 Back
BackBiological Membranes and Transport: Structure, Dynamics, and Function
Study Guide - Smart Notes

Biological Membranes and Transport
Principles of Membrane Structure
Biological membranes are fundamental to cellular structure and function, serving as selective barriers and platforms for biochemical processes. Their architecture is based on a lipid bilayer stabilized by the hydrophobic effect, with embedded proteins that perform diverse functions.
Lipid Bilayer: Composed of amphipathic lipids (glycerophospholipids, sphingolipids, sterols) that spontaneously form aggregates in water due to hydrophobic interactions.
Hydrophobic Effect: Drives the clustering of nonpolar regions to minimize exposure to water, stabilizing bilayers and vesicles.
Membrane Proteins: Embedded or associated with the bilayer, these proteins allow limited lateral motion and contribute to membrane function.
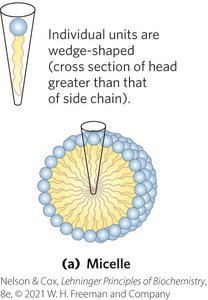
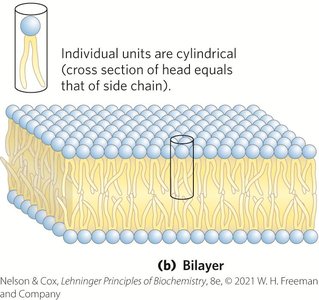
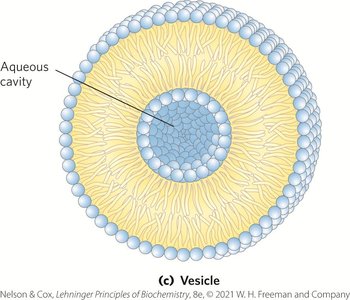
Membrane Architecture and Fluid Mosaic Model
The fluid mosaic model describes membranes as dynamic structures with lipids and proteins capable of lateral movement, maintaining permeability and flexibility.
Micelles: Spherical aggregates formed when the head group area is larger than the acyl chain area.
Bilayers: Two-dimensional sheets formed when head group and acyl chain areas are similar.
Vesicles (Liposomes): Hollow spheres formed by bilayer sheets folding back on themselves.
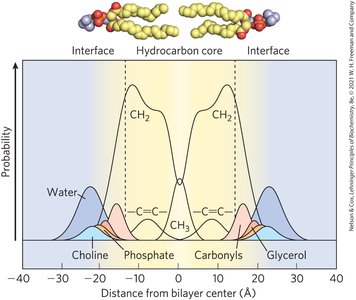
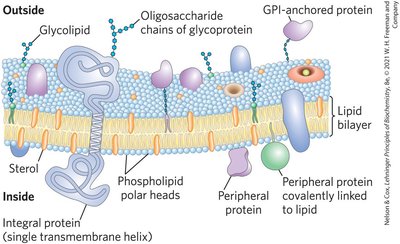
Functions of Biological Membranes
Membranes are essential for cellular processes, including shape changes, exocytosis, endocytosis, cell division, and molecular transport.
Transporters: Move specific solutes and ions across membranes.
Receptors: Sense extracellular signals and trigger cellular responses.
Ion Channels: Mediate electrical signaling.
Adhesion Molecules: Hold cells together.
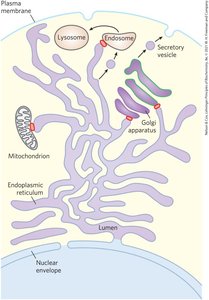
Endomembrane System and Membrane Trafficking
Internal membranes form a dynamic, interconnected system, with proteins and lipids synthesized in the ER and processed in the Golgi apparatus before being targeted to organelles or the plasma membrane.
Single Membrane: Surrounds ER, Golgi, lysosomes, vesicles.
Double Membrane: Surrounds nucleus, mitochondria, chloroplasts.
Membrane Trafficking: Involves covalent modification and sorting of proteins and lipids.
Lipid Rafts: Specialized regions with unique lipid and protein compositions.
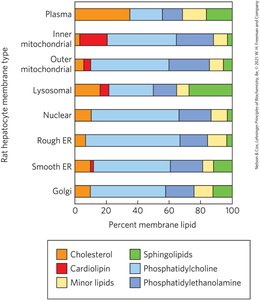
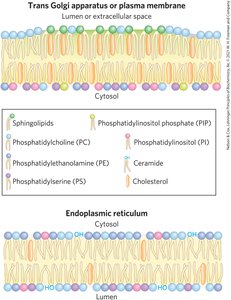
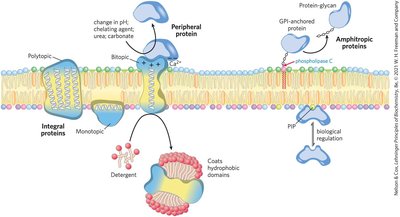
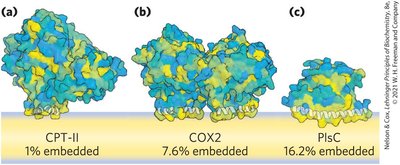
Membrane Protein Types and Associations
Membrane proteins are classified by their association with the bilayer:
Integral Proteins: Firmly embedded, often spanning the bilayer.
