 Back
BackMidterm
Study Guide - Smart Notes

Chemical Elements of Cells and Organisms
Major Elements in Living Systems
Living organisms are primarily composed of a few key elements: carbon (C), hydrogen (H), oxygen (O), and nitrogen (N). These elements form the backbone of biological macromolecules. Other essential elements include sulfur (S) and phosphorus (P), which are critical for protein structure and energy metabolism, respectively. Ions such as Na+, K+, Mg2+, Ca2+, and Cl- are also vital for cellular function.
Sulfur (S): Found in nearly all proteins.
Phosphorus (P): Essential for nucleic acids and energy transfer (e.g., ATP).
Inert gases (He, Ne): Abundant but not biologically active due to chemical inertness.
Biological Macromolecules
Classes and Monomeric Units
There are four major classes of biological macromolecules, each with distinct structures and functions:
Nucleic acids (DNA and RNA): Polymers of nucleotides.
Proteins: Polymers of amino acids.
Polysaccharides: Polymers of monosaccharides (carbohydrates).
Lipids: Not true polymers, but large complexes of fatty acids and other components.
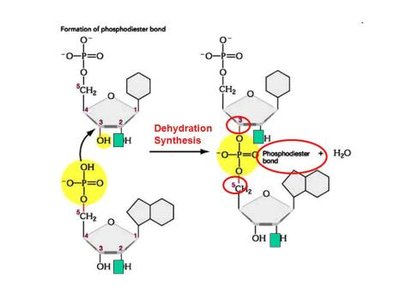
These macromolecules are assembled by dehydration synthesis (condensation), which removes a water molecule during the formation of covalent bonds between monomers.
Macromolecule | Monomer | Linkage |
|---|---|---|
Nucleic acids | Nucleotide | Phosphodiester |
Proteins | Amino acid | Peptide (amide) |
Polysaccharides | Monosaccharide | Glycoside (ether) |
Lipids (triacylglycerols) | Fatty acids | Ester |
Nucleic Acids
Structure and Function
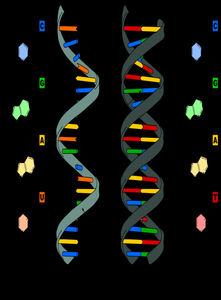
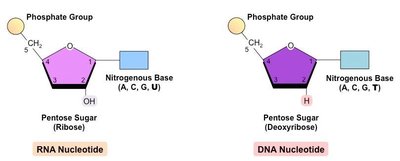
Nucleic acids (DNA and RNA) are polymers of nucleotides, each consisting of a sugar, a phosphate group, and a nitrogenous base. DNA stores genetic information, while RNA is involved in the expression and transmission of this information.
DNA: Double-stranded, contains deoxyribose, bases are A, T, G, C.
RNA: Single-stranded, contains ribose, bases are A, U, G, C.
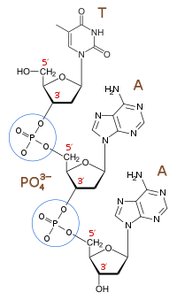
Nucleotides are joined by phosphodiester bonds between the 3' carbon of one sugar and the 5' carbon of the next.
DNA molecules are extremely large and complex, as seen in chromosomal DNA.
Proteins
Structure and Diversity
Proteins are polymers of 20 different amino acids, linked by peptide bonds. They exhibit remarkable structural diversity and perform a wide range of biological functions, including catalysis (enzymes), structural support, transport, signaling, and immune defense.
Primary structure: Linear sequence of amino acids.
Secondary structure: Local folding (α-helices, β-sheets).
Tertiary structure: Overall 3D shape of a single polypeptide.
Quaternary structure: Assembly of multiple polypeptide subunits.
