 Back
BackCh 10: Lipids – Structure, Function, and Biological Roles
Study Guide - Smart Notes

Lipids: Structure, Function, and Biological Roles
Introduction to Lipids
Lipids are a diverse group of hydrophobic or amphipathic molecules essential for energy storage, membrane structure, and signaling in biological systems. They include fatty acids, triacylglycerols, phospholipids, glycolipids, sterols, and fat-soluble vitamins.
Storage Lipids
Fatty Acids: Structure and Nomenclature
Fatty acids are carboxylic acids with long hydrocarbon chains, typically containing 4 to 36 carbon atoms. They can be saturated (no double bonds) or unsaturated (one or more double bonds), and may be branched or unbranched.
General Structure: A long hydrocarbon chain (hydrophobic) with a terminal carboxylic acid group (hydrophilic).
Nomenclature: Denoted as C:D(Δx,y,...) where C is the number of carbons, D is the number of double bonds, and Δx,y,... indicates the position of double bonds.
Omega Nomenclature: Double bonds are also described relative to the terminal methyl carbon (ω-carbon), e.g., omega-3 (ω-3) fatty acids.
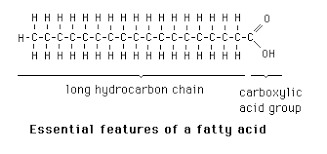
Table: Some Naturally Occurring Fatty Acids
Carbon Skeleton | Structure | Systematic Name | Common Name | Melting Point (°C) | Solubility in Water (mg/L) |
|---|---|---|---|---|---|
12:0 | CH3(CH2)10COOH | n-Dodecanoic acid | Lauric acid | 44.2 | 0.063 |
16:0 | CH3(CH2)14COOH | n-Hexadecanoic acid | Palmitic acid | 63.1 | 0.001 |
18:1(Δ9) | CH3(CH2)7CH=CH(CH2)7COOH | cis-9-Octadecenoic acid | Oleic acid | 13.4 | 0.003 |
18:2(Δ9,12) | CH3(CH2)4CH=CHCH2CH=CH(CH2)7COOH | cis,cis-9,12-Octadecadienoic acid | Linoleic acid | -5 | 0.006 |
20:4(Δ5,8,11,14) | CH3...COOH | cis,cis,cis,cis-5,8,11,14-Eicosatetraenoic acid | Arachidonic acid | -49.5 | 0.005 |
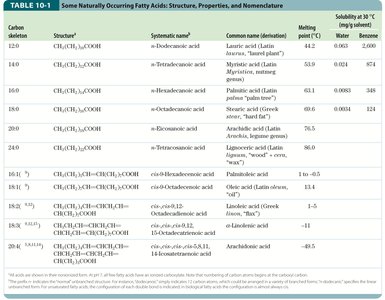
Physical Properties of Fatty Acids
Solubility: Decreases with increasing chain length and decreases with fewer double bonds.
Melting Point: Increases with chain length and decreases with unsaturation. Saturated fatty acids are waxy solids at room temperature, while unsaturated fatty acids are oily liquids.
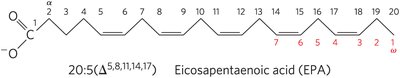
Polyunsaturated Fatty Acids (PUFAs) and Human Nutrition
PUFAs contain more than one double bond. Omega-3 and omega-6 fatty acids are essential in the human diet, as they cannot be synthesized de novo.
Omega-3 (ω-3) Fatty Acids: First double bond between C-3 and C-4 from the ω-carbon.
Omega-6 (ω-6) Fatty Acids: First double bond between C-6 and C-7 from the ω-carbon.
Essential Fatty Acids: α-Linolenic acid (ALA), eicosapentaenoic acid (EPA), and docosahexaenoic acid (DHA).
Triacylglycerols (Triglycerides)
Triacylglycerols are the main storage form of fatty acids, consisting of three fatty acids esterified to a glycerol backbone. They are nonpolar and hydrophobic, serving as energy reserves and insulation in animals and plants.