Peripheral Proteins: Associate via electrostatic interactions and hydrogen bonding.
Amphitropic Proteins: Reversibly associate with membranes, found in both membranes and cytosol.
Monotopic, Bitopic, Polytopic: Refer to the number of times a protein crosses the membrane.
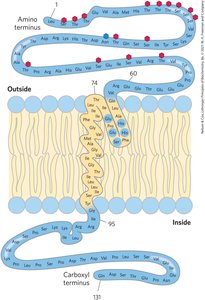
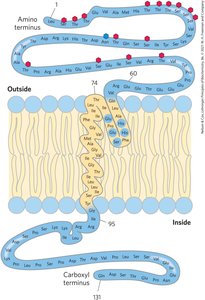
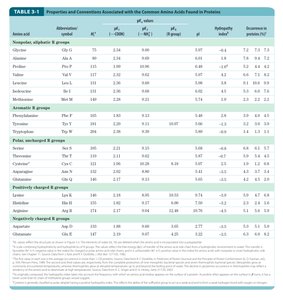
Membrane Protein Structure and Prediction
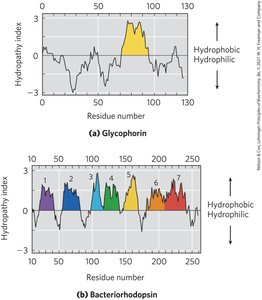
Integral membrane proteins often have α-helical segments long enough to span the bilayer. Hydropathy plots and indices help predict membrane-spanning regions.
Hydropathy Index: Measures free-energy change for moving an amino acid side chain from a hydrophobic environment to water.
Hydropathy Plot: Plots average hydropathy index against residue number to identify transmembrane segments.
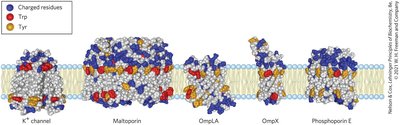
β-Barrel and Amino Acid Location in Membrane Proteins
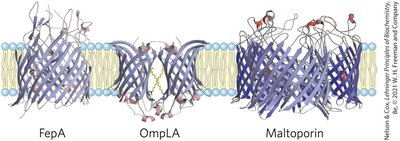
Some membrane proteins form β-barrel structures, especially in bacterial outer membranes. Amino acid location relative to the bilayer is critical for function.
β-Barrel: 20+ transmembrane segments form β-sheets lining a cylinder, stabilized by hydrogen bonds.
Porins: Allow polar solutes to cross bacterial membranes.
Positive-Inside Rule: Lys and Arg residues are more common on the cytoplasmic face.
Covalent Lipid Anchors and Membrane Protein Targeting
Some proteins are anchored or directed to membranes by covalently attached lipids, such as GPI anchors, and ionic attractions can enhance membrane affinity.
Membrane Dynamics and Fluidity
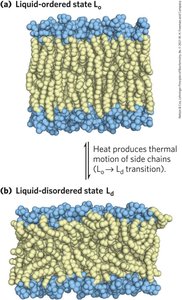
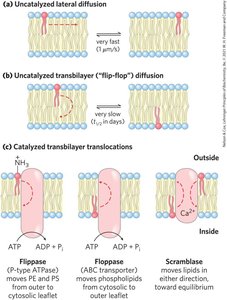
Membrane fluidity is influenced by fatty acid composition and sterol content, with acyl groups ordered to varying degrees. Lipids and proteins diffuse laterally, and transbilayer movement requires catalysis.
Liquid-Ordered (Lo) State: Gel-like, constrained motion.
Liquid-Disordered (Ld) State: Constant motion of hydrocarbon chains.
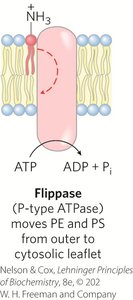
Flippases, Floppases, Scramblases: Enzymes catalyzing transbilayer movement of lipids.
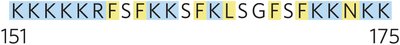
Lateral Diffusion and Membrane Rafts
Lipids and proteins undergo lateral diffusion, with microdomains (rafts) forming clusters of cholesterol and sphingolipids, making the bilayer thicker and more ordered.
