 Back
BackEnergetics of Biological Systems: Thermodynamics and Bioenergetics
Study Guide - Smart Notes

The Energetics of Biological Systems
Thermodynamic Systems and Living Organisms
Biological systems are governed by the principles of thermodynamics, which describe how energy is transferred and transformed. Understanding these principles is essential for biochemistry, as they explain how cells maintain order and drive metabolic processes.
System: Any defined part of the universe under study (e.g., a cell, a Petri dish).
Types of Systems:
Open systems: Exchange both matter and energy with surroundings (e.g., living cells).
Closed systems: Exchange energy but not matter.
Isolated systems: Exchange neither matter nor energy.
Surroundings: Everything outside the system's boundaries.
The First Law of Thermodynamics
The first law states that energy cannot be created or destroyed, only transformed. In biological systems, this principle governs the conservation of energy during metabolic reactions.
Energy transfer: Energy can be transferred as heat or work.
State function: Properties like enthalpy (H), free energy (G), and entropy (S) depend only on the initial and final states, not the pathway.
Enthalpy change:
The Second Law of Thermodynamics and Entropy
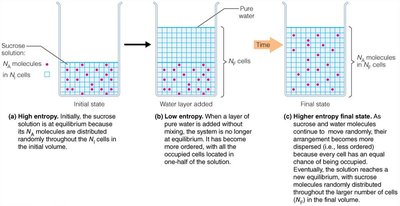
The second law states that the entropy of an isolated system tends to increase, leading to greater disorder. In living systems, processes are favorable if they increase the total entropy of the universe.
Entropy (S): A measure of disorder or randomness.
Favorable process:
Energy dissipation: Some energy is always lost as unusable energy (entropy).
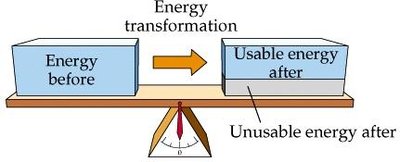
Free Energy and Biological Work
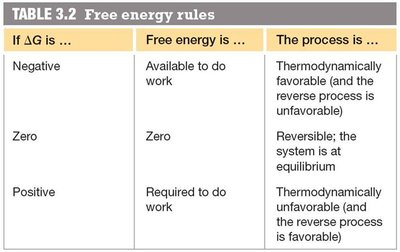
Free energy (Gibbs free energy, G) determines whether a process is thermodynamically favorable. The change in free energy () predicts the direction of chemical reactions and their ability to do work.
Free energy change:
Interpretation:
: Process is thermodynamically favorable (exergonic).
: System is at equilibrium.
: Process is thermodynamically unfavorable (endergonic).
If is ... | Free energy is ... | The process is ... |
|---|---|---|
Negative | Available to do work | Thermodynamically favorable (reverse is unfavorable) |
Zero | Zero | Reversible; system at equilibrium |
Positive | Required to do work | Thermodynamically unfavorable (reverse is favorable) |
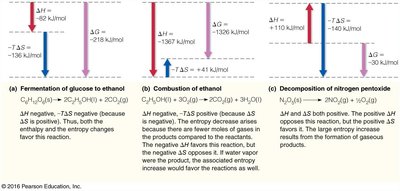
Contributions of Enthalpy and Entropy to Exergonic Processes
Both enthalpy and entropy contribute to the free energy change of a reaction. Their interplay determines whether a process is favorable under given conditions.
Examples:
Fermentation of glucose: negative, negative, negative (favorable).
Combustion of ethanol: positive, negative, positive (unfavorable).
Decomposition of nitrogen pentoxide: and positive, positive (unfavorable).
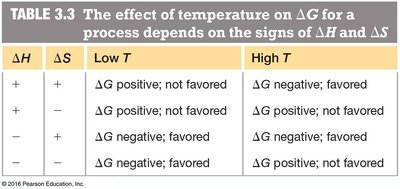
Temperature Effects on Free Energy
The effect of temperature on depends on the signs of and . This relationship is crucial for understanding biochemical reactions under physiological conditions.
Low T | High T | ||
|---|---|---|---|
+ | + | positive; not favored | negative; favored |
+ | - | positive; not favored | positive; not favored |
- | + | negative; favored | negative; favored |
- | - | negative; favored | positive; not favored |
Free Energy, Chemical Reactions, and Equilibrium
Chemical reactions reach equilibrium when the free energy change is zero. The equilibrium constant (K) and the reaction quotient (Q) are used to describe the position of equilibrium.
