 Back
BackCarbohydrates and Glycobiology: Structure, Function, and Analysis
Study Guide - Smart Notes

Carbohydrates: Structure and Classification
Definition and General Properties
Carbohydrates are organic molecules that function as aldehydes or ketones with at least two hydroxyl groups, or substances that yield such compounds upon hydrolysis. Many carbohydrates have the empirical formula (CH2O)n. They are essential for energy storage, structural integrity, and cellular communication.
Monosaccharides: Simple sugars consisting of a single polyhydroxy aldehyde or ketone unit (e.g., D-glucose).
Oligosaccharides: Short chains of monosaccharide units joined by glycosidic bonds.
Disaccharides: Oligosaccharides with two monosaccharide units (e.g., sucrose).
Polysaccharides: Sugar polymers with 10 or more monosaccharide units (e.g., cellulose, glycogen).
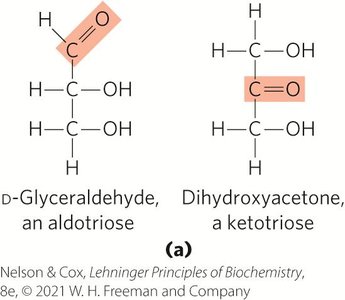
Principles of Carbohydrate Structure
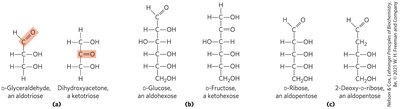
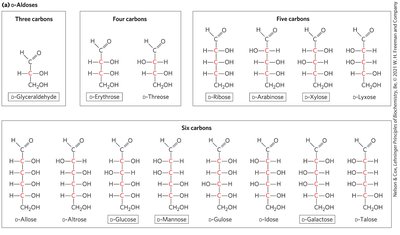
Principle 1: Carbohydrates can have multiple chiral carbons; the configuration of groups around each carbon atom determines interactions with other biomolecules. Biological evolution has selected the D-series for sugars.
Principle 2: Monomeric subunits (monosaccharides) serve as building blocks for large carbohydrate polymers. The specific sugar, linkage, and branching determine properties and function.
Principle 3: Storage of metabolites in polymeric form avoids high osmolarity that would result from storing them as individual monomers.
Principle 4: The sequences of complex polysaccharides are determined by biosynthetic enzymes, not by templates as in DNA, RNA, or proteins.
Principle 5: Polysaccharides assume three-dimensional structures with lowest-energy conformations, determined by covalent bonds, hydrogen bonds, charge interactions, and steric factors.
Principle 6: Molecular complementarity is central to function; recognition of oligosaccharides by lectins results from a perfect fit between lectin and ligand.
Principle 7: An almost infinite variety of discrete structures can be built from a small number of monomeric subunits.
Monosaccharides and Disaccharides
Stereoisomerism in Sugars
Sugar stereoisomers arise because many carbon atoms are chiral centers. Enzymes acting on sugars are stereospecific. A molecule with n chiral centers can have 2n stereoisomers.
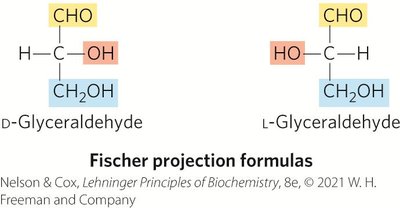
Aldoses and Ketoses
Monosaccharides have unbranched carbon chains with single bonds linking all carbon atoms. One carbon is double-bonded to oxygen (carbonyl group), others are bonded to hydroxyl groups.
Aldose: Carbonyl group at the end of the chain (aldehyde group).
Ketose: Carbonyl group at any other position (ketone group).
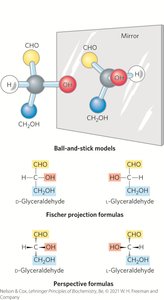
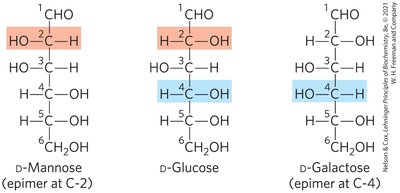
Epimers
Epimers are two sugars that differ only in the configuration around one carbon atom.
