side chains contain ionizable groups with characteristic pKa values, influencing protein charge and function.
Rare Amino Acids: Some amino acids occur infrequently in proteins but have specialized roles.
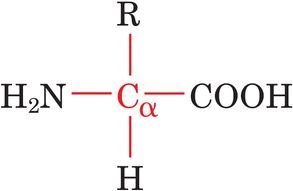
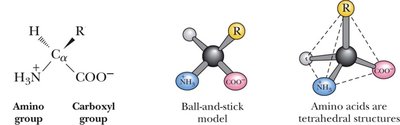
The 20 Common Amino Acids: Classification
The 20 standard amino acids are classified based on the properties of their side chains:
Nonpolar (Hydrophobic): Side chains are primarily hydrocarbons, making them water-insoluble. Examples: Alanine, Valine, Leucine, Isoleucine, Methionine, Proline, Phenylalanine, Tryptophan.
Polar, Uncharged: Side chains contain groups that can form hydrogen bonds but are not charged at physiological pH. Examples: Serine, Threonine, Asparagine, Glutamine, Cysteine, Tyrosine, Glycine.
Charged, Acidic: Side chains contain carboxyl groups and are negatively charged at physiological pH. Examples: Aspartic acid, Glutamic acid.
Charged, Basic: Side chains contain amino groups and are positively charged at physiological pH. Examples: Lysine, Arginine, Histidine.
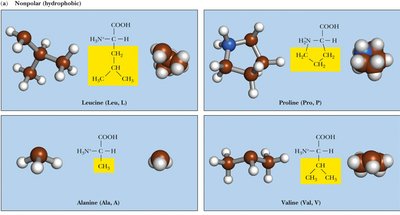
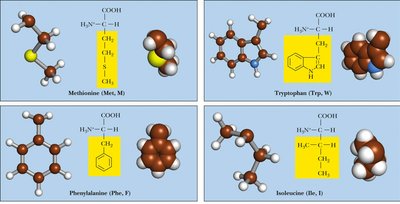
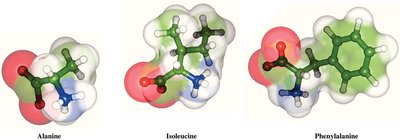
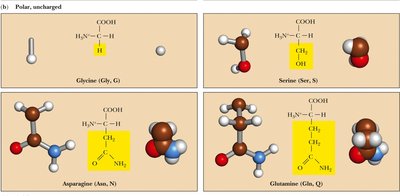
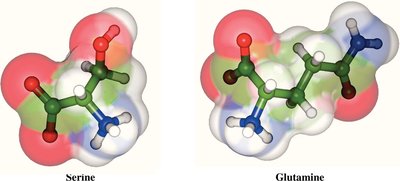
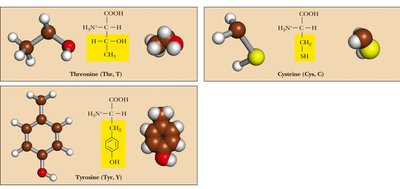
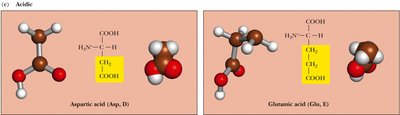
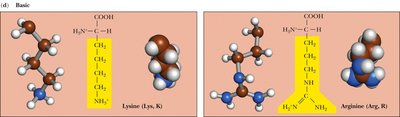
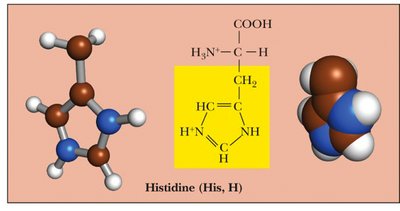
Table: Classification of Amino Acids by Side Chain Properties
Group | Examples | Key Features |
|---|---|---|
Nonpolar | Ala, Val, Leu, Ile, Met, Pro, Phe, Trp | Hydrophobic, often buried in protein cores |
Polar, Uncharged | Ser, Thr, Asn, Gln, Cys, Tyr, Gly | Hydrophilic, can form H-bonds |
Acidic | Asp, Glu | Negatively charged at pH 7 |
Basic | Lys, Arg, His | Positively charged at pH 7 |
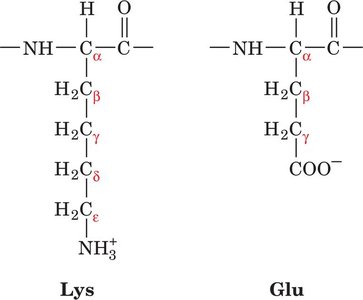
Naming the Side-Chain Carbons
Carbons in amino acid side chains are named using Greek letters (α, β, γ, δ, ε) starting from the α-carbon.