Equilibrium constant:
Reaction quotient:
Free energy change:
At equilibrium:
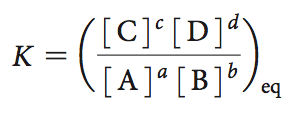
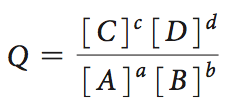
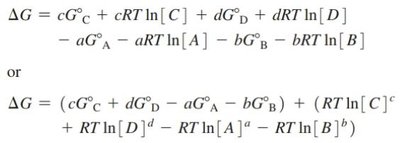
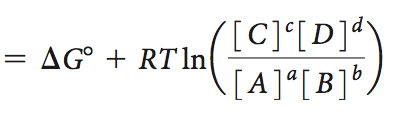
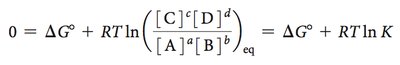
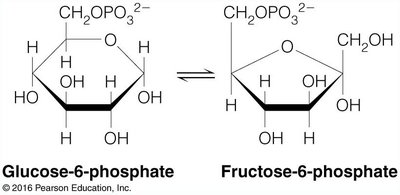
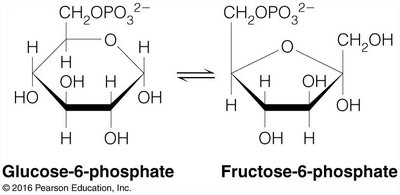
Example: Glucose-6-Phosphate and Fructose-6-Phosphate
The interconversion of glucose-6-phosphate and fructose-6-phosphate is an example of a reaction where the equilibrium lies toward the reactant due to a positive .
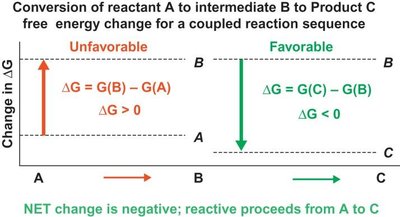
Coupling Reactions in Biological Systems
Cells drive thermodynamically unfavorable reactions by coupling them to favorable ones, ensuring the net free energy change is negative. This principle is fundamental to metabolism and cellular processes.
Coupled reactions: The sum of values determines overall favorability.
Example:
A → B ( kJ/mol)
C → D ( kJ/mol)
Overall: A + C → B + D ( kJ/mol)
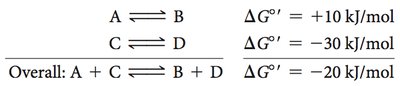
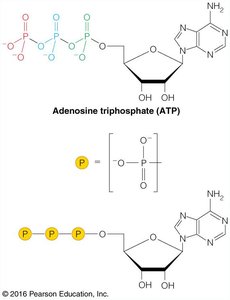
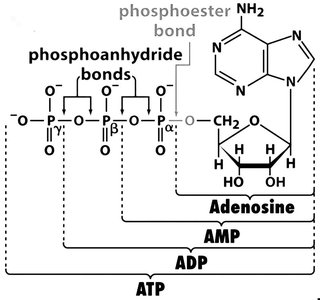
ATP: The Energy Currency of the Cell
Adenosine triphosphate (ATP) is the primary energy carrier in cells. Its hydrolysis releases energy that can be used to drive unfavorable reactions.
ATP structure: Contains three phosphate groups linked by phosphoanhydride bonds.
Hydrolysis: ATP → ADP + Pi ( kJ/mol), ADP → AMP + Pi ( kJ/mol)
Phosphoanhydride bonds: High-energy bonds whose cleavage releases significant free energy.
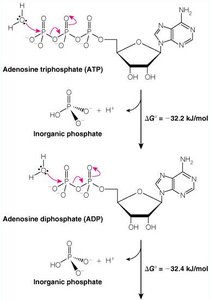
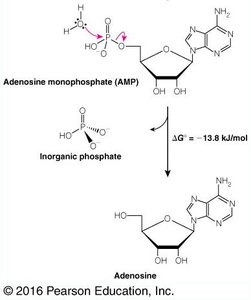
Why ADP Is More Stable Than ATP
ATP contains closely spaced negative charges, creating strain in the molecule. Hydrolysis relieves this strain, producing more stable products (ADP and Pi), which are less likely to recombine due to their charge and affinity for magnesium ions.
Electrostatic repulsion: Hydrolysis relieves repulsion between phosphate groups.
Magnesium ions: Stabilize ADP and Pi, favoring ATP hydrolysis.
Summary Table: Key Thermodynamic Concepts
Concept | Definition | Example/Application |
|---|---|---|
System | Defined part of the universe | Cell, Petri dish |
First Law | Energy conservation | Metabolic reactions |
Second Law | Entropy increases | Diffusion, mixing |
Free Energy () | Usable energy for work | ATP hydrolysis |
Coupling | Driving unfavorable reactions | ATP-driven biosynthesis |
Additional info: These notes expand on the original lecture content, providing definitions, examples, and equations for clarity and completeness. All images included are directly relevant to the explained concepts.