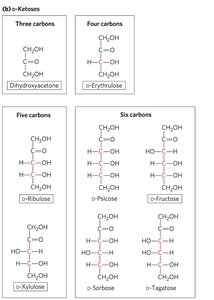
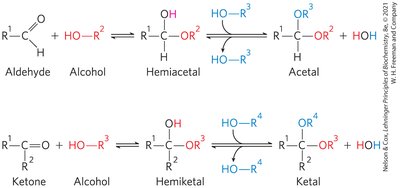
Cyclic Structures of Monosaccharides
In aqueous solution, aldotetroses and all monosaccharides with five or more backbone carbon atoms occur as cyclic structures. These are formed by covalent bonds between the carbonyl group and the oxygen of a hydroxyl group, resulting in hemiacetals or hemiketals.
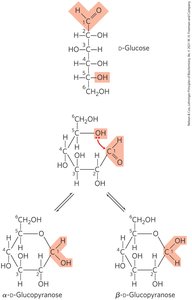
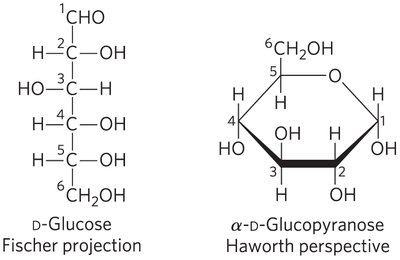
α and β Stereoisomeric Configurations
The reaction with the first alcohol molecule creates an additional chiral center (the carbonyl carbon), producing either α or β configurations. Anomers are isomeric forms differing only in their configuration about the hemiacetal or hemiketal carbon atom (anomeric carbon).
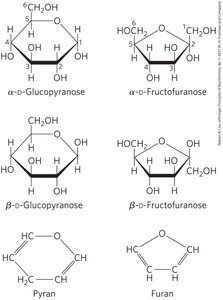
Pyranoses and Furanoses
Pyranoses are six-membered ring compounds, while furanoses are five-membered ring compounds. These rings are formed by reactions between hydroxyl and keto groups within the same molecule.
Haworth Perspective Formulas
Haworth perspective formulas provide a more accurate representation of cyclic sugar structures than Fischer projections. The six-membered ring is tilted, and bonds closest to the reader are drawn thicker.
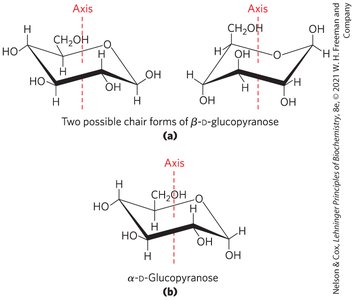
Conformational Formulas of Pyranoses
Pyranose rings tend to assume either of two "chair" conformations, which are interconvertible without breaking covalent bonds but require energy input.

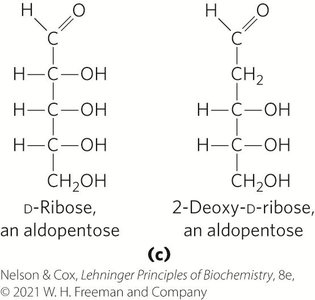
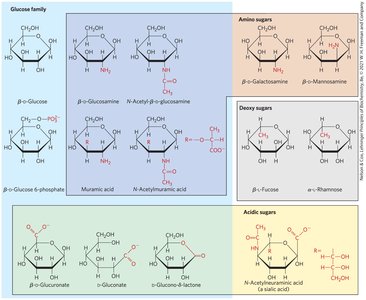
Hexose Derivatives
Organisms contain a variety of hexose derivatives, including amino sugars, deoxy sugars, and acidic sugars.
Aldonic and Uronic Acids
Aldonic acids: Formed by oxidation of the carbonyl carbon of aldoses.
Uronic acids: Formed by oxidation at C-6.
Both form stable intramolecular esters called lactones.
Phosphorylated Derivatives
Some sugar intermediates are phosphate esters (e.g., glucose 6-phosphate), which are stable at neutral pH and bear a negative charge, trapping sugar inside the cell.
Reducing Sugars
Sugars that are, or can form, aldehydes are reducing sugars. They undergo characteristic redox reactions where free aldehyde groups react with Cu2+ under alkaline conditions, forming a brick-red precipitate.