Acid-Base Properties and pKa Values
Amino acids can exist in different ionic forms depending on the pH of the environment. The α-carboxyl group (pKa ≈ 2) and α-amino group (pKa ≈ 9) are always present, while some side chains are also ionizable.
Zwitterion: At neutral pH, amino acids exist as zwitterions, with both the amino and carboxyl groups ionized.
pKa Values: The pKa of side chains determines their charge at physiological pH.
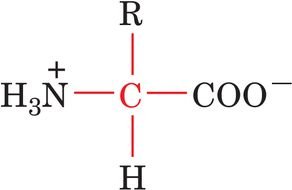
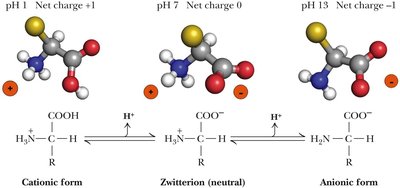
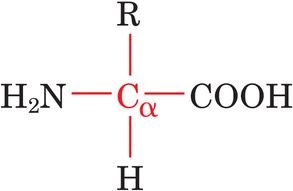
Table: pKa Values of Ionizable Groups
Group | pKa | Charge at pH 7 |
|---|---|---|
α-Carboxyl | 2 | – |
α-Amino | 9 | + |
Asp (β-carboxyl) | 3.9 | – |
Glu (γ-carboxyl) | 4.1 | – |
His (imidazole) | 6.0 | 0 |
Cys (SH) | 8.4 | 0 |
Tyr (OH) | 10.5 | 0 |
Lys (ε-amino) | 10.5 | + |
Arg (guanidino) | 12.5 | + |
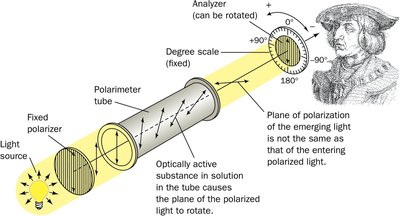
Stereochemistry of Amino Acids
Most amino acids are chiral, with the exception of glycine. The L-stereoisomer is the predominant form in proteins.
Chirality: The α-carbon is a stereocenter in all amino acids except glycine.
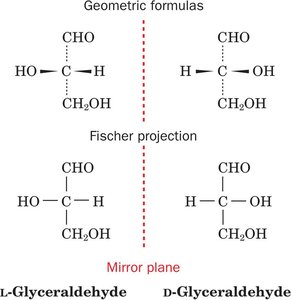
L- and D- Forms: The L-form is found in proteins; D-forms are rare in nature.
Fischer Projections: Used to depict absolute configuration.
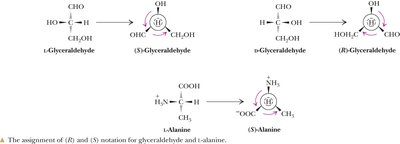
R,S System: Provides unambiguous naming, especially for amino acids with multiple chiral centers.

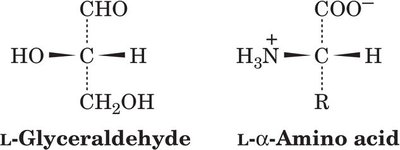
Spectroscopic Properties of Amino Acids
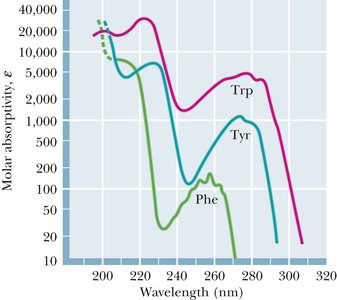
Certain amino acids, especially those with aromatic side chains, absorb ultraviolet light. This property is useful for protein quantification and structural studies.
UV Absorbance: Tryptophan (Trp), Tyrosine (Tyr), and Phenylalanine (Phe) absorb UV light, with maxima near 280 nm for Trp and Tyr.
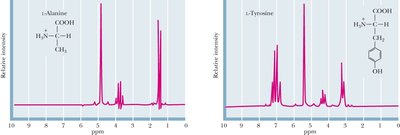
NMR Spectroscopy: Each amino acid has a characteristic NMR spectrum, useful for protein structure determination.