Hop Diffusion: Lipid molecules diffuse within small regions, movement between regions is rare.
Lipid Rafts: Enriched in proteins with long-chain saturated fatty acids and GPI anchors.
Caveolae: Specialized rafts formed by caveolin, inducing membrane curvature.
Membrane Curvature and Fusion
Membrane curvature and fusion are central to processes such as vesicle trafficking, cell division, and neurotransmitter release. Proteins like BAR domains and septins induce or recognize curvature, while SNAREs mediate membrane fusion.
Solute Transport Across Membranes
Membranes are impermeable to charged or polar solutes, requiring transporters and ion channels for transmembrane movement. Transport can be passive or active, driven by concentration or electrochemical gradients.
Passive Transport: Facilitated diffusion down a gradient.
Active Transport: Moves substrates against a gradient, using energy from ATP or coupled transport.
Electrochemical Gradient: Determines direction of charged solute movement.
Transporter and Ion Channel Mechanisms
Transporters reduce activation energy for diffusion by forming noncovalent interactions and providing hydrophilic pathways. Ion channels allow rapid diffusion of ions, often gated by biological signals.
Glucose Transport and Kinetics
Glucose enters erythrocytes via GLUT1, a passive transporter. The process is analogous to enzyme kinetics and follows a Michaelis-Menten-like equation:
Equation:
GLUT1: Integral membrane protein with 12 transmembrane helices.
GLUT Family: Different isoforms with tissue-specific expression and kinetic properties.
Active Transport and ATPases
Active transporters include P-type, V-type, F-type ATPases, and ABC transporters, each with distinct mechanisms and substrate specificities.
P-type ATPases: Undergo phosphorylation during transport cycles (e.g., Na+/K+ ATPase).
V-type ATPases: Acidify intracellular compartments.
F-type ATPases: Drive ATP synthesis using proton gradients.
ABC Transporters: Use ATP to transport a wide variety of substrates.
Free-Energy Equations for Transport
General Free-Energy Change:
Uncharged Solute:
Ion Transport:
Specialized Transport Systems
Cotransport: Simultaneous transport of two solutes (antiport or symport).
Uniport: Transport of a single substrate.
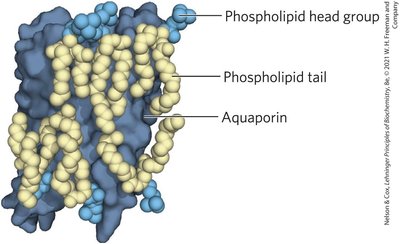
Aquaporins: Provide channels for water movement across membranes.
Ion-Selective Channels: Allow rapid movement of specific ions, often gated by ligands or voltage.
Summary Table: Glucose Transporters in Humans
Transporter | Tissue(s) where expressed | Kt (mM) | Role/characteristics |
|---|---|---|---|
GLUT1 | Erythrocytes, blood-brain barrier, placenta, most tissues | 3 | Basal glucose uptake; defective in De Vivo disease |
GLUT2 | Liver, pancreatic islets, intestine, kidney | 17 | Removal of excess glucose from blood; regulation of insulin release |
GLUT3 | Brain (neuron), testis (sperm) | 1.4 | Basal glucose uptake; high turnover number |
GLUT4 | Muscle, fat, heart | 5 | Activity increased by insulin |
GLUT5 | Intestine, testis, kidney | 6 | Primarily fructose transport |
Summary Table: ABC Transporters in Humans
Gene(s) | Role/characteristics |
|---|---|
ABCA1 | Reverse cholesterol transport; defect causes Tangier disease |
ABCB1 | Multidrug resistance P-glycoprotein 1; transport across blood-brain barrier |
ABCC7 | CFTR (Cl⁻ channel); defect causes cystic fibrosis |
ABCG2 | Breast cancer resistance protein; major exporter of anticancer drugs |
Summary Table: Aquaporins in Mammals
Aquaporin | Permeant | Tissue distribution | Primary subcellular distribution |
|---|---|---|---|
AQP1 | Water (high) | Erythrocyte, kidney, lung, vascular endothelium, brain, eye | Plasma membrane |
AQP2 | Water (high) | Kidney, vas deferens | Apical plasma membrane, intracellular vesicles |
AQP3 | Water (high), glycerol (high), urea (moderate) | Kidney, skin, lung, eye, colon | Basolateral plasma membrane |
Key Equations
Michaelis-Menten-like for Transport:
Free-Energy Change:
Uncharged Solute Transport:
Ion Transport:
Additional info: Academic context and explanations have been expanded for clarity and completeness. Only directly relevant images have been included to reinforce key concepts.
