 Back
BackBuffer Systems in Blood and Hemoglobin Function: Biochemistry Study Notes
Study Guide - Smart Notes

Buffer Systems in Blood
Introduction to Buffer Systems
Buffer systems are essential for maintaining the pH of body fluids within a narrow range, which is critical for proper physiological function. The primary buffer system in blood involves the equilibrium between carbon dioxide (CO2), carbonic acid (H2CO3), bicarbonate ion (HCO3-), and hydrogen ion (H+).
CO2 Production: CO2 is a major metabolic product from the oxidation of ingested carbon compounds.
Carbonic Anhydrase: This enzyme in red blood cells catalyzes the conversion of CO2 to H2CO3, which then dissociates into HCO3- and H+.
Buffering: The generated H+ ions are buffered by hemoglobin and other proteins, minimizing changes in blood pH.
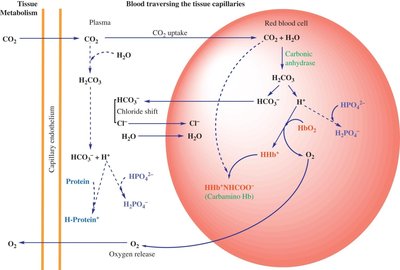
CO2 Transport in Blood
CO2 produced by tissues diffuses into blood, where it is transported in three main forms:
Dissolved CO2: A small fraction is transported physically dissolved in plasma.
Bicarbonate Ion (HCO3-): The majority of CO2 is converted to HCO3- in red blood cells and transported in plasma.
Carbaminohemoglobin: A minor fraction binds to the amino groups of hemoglobin, forming carbaminohemoglobin.
The following equilibrium is central to CO2 buffering:
Chloride Shift and Electrolyte Balance
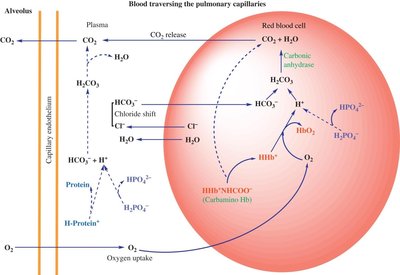
As HCO3- accumulates in red blood cells, it is transported out to plasma in exchange for Cl- ions, a process known as the chloride shift. This maintains electrostatic balance across the red blood cell membrane.
Buffering by Hemoglobin and Other Proteins
Hemoglobin acts as a major buffer by binding H+ ions generated from carbonic acid dissociation. Other proteins and phosphate also contribute to buffering capacity.
Hemoglobin Structure and Oxygen Binding
Heme Group and Oxygen Coordination
Hemoglobin contains a heme group, which consists of a porphyrin ring coordinated to an iron (Fe2+) atom. The iron atom binds oxygen reversibly, allowing hemoglobin to transport O2 from the lungs to tissues.
Coordination Sites: The iron atom is coordinated to four nitrogen atoms of the porphyrin ring, one nitrogen from a proximal histidine residue, and can bind O2 at the sixth coordination site.
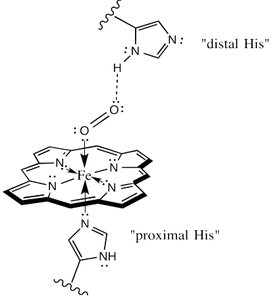
Conformational Changes Upon Oxygen Binding
Oxygen binding induces a conformational change in hemoglobin:
Deoxyhemoglobin: The iron atom is slightly out of the plane of the porphyrin ring.
Oxyhemoglobin: Oxygen binding pulls the iron atom into the plane, altering the protein's structure and increasing its affinity for additional O2 molecules (cooperativity).
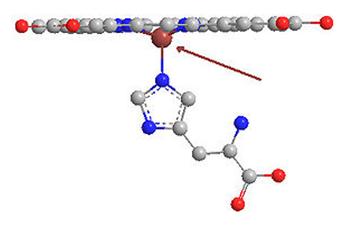
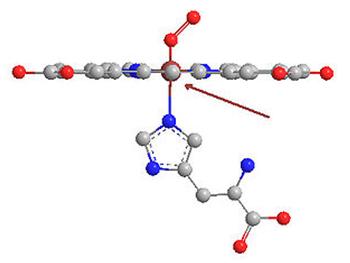
Bohr Effect and Regulation of Oxygen Binding
The Bohr Effect
The Bohr effect describes how increases in CO2 and H+ (lower pH) reduce hemoglobin's affinity for oxygen, facilitating O2 release in tissues where it is needed most.
Mechanism: H+ and CO2 bind to hemoglobin, stabilizing the deoxy form and promoting O2 release.
Physiological Importance: Enhances O2 delivery to metabolically active tissues producing more CO2 and H+.
Oxygen-Hemoglobin Dissociation Curve
The oxygen-hemoglobin dissociation curve is sigmoidal, reflecting cooperative binding. Factors such as pH, CO2, and temperature shift the curve:
Right Shift: Lower pH, higher CO2, or higher temperature decrease affinity, facilitating O2 release.
Left Shift: Higher pH, lower CO2, or lower temperature increase affinity, favoring O2 uptake.
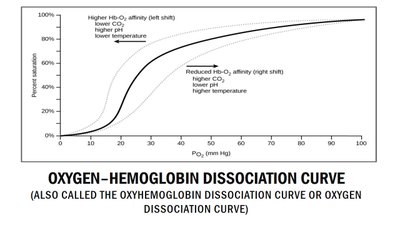
Summary Table: Factors Affecting Hemoglobin-Oxygen Affinity
Factor | Effect on Curve | Physiological Outcome |
|---|---|---|
Increased H+ (lower pH) | Right shift | O2 release favored |
Increased CO2 | Right shift | O2 release favored |
Increased temperature | Right shift | O2 release favored |
Decreased H+ (higher pH) | Left shift | O2 uptake favored |
Decreased CO2 | Left shift | O2 uptake favored |
Decreased temperature | Left shift | O2 uptake favored |
CO2 Transport and Exchange in Lungs and Tissues
Peripheral Tissues
In tissues, CO2 produced by metabolism diffuses into red blood cells, where it is converted to HCO3- and H+. HCO3- is transported to plasma, and H+ is buffered by hemoglobin.

Lungs
In the lungs, the process is reversed: HCO3- re-enters red blood cells, combines with H+ to form H2CO3, which is then converted to CO2 and exhaled.
Summary
Buffer systems, especially the bicarbonate buffer, are vital for blood pH regulation.
Hemoglobin plays a dual role in O2 transport and buffering H+ ions.
The Bohr effect and the oxygen-hemoglobin dissociation curve explain how O2 delivery is matched to tissue metabolic needs.
