 Back
BackChapter 10: Lipids – Structure, Function, and Biochemical Roles
Study Guide - Smart Notes

10.1 Storage Lipids
Fatty Acids: Structure and Properties
Fatty acids are water-insoluble hydrocarbons that serve as a primary form of cellular energy storage. Their highly reduced structure makes them a rich source of stored chemical energy. Fatty acids are typically stored as triacylglycerols, which are hydrophobic and do not require water for hydration, making storage highly efficient.
Definition: Fatty acids are carboxylic acids with hydrocarbon tails ranging from 4 to 36 carbons (C4–C36).
Classification: Can be saturated (no double bonds) or unsaturated (one or more double bonds); may be branched or unbranched.
Energy Yield: Oxidation of fatty acids to CO2 and H2O is highly exergonic.
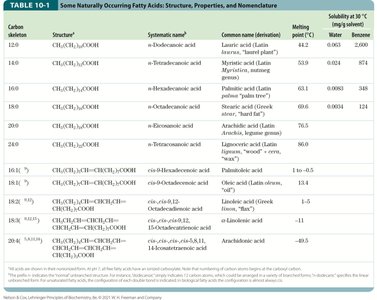
Nomenclature: Unbranched fatty acids are named by chain length and number of double bonds (e.g., 18:1(Δ9)). Numbering starts at the carboxyl carbon; double bond positions are indicated by Δ and superscript.
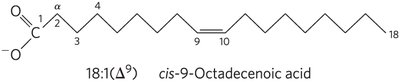
Common Patterns: Most fatty acids have even numbers of carbons (12–24). Monounsaturated fatty acids usually have a double bond at Δ9; polyunsaturated fatty acids (PUFAs) often have double bonds at Δ12 and Δ15, separated by methylene groups, and are typically in the cis configuration.
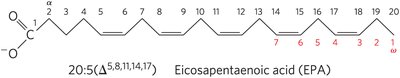
PUFAs and Human Nutrition: Omega-3 (ω-3) and omega-6 (ω-6) fatty acids are essential; humans must obtain α-linolenic acid (ALA, 18:3(Δ9,12,15)) from their diet, which is used to synthesize EPA and DHA. The optimal dietary ratio of omega-6 to omega-3 is 1:1 to 4:1.
Triacylglycerols: Structure and Function
Triacylglycerols are the simplest lipids constructed from fatty acids, consisting of three fatty acids esterified to a single glycerol molecule. They can be simple (one kind of fatty acid) or mixed (different fatty acids), and are non-polar and hydrophobic.
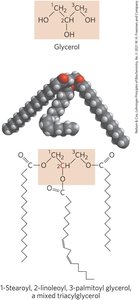
Solubility: Poor solubility in water due to the nonpolar hydrocarbon chain; longer chains and fewer double bonds decrease solubility.
Melting Points: Saturated fatty acids are waxy at room temperature; unsaturated fatty acids are oily. Packing depends on saturation.
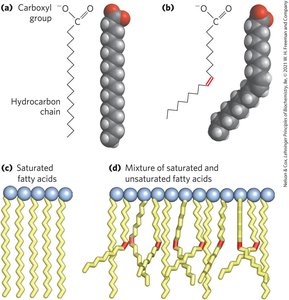
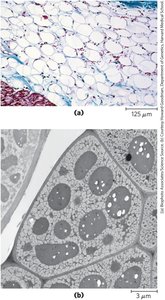
Energy Storage and Insulation: Vertebrates store triacylglycerols in adipocytes; plants store them in seeds. Triacylglycerols provide both energy and insulation, especially in polar and hibernating animals.
Lipases: Enzymes that hydrolyze stored triacylglycerols, releasing fatty acids for export and use as fuel.
Partial Hydrogenation and Trans Fats
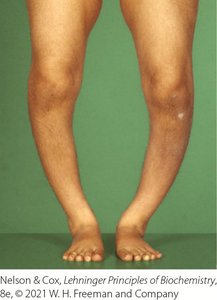
Partial hydrogenation of cooking oils improves stability and shelf life but creates trans fatty acids, which are linked to increased cardiovascular disease risk.
Trans Fats: Raise blood triacylglycerols and LDL cholesterol, lower HDL cholesterol, and increase inflammation.
Waxes: Structure and Function
Biological waxes are esters of long-chain fatty acids and alcohols, with higher melting points and water-repellent properties. They serve as energy stores and protective coatings.

