 Back
BackCarbohydrates: Structure, Stereochemistry, and Biological Roles
Study Guide - Smart Notes

Carbohydrates: Structure and Classification
Definition and General Properties
Carbohydrates are polyhydroxy aldehydes or ketones, or compounds that yield such molecules upon hydrolysis. They are essential biomolecules that serve as energy sources, structural components, and participate in cell signaling. The general formula for many carbohydrates is Cn(H2O)n.
Monosaccharides: Simple sugars with 3–7 carbon atoms.
Disaccharides: Composed of two monosaccharide units.
Polysaccharides: Long chains of monosaccharide units, can be linear or branched.
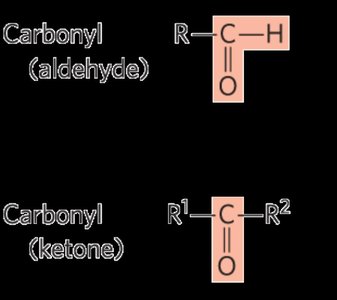
Carbohydrates are classified based on the number of carbon atoms (e.g., triose, tetrose, pentose, hexose) and the type of carbonyl group present (aldose for aldehyde, ketose for ketone).
Monosaccharide Structure and Stereochemistry
Key Functional Groups
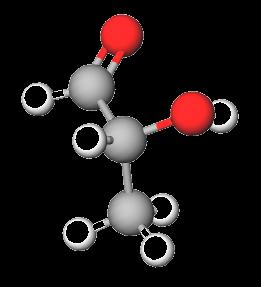
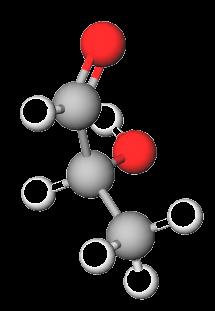
Monosaccharides contain both carbonyl (aldehyde or ketone) and multiple hydroxyl groups. The simplest monosaccharides are glyceraldehyde (an aldotriose) and dihydroxyacetone (a ketotriose).
Stereochemistry and Isomerism
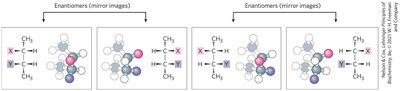
The presence of chiral centers in monosaccharides leads to multiple stereoisomers. The D- and L- notation refers to the configuration around the chiral carbon farthest from the carbonyl group, based on the reference molecule glyceraldehyde.
Enantiomers: Non-superimposable mirror images (e.g., D- and L-glyceraldehyde).
Diastereomers: Stereoisomers that are not mirror images; differ at one or more (but not all) chiral centers.
Epimers: Diastereomers that differ at only one chiral center.
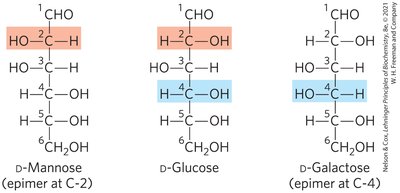
Epimers of Glucose
D-Mannose and D-galactose are epimers of D-glucose, differing at C-2 and C-4, respectively.
Physical and Chemical Properties of Carbohydrates
Reducing Properties
Aldehyde and ketone groups in carbohydrates can act as reducing agents. Aldoses are generally stronger reductants than ketoses. The reducing ability of sugars is the basis for several biochemical tests, such as Tollen's test for aldehydes.

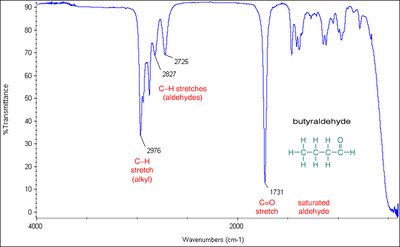
Infrared Spectroscopy
Carbonyl groups absorb strongly in the infrared region. However, the IR spectra of carbohydrates are often less distinct due to extensive hydrogen bonding and the presence of multiple functional groups.
Cyclization of Monosaccharides
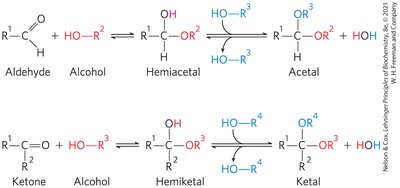
Formation of Hemiacetals and Hemiketals
Monosaccharides with five or more carbons can cyclize via intramolecular reactions between a carbonyl group and a hydroxyl group, forming a ring structure. The new chiral center formed is called the anomeric carbon.
