 Back
BackChapter 1: Biochemistry and the Language of Chemistry – Study Notes
Study Guide - Smart Notes

Biochemistry and the Language of Chemistry
Introduction to Biochemistry
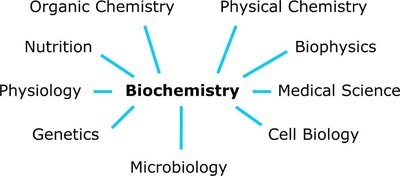
Biochemistry is the study of the chemical processes and substances that occur within living organisms. It bridges the gap between biology and chemistry, focusing on the molecular mechanisms that underlie life. The discipline is inherently interdisciplinary, drawing upon concepts and techniques from organic chemistry, physical chemistry, biophysics, genetics, cell biology, physiology, microbiology, nutrition, and medical science.
Macromolecules (e.g., proteins, nucleic acids) and metabolites (e.g., glucose, glycerol) are the two main classes of molecules studied in biochemistry.
Biochemistry investigates the structure, function, and interactions of these molecules to explain life processes.
Historical Foundations of Biochemistry
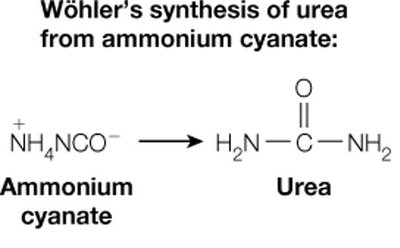
The field of biochemistry emerged from the realization that biological processes can be explained by chemical and physical principles. A pivotal moment was Friedrich Wöhler's synthesis of urea from ammonium cyanate in 1828, disproving the theory of vitalism, which posited that biological reactions required a 'life force.'
Vitalism was further challenged when Eduard and Hans Buchner demonstrated that cell extracts could ferment sugar to ethanol outside living cells (in vitro), leading to the discovery of enzymes as biological catalysts.
Enzymes are proteins whose synthesis is controlled by genes, the hereditary units encoded in DNA.
The Chemical Foundation of Life
Elements and Molecules of Living Systems
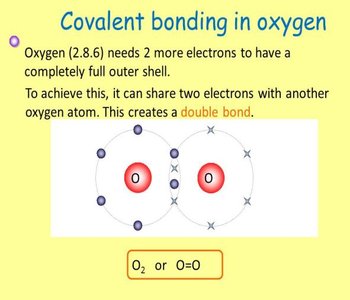
All living organisms are composed of a limited set of chemical elements, primarily carbon (C), hydrogen (H), oxygen (O), and nitrogen (N). These elements form the backbone of biological molecules due to their ability to form stable covalent bonds by sharing electrons.
Other essential elements include sulfur (S), phosphorus (P), and various ions such as Na+, K+, Mg2+, Ca2+, and Cl-.
The periodic table relevant to biochemistry highlights the abundance and roles of these elements in living systems.
Biological Macromolecules
There are four major classes of biological macromolecules essential for life:
Nucleic acids (DNA and RNA)
Proteins
Polysaccharides (complex carbohydrates)
Lipids
These macromolecules are polymers (except lipids), assembled from monomeric subunits via dehydration synthesis (condensation reactions), which remove water to form covalent bonds.
Macromolecule | Monomer | Linkage |
|---|---|---|
Nucleic acids | Nucleotide | Phosphodiester |
Proteins | Amino acid | Peptide (amide) |
Polysaccharides | Monosaccharide | Glycoside (ether) |
Lipids* | Fatty acids | Ester |
*Lipids are not true polymers but exist in large complexes.