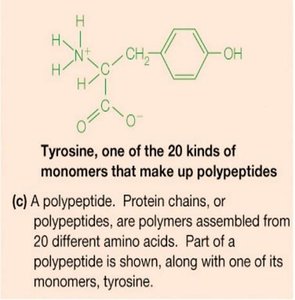
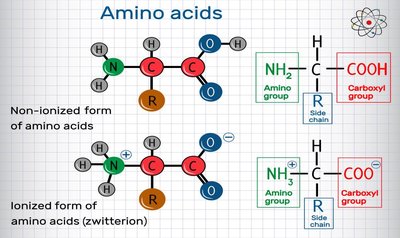
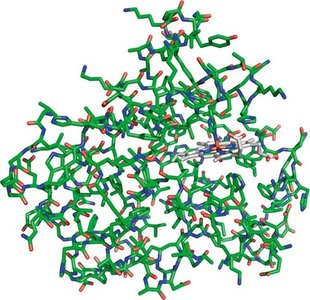
Polysaccharides (Carbohydrates)
Structure and Function
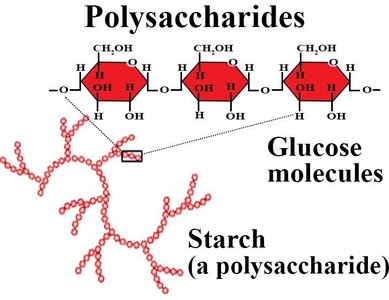
Polysaccharides are long chains of monosaccharide units. They serve as energy storage (e.g., starch, glycogen) and structural components (e.g., cellulose in plants).
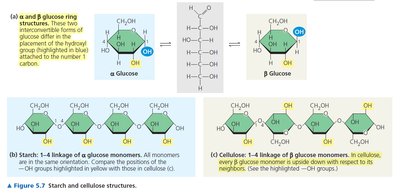
Starch: Main storage polysaccharide in plants; composed of α-1,4 glycosidic linkages.
Cellulose: Structural polysaccharide in plants; composed of β-1,4 glycosidic linkages.
Glycogen: Main storage polysaccharide in animals; highly branched.
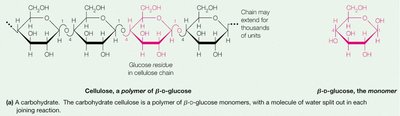
Lipids
Structure and Types
Lipids are a chemically diverse group of hydrophobic molecules, including fats, phospholipids, and steroids. They are essential for energy storage, membrane structure, and signaling.
Triacylglycerols (triglycerides): Energy storage molecules formed by esterification of three fatty acids to glycerol.
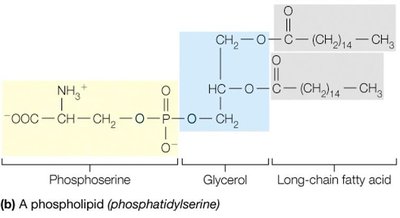
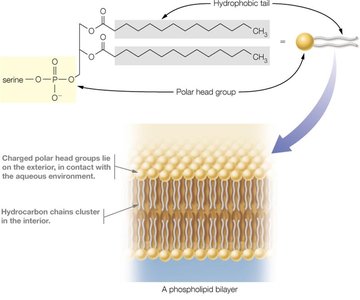
Phospholipids: Major components of cell membranes; amphipathic molecules with hydrophilic heads and hydrophobic tails.
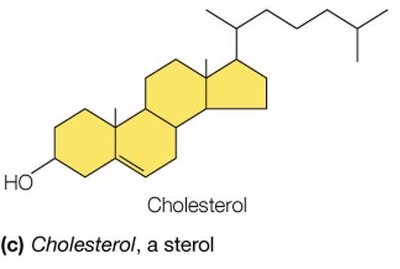
Sterols (e.g., cholesterol): Structural components of membranes and precursors to steroid hormones.
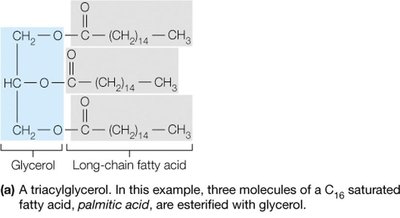
Membrane Formation
Phospholipids spontaneously form bilayers in aqueous environments, with hydrophobic tails facing inward and hydrophilic heads facing outward, forming the basis of biological membranes.