Reactions and Modifications of Amino Acids
Amino acid side chains can undergo various chemical modifications, which are important for protein function and regulation.
Schiff Bases and Amides: Amino groups can react to form Schiff bases; carboxyl groups can form amides and esters.
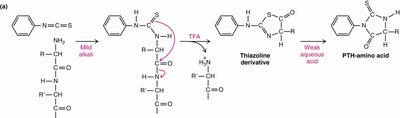
Edman Degradation: Edman’s reagent is used to sequence peptides by labeling the N-terminal amino acid.
Disulfide Bonds: Cysteine residues can form covalent disulfide bonds, stabilizing protein structure.
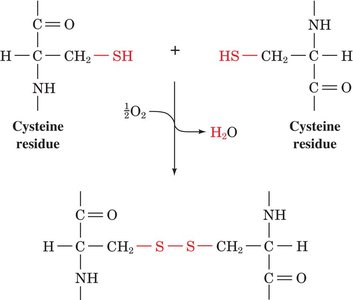
Biologically Active and Modified Amino Acids
Some amino acids and their derivatives serve specialized roles as neurotransmitters, hormones, or structural components. Proteins may also contain post-translationally modified amino acids.
Rare Amino Acids: Selenocysteine, pyrrolysine, hydroxylysine, hydroxyproline, carboxyglutamate, and pyroglutamate are examples of rare or modified amino acids.
Neurotransmitters and Hormones: GABA, epinephrine, histamine, and serotonin are amino acid derivatives with signaling roles.
Phosphorylation: Phosphorylated amino acids are key in cell signaling.
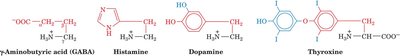
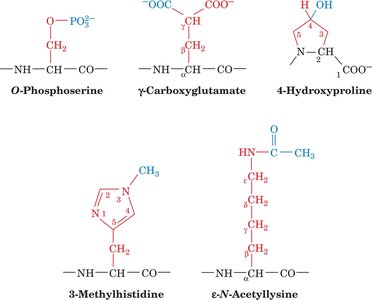
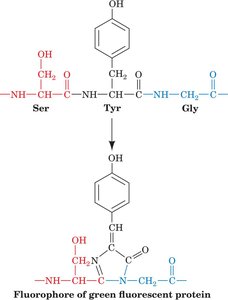
Green Fluorescent Protein (GFP) and Fluorescent Proteins
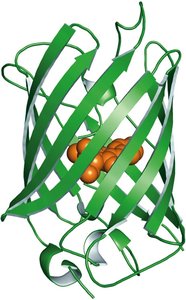
Green Fluorescent Protein (GFP) is a naturally fluorescent protein from the jellyfish Aequorea victoria. It is widely used as a molecular tag in cell biology and biotechnology.
Structure: GFP contains a chromophore formed from a specific sequence of amino acids within a β-barrel structure.
Applications: GFP and its variants are used to visualize proteins, organelles, and cells in living organisms.
Brainbow Technique: Expression of multiple fluorescent proteins enables visualization of individual neurons in different colors.

Summary Table: Key Amino Acid Properties
Amino Acid | 3-Letter Code | 1-Letter Code | Side Chain Property | pKa (side chain) |
|---|---|---|---|---|
Aspartic Acid | Asp | D | Acidic | 3.9 |
Glutamic Acid | Glu | E | Acidic | 4.1 |
Histidine | His | H | Basic | 6.0 |
Cysteine | Cys | C | Polar, uncharged | 8.4 |
Tyrosine | Tyr | Y | Polar, uncharged | 10.5 |
Lysine | Lys | K | Basic | 10.5 |
Arginine | Arg | R | Basic | 12.5 |
Key Equations
Henderson-Hasselbalch Equation:
Peptide Bond Formation (Condensation Reaction):
Example: Net Charge of a Tripeptide at Neutral pH
For a tripeptide Cys–Gly–Asn at pH 7:
Cys side chain is neutral at pH 7 (pKa = 8.4).
Gly and Asn side chains are neutral.
N-terminus (+1), C-terminus (–1): Net charge = 0.
Checkpoint Questions for Review
Draw and label the general structure of an amino acid, indicating the α-carbon and its substituents.
Classify the 20 standard amino acids by polarity, structure, and acid–base properties.
Explain the significance of pKa values for amino acid side chains.
Describe the difference between L- and D-amino acids and their biological relevance.
List common covalent modifications of amino acids in proteins and their functional consequences.