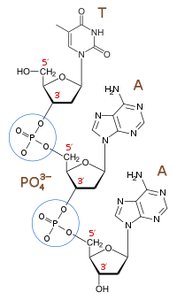
Nucleic Acids
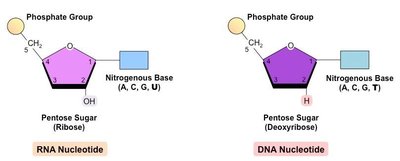
Nucleic acids are polymers of nucleotides, joined by 3',5'-phosphodiester bonds. DNA and RNA differ in their sugar (deoxyribose vs. ribose) and nitrogenous bases (DNA: A, T, C, G; RNA: A, U, C, G).
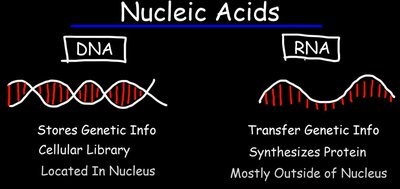
DNA stores genetic information in the nucleus.
RNA is involved in the expression and transmission of genetic information, mainly outside the nucleus.
Proteins
Proteins are polymers of 20 different amino acids, linked by peptide bonds. They exhibit remarkable structural diversity and perform a wide range of biological functions, including catalysis (enzymes), structural support, transport, signaling, and immune defense.
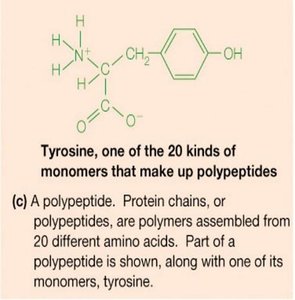
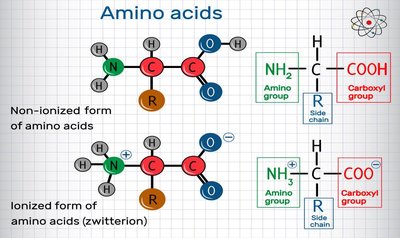
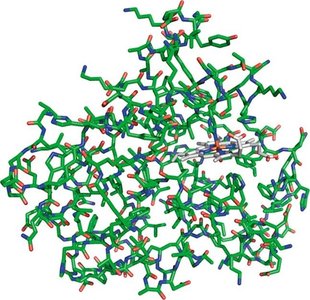
Enzymes are highly specialized proteins that catalyze specific biochemical reactions with high efficiency and specificity.
Polysaccharides
Polysaccharides are long chains of monosaccharide units. They serve as energy storage (e.g., starch in plants, glycogen in animals) and structural components (e.g., cellulose in plants).
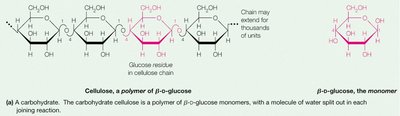
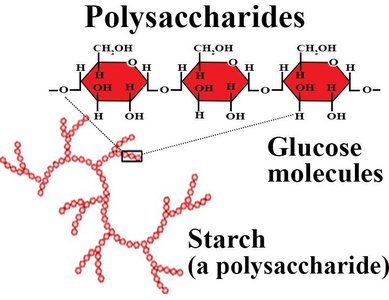
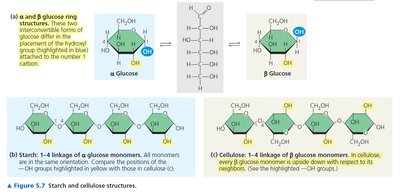
The main difference between animal and plant polysaccharides is the type of glycosidic linkage: α(1→4) in starch/glycogen (digestible by animals) vs. β(1→4) in cellulose (indigestible by most animals).