Simple Triacylglycerols: All three fatty acids are the same.
Mixed Triacylglycerols: Two or three different fatty acids.
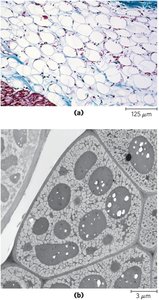
Biological Role: Stored in adipocytes (animals) and seeds (plants).
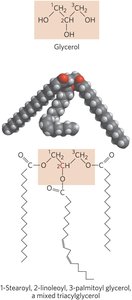
Waxes
Waxes are esters of long-chain fatty acids with long-chain alcohols. They serve as energy stores and water repellents in nature, with higher melting points than triacylglycerols.
Examples: Beeswax, plant cuticles, and animal fur coatings.

Structural Lipids in Membranes
Biological Membranes and Amphipathic Lipids
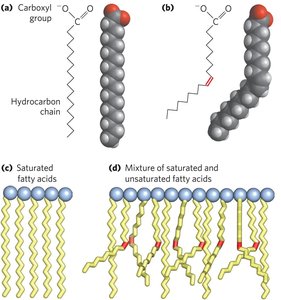
Biological membranes are composed of a double layer of amphipathic lipids, which have both hydrophobic and hydrophilic regions. The hydrophobic regions interact with each other, while the hydrophilic regions interact with water.
Major Classes: Phospholipids, glycolipids, and sterols.
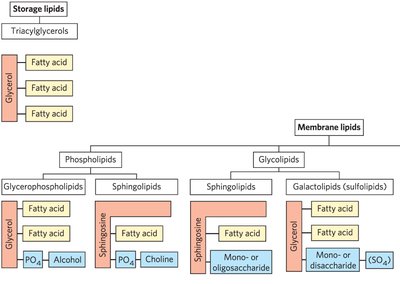
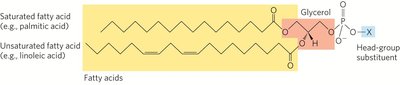
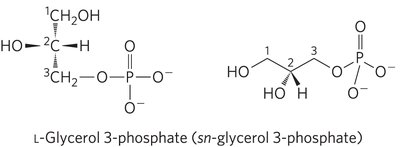
Glycerophospholipids
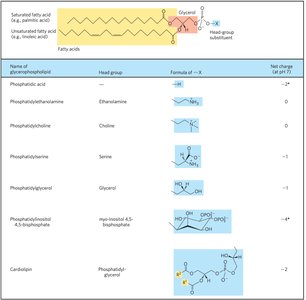
Glycerophospholipids are the main structural lipids in membranes, consisting of two fatty acids esterified to glycerol and a phosphate group with a polar head group.
Phosphatidic Acid: The simplest glycerophospholipid, with a phosphate group as the head group.
Head Groups: Can be serine, ethanolamine, choline, inositol, or glycerol.
Fatty Acid Composition: Typically, C-1 is a saturated fatty acid, and C-2 is an unsaturated fatty acid.
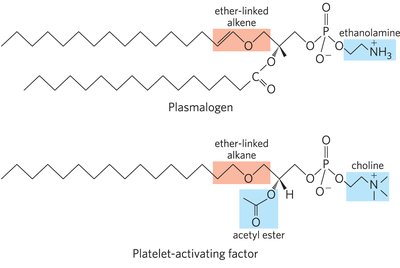
Ether Lipids and Platelet-Activating Factor
Some glycerophospholipids contain ether-linked fatty acids. Plasmalogens and platelet-activating factor are biologically important ether lipids.
Plasmalogens: Contain an ether-linked alkene at C-1 of glycerol.
Platelet-Activating Factor: An ether lipid involved in inflammation and allergic responses.