Hemiacetal: Formed from an aldehyde and an alcohol.
Hemiketal: Formed from a ketone and an alcohol.
Alpha and Beta Anomers
The cyclization creates two possible configurations at the anomeric carbon: alpha (α) and beta (β) anomers. If the hydroxyl group on the anomeric carbon is on the opposite side of the ring from the CH2OH group, it is α; if on the same side, it is β.
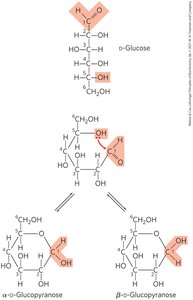
Mutarotation
When monosaccharides are dissolved in water, they interconvert between α and β forms via the open-chain structure, a process called mutarotation. This results in a change in optical rotation until equilibrium is reached.
Glycosidic Bond Formation
Disaccharides and Polysaccharides
Monosaccharides can be linked by glycosidic bonds (covalent bonds formed via condensation reactions between the anomeric carbon of one sugar and a hydroxyl group of another). The bond can be described by the carbon numbers involved and the α/β configuration (e.g., α1→4).
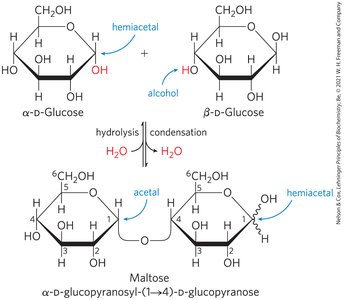
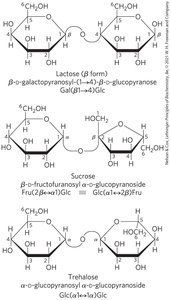
Reducing and Non-Reducing Disaccharides
Reducing disaccharides: Have a free hemiacetal/hemiketal group and can act as reducing agents (e.g., maltose).
Non-reducing disaccharides: Both anomeric carbons are involved in the glycosidic bond, so no reducing end is present (e.g., sucrose).
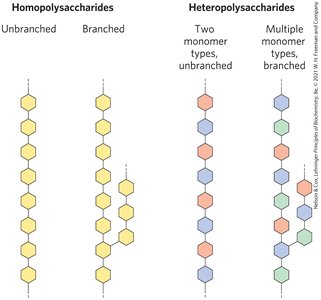
Polysaccharides: Structure and Function
Types of Polysaccharides
Homopolysaccharides: Composed of one type of monosaccharide (e.g., starch, glycogen, cellulose).
Heteropolysaccharides: Composed of two or more types of monosaccharides (e.g., agarose, peptidoglycan).
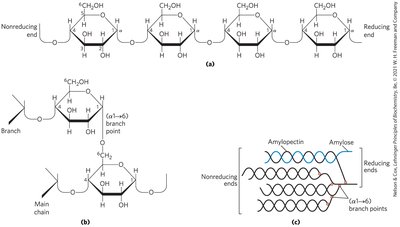
Starch and Glycogen
Both are storage polysaccharides of glucose. Starch (plants) consists of amylose (unbranched, α1→4) and amylopectin (branched, α1→4 and α1→6). Glycogen (animals) is highly branched (α1→4 and α1→6, with more frequent branches).
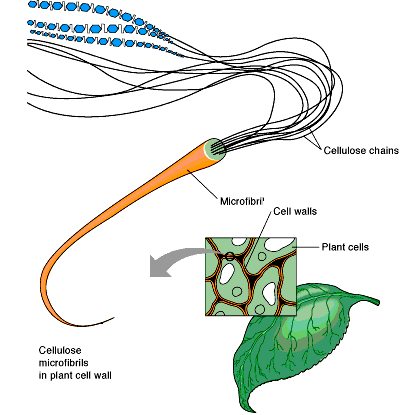
Cellulose
Cellulose is a linear homopolysaccharide of glucose with β1→4 linkages. Extensive hydrogen bonding between chains gives cellulose its rigidity and insolubility, making it a major structural component in plants.