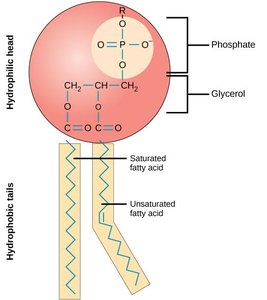
Cell Types and Organization
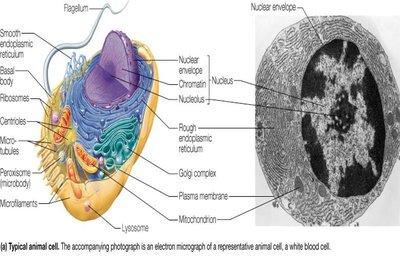
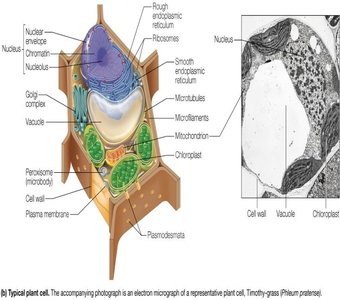
Prokaryotic and Eukaryotic Cells
Cells are the fundamental units of life. There are three major domains of life: Bacteria, Archaea (both prokaryotic), and Eukarya (eukaryotic). Prokaryotic cells lack membrane-bound organelles, while eukaryotic cells contain organelles such as the nucleus, mitochondria, and endoplasmic reticulum.
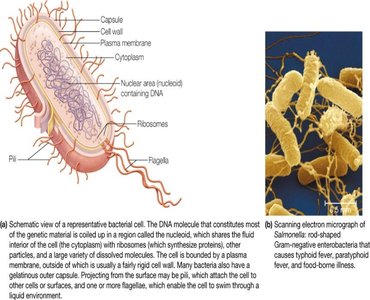
Noncovalent Interactions in Biomolecules
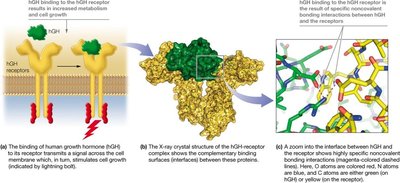
Types and Importance
Noncovalent interactions, including hydrogen bonds, ionic interactions, van der Waals forces, and hydrophobic effects, are crucial for the structure and function of biomolecules. They are weaker than covalent bonds but allow for dynamic molecular interactions essential for biological processes.
Summary Table: Macromolecules, Monomers, and Linkages
Macromolecule | Monomer | Bond/Linkage |
|---|---|---|
Nucleic Acid | Nucleotide | Phosphodiester |
Protein | Amino Acid | Peptide (Amide) |
Polysaccharide | Monosaccharide | Glycosidic (Ether) |
Lipid | Fatty Acid | Ester |
Key Chemical Reactions
Dehydration Synthesis (Condensation)
Macromolecules are assembled by dehydration synthesis, where a water molecule is removed to form a covalent bond between monomers.
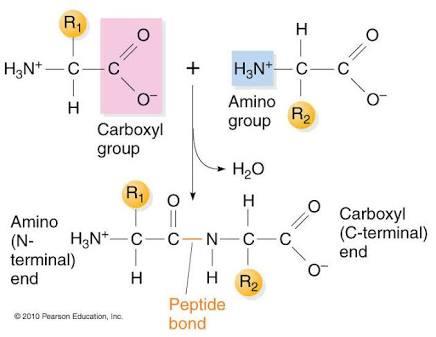
Peptide Bond Formation
Amino acids are linked by peptide bonds, formed by condensation between the carboxyl group of one amino acid and the amino group of another.
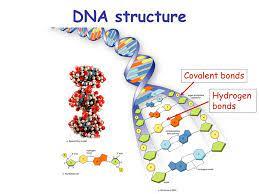
DNA Structure and Stability
Covalent and Noncovalent Bonds in DNA
DNA's backbone is stabilized by covalent phosphodiester bonds, while the double helix is stabilized by hydrogen bonds between complementary bases (A-T, G-C).
Conclusion
Understanding the structure, function, and assembly of biological macromolecules is fundamental to biochemistry. The interplay of covalent and noncovalent interactions, the diversity of monomeric units, and the organization of cells provide the molecular basis for life.