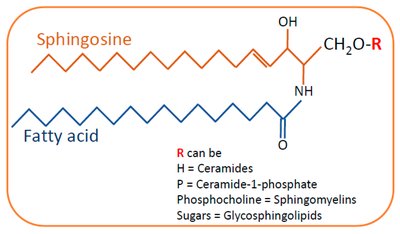
Sphingolipids
Sphingolipids are membrane lipids derived from sphingosine, a long-chain amino alcohol. They have a polar head group and two nonpolar tails but lack glycerol.
Ceramide: The parent compound, formed by attaching a fatty acid to the amino group of sphingosine.
Sphingomyelins: Contain phosphocholine or phosphoethanolamine as the head group.
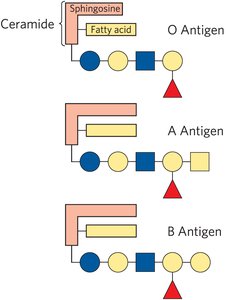
Glycosphingolipids: Have one or more sugars as the head group; important in cell recognition and signaling.
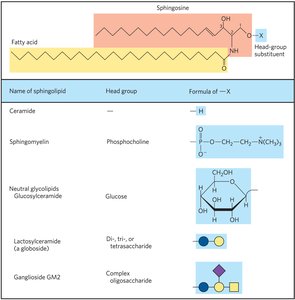
Gangliosides and Biological Recognition
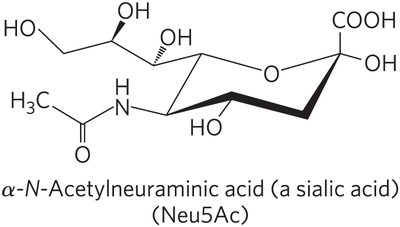
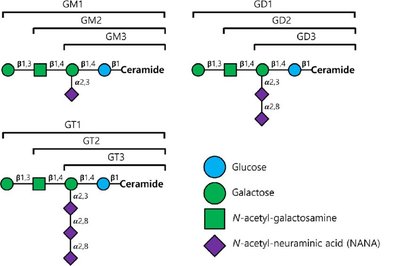
Gangliosides are glycosphingolipids with complex oligosaccharide head groups containing sialic acid residues. They are abundant in neuronal membranes and play roles in cell recognition, including blood group antigens.
Lipid Degradation
Phospholipids and sphingolipids are degraded in lysosomes by specific enzymes. Defects in these enzymes can lead to lysosomal storage diseases.
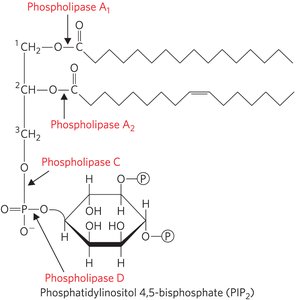
Phospholipases: Remove fatty acids from phospholipids.
Lysosomal Enzymes: Remove sugar units from gangliosides.
Sterols
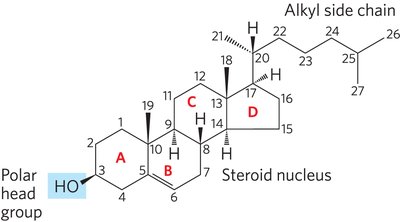
Sterols are structural lipids with a characteristic four-fused-ring system. Cholesterol is the major sterol in animal tissues, serving as a membrane component and precursor for steroid hormones and bile acids.
Cholesterol: Amphipathic molecule with a polar head group and nonpolar body.
Bile Acids: Derived from cholesterol, aid in fat digestion.
Lipids as Signals, Cofactors, and Pigments
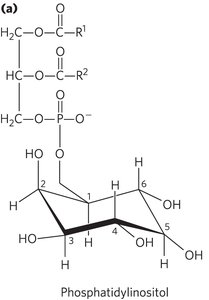
Phosphatidylinositols and Sphingosine Derivatives as Intracellular Signals
Phosphatidylinositol and its phosphorylated derivatives (e.g., PIP2, PIP3) regulate cell structure and metabolism by serving as reservoirs and points of nucleation for signaling proteins. Sphingolipid derivatives such as ceramide regulate cell division, differentiation, and apoptosis.