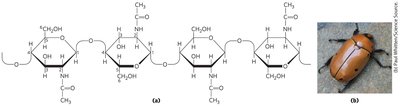
Chitin
Chitin is a linear homopolysaccharide of N-acetylglucosamine with β1→4 linkages. It forms tough, flexible structures in fungal cell walls and arthropod exoskeletons.
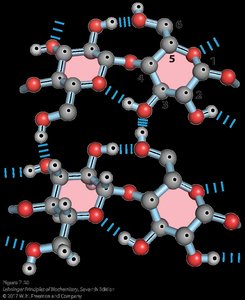
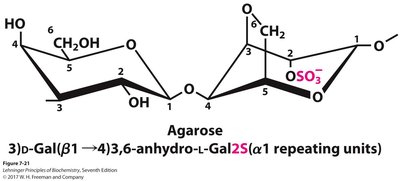
Agar and Agarose
Agar is a branched heteropolysaccharide from seaweed, used as a gel matrix in microbiology. Agarose, a component of agar, is used for DNA electrophoresis and consists of alternating β1→4 and α1→3 linked sugars.
Summary Table: Structures and Roles of Some Polysaccharides
Polymer | Type | Repeating Unit | Size | Role/Significance |
|---|---|---|---|---|
Starch (Amylose/Amylopectin) | Homo | (α1→4) Glc, (α1→6) branches | 50–106 | Energy storage in plants |
Glycogen | Homo | (α1→4) Glc, (α1→6) branches | Up to 50,000 | Energy storage in animals |
Cellulose | Homo | (β1→4) Glc | Up to 15,000 | Structural in plants |
Chitin | Homo | (β1→4) GlcNAc | Very large | Structural in exoskeletons |
Agarose | Hetero | 3)D-Gal(β1→4)3,6-anhydro-L-Gal(α1 | ~1,000 | Structural in algae, lab gels |
Monosaccharide Derivatives and Glycoconjugates
Monosaccharide Derivatives
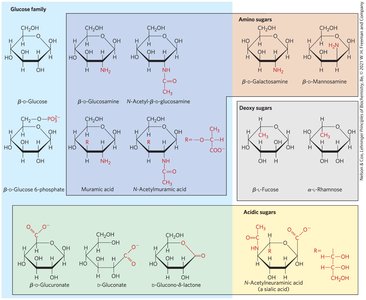
Carbohydrates can be chemically modified to form important biological molecules, such as deoxyribose (in DNA), amino sugars (e.g., glucosamine), and acidic sugars.
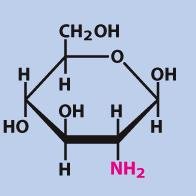
Glucosamine
Glucosamine is an amino sugar derived from glucose, commonly found in connective tissues and as a dietary supplement for joint health.

Glycoconjugates: Glycolipids and Glycoproteins
Carbohydrates can be covalently linked to lipids (glycolipids) or proteins (glycoproteins), playing crucial roles in cell recognition, signaling, and immune response.
Glycolipids: Lipids with attached oligosaccharides, important in cell membranes and blood group antigens.
Glycoproteins: Proteins with attached oligosaccharides, involved in protein-protein recognition and immune evasion by viruses.
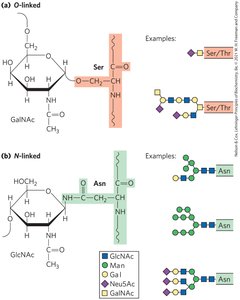
O-Linked and N-Linked Glycoproteins
O-linked oligosaccharides are attached to the hydroxyl group of serine or threonine, while N-linked oligosaccharides are attached to the amide nitrogen of asparagine.
Extracellular Matrix (ECM)
The ECM is a complex network of proteoglycans, collagen, and elastin that provides structural support, elasticity, and acts as a barrier to tumor cell invasion. Some tumor cells secrete enzymes that degrade ECM components to facilitate metastasis.
Summary
Carbohydrates are essential for energy storage, structure, and cell signaling.
Monosaccharides exhibit stereoisomerism, cyclization, and can form glycosidic bonds to create complex carbohydrates.
Polysaccharides serve diverse roles, from energy storage (starch, glycogen) to structural support (cellulose, chitin).
Carbohydrate derivatives and glycoconjugates are critical in biological recognition and extracellular structure.