10.2 Structural Lipids in Membranes
Membrane Lipid Types and Properties
Membrane lipids are amphipathic, with hydrophobic tails and polar head groups. They form biological membranes, which act as barriers to polar molecules and ions.
Phospholipids: Hydrophobic regions composed of two fatty acids joined to glycerol or sphingosine.
Glycolipids: Contain sugars at the polar ends.
Archaeal tetraether lipids: Two long alkyl chains ether-linked to glycerol.
Sterols: Four fused hydrocarbon rings.
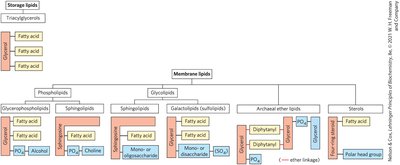
Glycerophospholipids
Glycerophospholipids are membrane lipids with two fatty acids esterified to glycerol and a polar head group attached via a phosphodiester linkage.
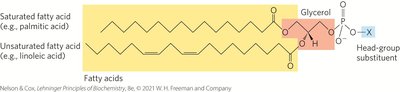
Fatty Acid Composition: Typically, C16 or C18 saturated fatty acid at C-1 and C18 or C20 unsaturated fatty acid at C-2.
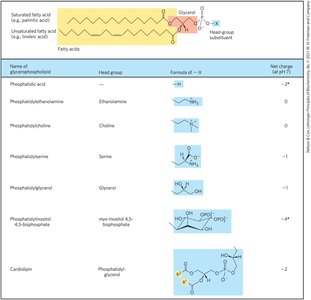
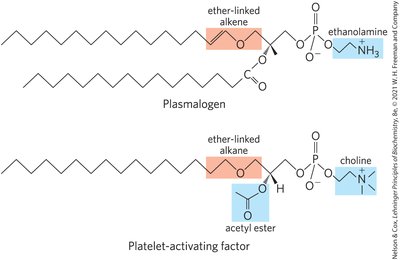
Ether Lipids: One acyl chain attached via ether linkage; plasmalogens and platelet-activating factor are examples.
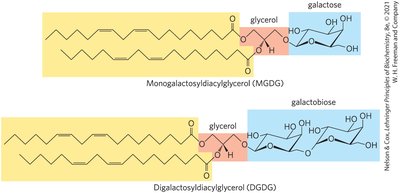
Galactolipids and Archaeal Lipids
Galactolipids predominate in plant cells, with galactose residues linked to diacylglycerol. Archaeal lipids are environmental adaptations with ether linkages.
Sphingolipids
Sphingolipids are membrane phospholipids and glycolipids with a sphingosine backbone, a fatty acid attached via amide linkage, and a polar head group.
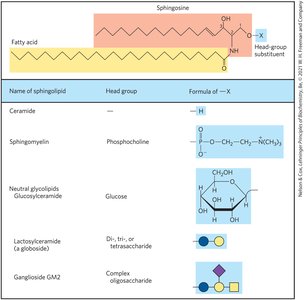
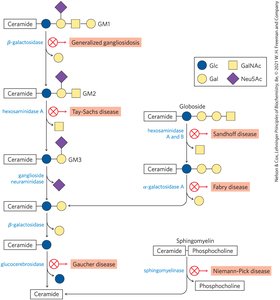
Subclasses: Sphingomyelins (phosphocholine head group), glycosphingolipids (sugar head groups), cerebrosides (single sugar), globosides (multiple sugars), gangliosides (oligosaccharides with sialic acid).
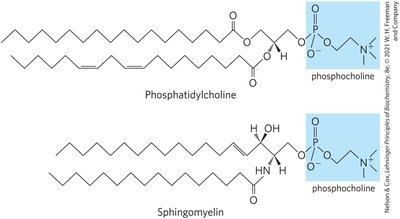
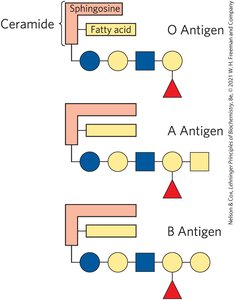
Biological Recognition: Sphingolipids are prominent in neuron membranes and determine human blood groups via their oligosaccharide head groups.
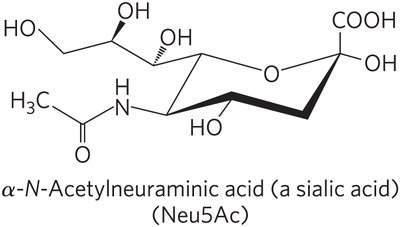
Degradation and Disease
Phospholipids and sphingolipids are degraded in lysosomes by specific enzymes. Genetic defects in these enzymes lead to accumulation of gangliosides and associated diseases.