Eicosanoids: Local Hormones Derived from Arachidonic Acid
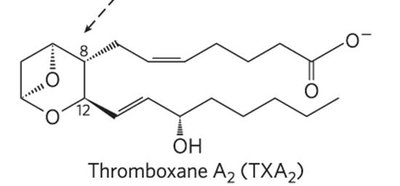
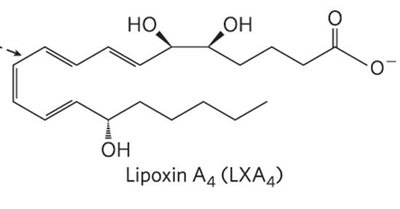
Eicosanoids are paracrine hormones derived from arachidonic acid. They include prostaglandins, thromboxanes, leukotrienes, and lipoxins, each with distinct biological functions.
Prostaglandins: Five-membered ring; involved in inflammation, pain, and smooth muscle contraction.
Thromboxanes: Six-membered ring; involved in blood clot formation.
Leukotrienes: Linear with conjugated double bonds; mediate allergic and inflammatory responses.
Lipoxins: Linear with hydroxyl groups; anti-inflammatory agents.
Steroid Hormones
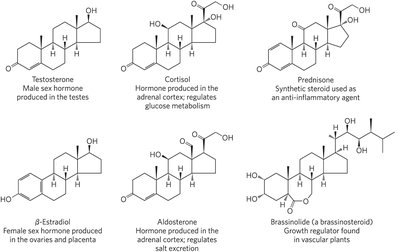
Steroid hormones are oxidized derivatives of sterols, more polar than cholesterol, and function as signaling molecules that regulate gene expression in target tissues.
Examples: Testosterone, estradiol, cortisol, aldosterone, prednisone, brassinolide.
Fat-Soluble Vitamins
Vitamins A, D, E, and K are lipid-soluble and essential for various biological functions.
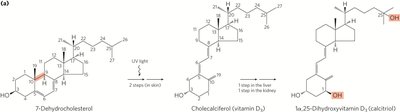
Vitamin D: Synthesized from 7-dehydrocholesterol in the skin; regulates calcium metabolism.
Vitamin A: Derived from β-carotene; involved in vision and gene regulation.
Vitamin E (Tocopherols): Antioxidants that protect membrane lipids from oxidation.
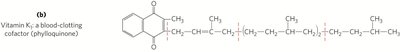
Vitamin K: Essential for blood clotting; undergoes oxidation-reduction cycles.
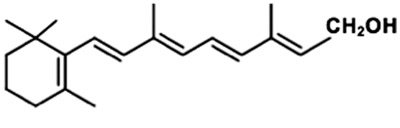
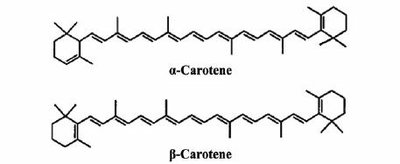
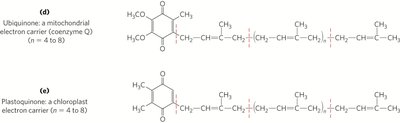
Lipid Quinones and Dolichols
Ubiquinone (coenzyme Q) and plastoquinone are isoprenoid quinones that function as electron carriers in mitochondrial and chloroplast electron transport chains. Dolichols anchor and activate sugars for the biosynthesis of complex carbohydrates, glycolipids, and glycoproteins.
Lipidic Pigments
Many natural pigments are lipids with conjugated double bonds (dienes), allowing them to absorb visible light. Examples include carotenoids, which are precursors to vitamin A.
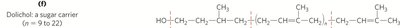
Additional info: This guide covers the structure, classification, and biological roles of lipids, integrating textbook-level explanations and relevant visual aids for biochemistry students.