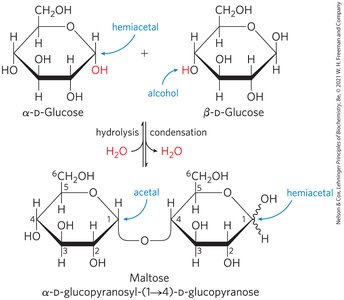
O-Glycosidic Bonds
An O-glycosidic bond is a covalent linkage joining two monosaccharides, formed when a hydroxyl group of one sugar reacts with the anomeric carbon of the other. This bond is readily hydrolyzed by acid.
The Reducing End
Formation of a glycosidic bond renders a sugar nonreducing. The reducing end is the end of a disaccharide or polysaccharide chain with a free anomeric carbon.
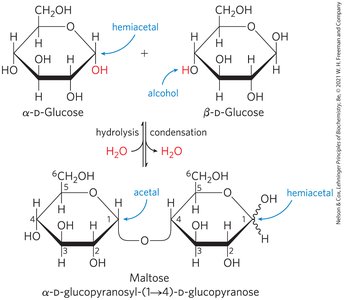
Naming Reducing Oligosaccharides
With the nonreducing end on the left, give the configuration (α or β) at the anomeric carbon joining the first unit to the second.
Name the nonreducing residue using "furano" or "pyrano".
Indicate in parentheses the two carbon atoms joined by the glycosidic bond, with an arrow connecting the two numbers.
Name the second residue and repeat for additional residues.
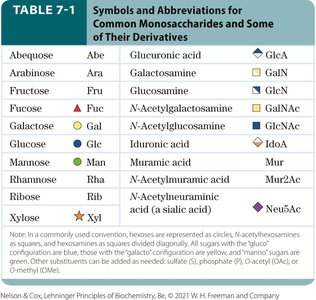
Symbols and Abbreviations for Monosaccharides
Monosaccharide | Abbreviation | Derivative |
|---|---|---|
Glucose | Glc | Glucuronic acid (GlcA) |
Galactose | Gal | Galactosamine (GalN) |
Fructose | Fru | Glucosamine (GlcN) |
Mannose | Man | N-Acetylgalactosamine (GalNAc) |
Ribose | Rib | N-Acetylglucosamine (GlcNAc) |
Xylose | Xyl | N-Acetylneuraminic acid (Neu5Ac) |
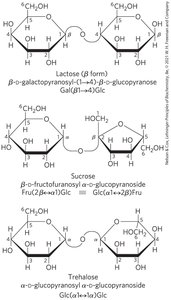
Three Common Disaccharides
Lactose: Reducing disaccharide.
Sucrose and Trehalose: Nonreducing sugars.
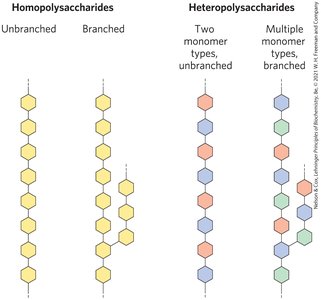
Polysaccharides
Homopolysaccharides and Heteropolysaccharides
Polysaccharides are the most abundant carbohydrates in nature and are also called glycans. Homopolysaccharides contain only a single monomeric sugar species, while heteropolysaccharides contain two or more kinds of monomers.
Storage and Structural Roles
Starch: Contains amylose (unbranched) and amylopectin (branched).
Glycogen: Polymer of (α1→4)-linked glucose subunits, with (α1→6)-linked branches; more extensively branched and compact than starch.
Cellulose: Linear, unbranched homopolysaccharide consisting of D-glucose units linked by (β1→4) glycosidic bonds; animals lack the enzyme to hydrolyze these bonds.
Chitin: Linear homopolysaccharide composed of N-acetylglucosamine residues in (β1→4) linkage; more hydrophobic and water-resistant than cellulose.
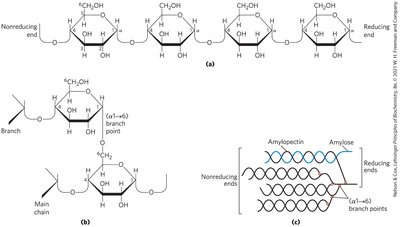

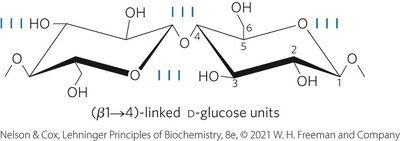
Polysaccharide Folding and Conformation
Three-dimensional structures are stabilized by weak interactions, especially hydrogen bonding due to the high number of hydroxyl groups. Steric hindrance limits free rotation about C—O bonds linking residues.