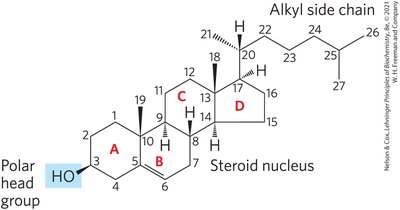
Sterols
Sterols are structural lipids with a rigid system of four fused carbon rings. Cholesterol is the major sterol in animal tissues, amphipathic, and a membrane constituent.
Biological Activities: Sterols serve as precursors for steroid hormones and bile acids, which regulate gene expression and emulsify dietary fats.
10.3 Lipids as Signals, Cofactors, and Pigments
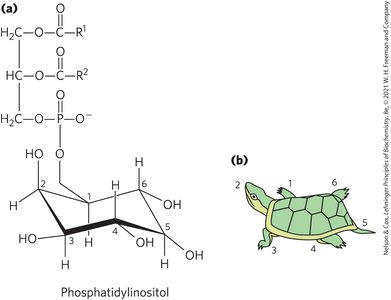
Phosphatidylinositols and Sphingosine Derivatives
Phosphatidylinositol (PI) and its derivatives regulate cell structure and metabolism. PIP2 serves as a reservoir of messenger molecules, hydrolyzed to IP3 and diacylglycerol by phospholipase C.
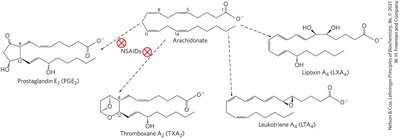
Eicosanoids
Eicosanoids are paracrine hormones derived from arachidonic acid, including prostaglandins, thromboxanes, leukotrienes, and lipoxins. They mediate inflammation, blood clotting, and other local cellular responses.
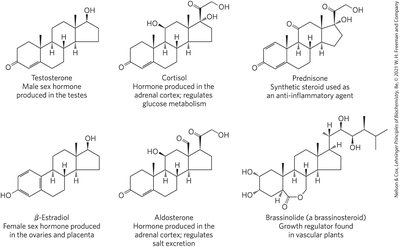
Steroid Hormones
Steroid hormones are oxidized derivatives of sterols, more polar than cholesterol, and regulate gene expression in target tissues.
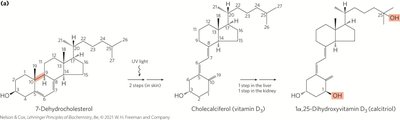
Vitamins A and D
Fat-soluble vitamins (A, D, E, K) are essential compounds that cannot be synthesized by humans. Vitamin D3 is produced in the skin and converted to calcitriol, which regulates calcium uptake. Vitamin A1 (all-trans-retinol) is involved in vision and cell differentiation.

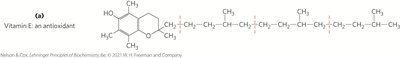
Vitamins E and K, Lipid Quinones
Vitamin E (tocopherols) are biological antioxidants, while vitamin K is essential for blood clotting. Ubiquinone and plastoquinone are lipophilic electron carriers in ATP synthesis.
10.4 Working with Lipids
Lipid Extraction and Separation
Lipids are insoluble in water and are extracted using organic solvents. Separation is based on polarity or solubility, using methods such as adsorption chromatography (HPLC, TLC) and gas chromatography (GC).
Structural Determination
Specific hydrolysis and mass spectrometry are used to determine lipid structure, including chain length and double bond positions.
Lipidomics
Lipidomics catalogs all lipids and their functions in cells. The lipidome is the full complement of lipids present in a specific cell type under particular conditions.
Category | Category code | Examples |
|---|---|---|
Fatty acids | FA | Oleate, stearoyl-CoA, palmitoylcarnitine |
Glycerolipids | GL | Di- and triaclyglycerols |
Glycerophospholipids | GP | Phosphatidylcholine, phosphatidylserine, phosphatidyethanoloamine |
Sphingolipids | SP | Sphingomyelin, ganglioside, GM2 |
Sterol lipids | ST | Cholesterol, progesterone, bile acids |
Prenol lipids | PR | Farnesol, geraniol, retinol, ubiquinone |
Saccharolipids | SL | Lipopolysaccharide |
Polyketides | PK | Tetracycline, erythromycin, aflatoxin B1 |
Additional info: The notes above expand on brief points with academic context, definitions, and examples, and include relevant images and tables to reinforce key concepts in lipid biochemistry.