Lipids
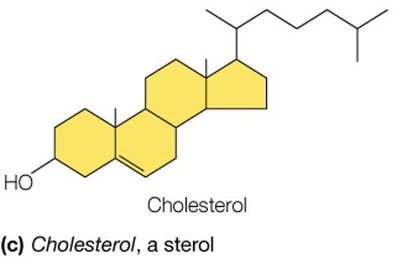
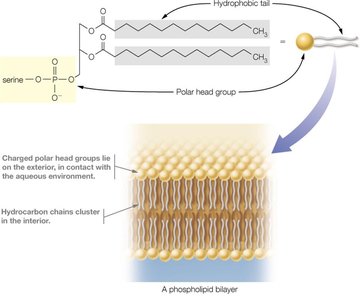
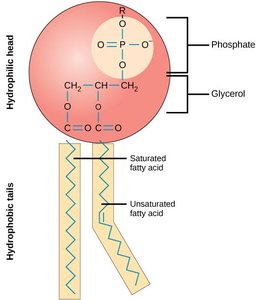
Lipids are a chemically diverse group of hydrophobic molecules, including fats (triacylglycerols), phospholipids, and sterols (e.g., cholesterol). They are essential for energy storage, membrane structure, and signaling.

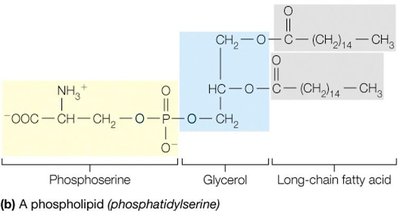
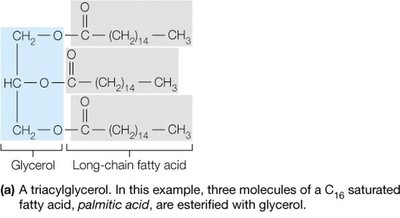
Phospholipids are amphipathic, containing both hydrophobic (fatty acid tails) and hydrophilic (phosphate head) regions, enabling them to form bilayers that make up biological membranes.
Distinguishing Characteristics of Living Systems
Seven Pillars of Life
Daniel Koshland identified seven essential attributes that distinguish living from nonliving systems:
Program: Genetic information stored in DNA.
Improvisation: Evolutionary adaptation through mutation and natural selection.
Compartmentation: Separation from the environment, allowing controlled biochemical reactions.
Energy: Maintenance of order and complexity by utilizing energy from the environment.
Regeneration: Repair and maintenance mechanisms.
Adaptability: Ability to respond to environmental changes.
Seclusion: Specificity and isolation of biochemical pathways.
The Unit of Biological Organization: The Cell
Types of Cells
Cells are the fundamental units of life. There are three major domains of life based on cell type and genetic analysis:
Bacteria (prokaryotic)
Archaea (prokaryotic)
Eukarya (eukaryotic: includes plants, animals, fungi, and protists)
Prokaryotic cells lack membrane-bound organelles, while eukaryotic cells are compartmentalized with specialized organelles.
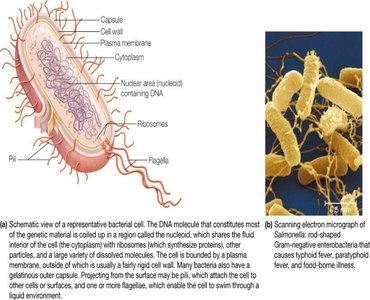
Prokaryotes: Surrounded by a plasma membrane and often a cell wall; genetic material is free in the cytosol.
Eukaryotes: Larger, with organelles such as nucleus, mitochondria, endoplasmic reticulum, and Golgi apparatus.
Biochemistry and the Information Explosion
Bioinformatics and Omics
Modern biochemistry utilizes computational tools to analyze vast amounts of molecular data. Bioinformatics applies information science to biology, including DNA sequence analysis, metabolic pathway simulation, and drug target identification.
Genomics: Study of the entire genome.
Transcriptomics: Study of gene expression (mRNA).
Proteomics: Study of the entire set of proteins.
Metabolomics: Study of metabolites.
Interactomics: Study of molecular interactions.
Summary
Biochemistry seeks to explain life at the molecular level, integrating concepts from chemistry and biology.
Living systems are composed of cells, which are classified as bacterial, archaeal, or eukaryotic.
Biochemistry is an experimental and data-driven science, increasingly reliant on computational analysis.