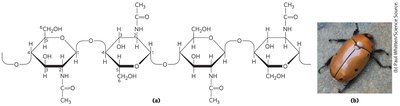
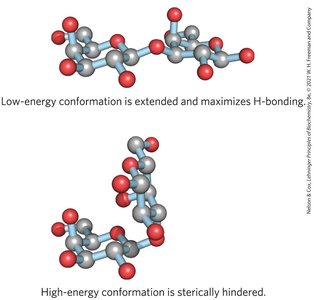
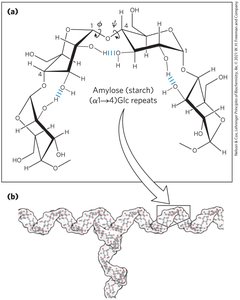
Table: Structures and Roles of Some Polysaccharides
Polymer | Type | Repeating Unit | Size | Roles/Significance |
|---|---|---|---|---|
Starch: Amylose | Homo | (α1→4)Glc, linear | 50–5,000 | Energy storage in plants |
Starch: Amylopectin | Homo | (α1→4)Glc, (α1→6) branches | Up to 106 | Energy storage in plants |
Glycogen | Homo | (α1→4)Glc, (α1→6) branches | Up to 50,000 | Energy storage in animals |
Cellulose | Homo | (β1→4)Glc | Up to 15,000 | Structural in plants |
Chitin | Homo | (β1→4)GlcNAc | Very large | Structural in exoskeletons |
Peptidoglycan | Hetero | Mur2Ac(β1→4)GlcNAc | Very large | Structural in bacteria |
Hyaluronan | Hetero | GlcA(β1→3)GlcNAc | Up to 100,000 | Structural in vertebrates |
Glycoconjugates: Proteoglycans, Glycoproteins, and Glycolipids
Glycoconjugates
Glycoconjugates are biologically active molecules consisting of an informational carbohydrate joined to a protein or lipid. They play critical roles in cell signaling, recognition, and structural integrity.
Proteoglycans
Proteoglycans are macromolecules of the cell surface or extracellular matrix consisting of one or more sulfated glycosaminoglycan chains joined covalently to a membrane or secreted protein.
Glycoproteins
Glycoproteins have one or several oligosaccharides joined covalently to a protein. They are found on the outer face of the plasma membrane, in the extracellular matrix, blood, and organelles.
Glycolipids and Glycosphingolipids
Glycolipids are plasma membrane components with hydrophilic head groups as oligosaccharides. Glycosphingolipids are a class of glycolipids with specific backbone structure, abundant in neurons and involved in signal transduction.
Carbohydrates as Informational Molecules: The Sugar Code
Lectins and Glycobiology
Lectins are proteins that bind carbohydrates with high specificity and affinity, mediating cell-cell recognition, signaling, adhesion, and intracellular targeting. Glycobiology is the study of the structure and function of glycoconjugates.
Oligosaccharide Structures
Oligosaccharides are information-dense due to their branched structures, stereochemistry, and glycosidic bond positions. This diversity allows for specific molecular recognition.
Methods of Carbohydrate Analysis
Determining Oligosaccharide and Polysaccharide Structures
Traditional chemical and enzymatic approaches
Mass spectrometry
High-resolution NMR spectroscopy
Solid-Phase Synthetic Methods
Carbohydrate chemists can synthesize short segments of almost any glycosaminoglycan using solid-phase oligosaccharide synthesis, useful for exploring lectin-oligosaccharide interactions.
Summary Table: Key Carbohydrate Terms
Term | Definition |
|---|---|
Monosaccharide | Single sugar unit |
Disaccharide | Two sugar units |
Polysaccharide | Many sugar units |
Glycosidic bond | Covalent bond joining sugars |
Reducing sugar | Sugar with free aldehyde or ketone group |
Epimer | Sugars differing at one chiral center |
Anomer | Isomers differing at the anomeric carbon |
Lectin | Carbohydrate-binding protein |
