 Back
BackEnzymes: Properties, Mechanisms, Classification, and Regulation
Study Guide - Smart Notes

Enzymes: Properties, Mechanisms, Classification, and Regulation
Chemical Nature and Properties of Enzymes
Enzymes are essential biological catalysts that facilitate and regulate nearly all biochemical reactions in living organisms. Their unique properties distinguish them from other catalysts and make them central to life processes.
Definition: Enzymes are mostly proteins (except ribozymes) that catalyze biochemical reactions without being consumed.
Origin: The term 'enzyme' was introduced by Kuhne in 1878 to describe biological catalysts.
Function: Enzymes can both break down large molecules and synthesize complex molecules from smaller ones.
Molecular Weight: Enzymes are macromolecules, with molecular weights ranging from thousands to millions.
Catalytic Power: Enzymes increase reaction rates by 105 to 1010 times compared to uncatalyzed reactions.
Efficiency: Enzymes are far more efficient than non-biological (man-made) catalysts.
Equilibrium: Enzymes accelerate reactions but do not alter the equilibrium constant ().
Substrate: The molecule upon which an enzyme acts is called the substrate; the result is the product.
Example: Carbonic anhydrase can hydrate thousands of CO2 molecules per second.
Medical and Biological Importance of Enzymes
Enzymes are vital for health, diagnosis, and industry.
Regulation: Enzymes regulate physiological processes; defects cause diseases.
Diagnostics: Enzyme activity in plasma is used for medical diagnosis.
Therapeutics: Enzymes are used as drugs and in biosensors (e.g., ELISA for AIDS detection).
Industry: Immobilized enzymes are used in clinical labs and pharmaceutical production.
Cleaning: Enzymes are used in detergents.
Mechanism of Enzyme Action
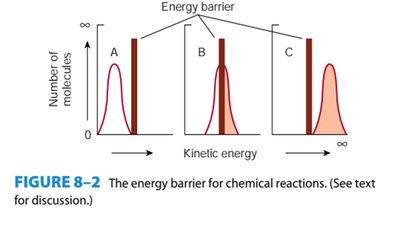
Enzymes lower the activation energy required for reactions, thereby increasing the rate of reaction. This is achieved through the formation of an enzyme-substrate (ES) complex.
Activation Energy: The energy required to convert a substrate from ground state to transition state is called activation energy.
Transition State Theory: Enzymes stabilize the transition state, reducing the activation energy.



Enzyme Specificity
Enzymes exhibit high specificity for their substrates and reactions, which is crucial for metabolic regulation.
Substrate Specificity: Enzymes act only on specific substrates (e.g., glucokinase acts on glucose, galactokinase on galactose).
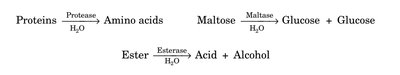
Reaction Specificity: Enzymes catalyze only specific types of reactions (e.g., lipases hydrolyze lipids, urease hydrolyzes urea).
Group Specificity: Some enzymes act on specific groups (e.g., proteases on peptide bonds, glycosidases on glycosidic bonds).
Absolute Group Specificity: Certain enzymes show high specificity for particular groups (e.g., chymotrypsin hydrolyzes peptide bonds with aromatic amino acids).



Models of Enzyme-Substrate Interaction
Two main models explain how enzymes bind substrates:
Lock and Key Model (Emil Fisher): The enzyme's active site is rigid and complementary to the substrate, like a key fitting into a lock.
Induced Fit Model (Daniel E Koshland): The enzyme's active site is flexible and changes shape upon substrate binding, enhancing specificity and catalysis.


Classification of Enzymes
Enzymes are classified by the International Union of Biochemistry (IUB) into six major classes, each with a unique EC number.
Class | Function | Examples |
|---|---|---|
EC-1: Oxidoreductases | Oxidation-reduction reactions | Dehydrogenases, Reductases, Oxidases, Peroxidases |
EC-2: Transferases | Transfer of functional groups | Hexokinase, Choline acyl transferase |
EC-3: Hydrolases | Hydrolysis of bonds | Proteases, Maltase, Esterase |
EC-4: Lyases | Cleavage of bonds without hydrolysis | Decarboxylase |
EC-5: Isomerases | Isomerization reactions | Triose phosphate isomerase, Alanine racemase |
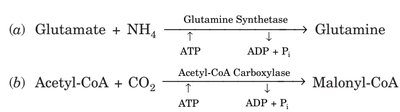
EC-6: Ligases | Joining of two molecules | Glutamine synthetase, Acetyl-CoA carboxylase |




Factors Affecting Enzyme Activity
Several factors influence the rate of enzyme-catalyzed reactions:
Temperature: Increasing temperature raises kinetic energy and collision frequency, thus increasing reaction rate up to an optimum.
Enzyme Concentration: Reaction rate is directly proportional to enzyme concentration.
pH: Each enzyme has an optimum pH; deviations affect activity due to changes in charge and conformation.
Substrate Concentration: Reaction rate increases with substrate concentration until a maximum (Vmax) is reached, following Michaelis-Menten kinetics.
Michaelis-Menten Kinetics
The relationship between substrate concentration and reaction velocity is described by the Michaelis-Menten equation:
Km (Michaelis constant): Substrate concentration at which velocity is half-maximal; indicates enzyme affinity for substrate.
Low Km: High affinity; High Km: Low affinity.
Example: Hexokinase (Km = M) phosphorylates glucose more efficiently than glucokinase (Km = M).
Lineweaver-Burk Plot
For more accurate determination of Km and Vmax, the Lineweaver-Burk (double reciprocal) plot is used:
Y-intercept gives ; slope gives .
Used to determine type of inhibition and inhibition constant (Ki).
Enzyme Regulation and Inhibition
Enzyme activity is regulated by inhibitors and feedback mechanisms.
Competitive Inhibition: Inhibitor resembles substrate and competes for active site. Increases Km, does not affect Vmax. Reversible by increasing substrate concentration.
Non-Competitive Inhibition: Inhibitor binds elsewhere on enzyme, not competing with substrate. Decreases Vmax, Km remains unchanged. Often irreversible (enzyme poisons).
Feedback Inhibition: End product of a pathway inhibits an enzyme earlier in the pathway, regulating biosynthesis.
Examples:
Competitive: Malonate inhibits succinate dehydrogenase; methotrexate inhibits dihydrofolate reductase.
Non-Competitive: Iodoacetate inhibits glyceraldehyde-3-phosphate dehydrogenase; fluoride inhibits enolase; heavy metals inhibit enzymes with –SH groups.
Feedback: CTP inhibits aspartate transcarbamoylase; cholesterol inhibits HMG-CoA reductase; heme inhibits ALA-synthase; tryptophan inhibits anthranilate synthetase.
Summary Table: Types of Enzyme Inhibition
Type | Binding Site | Effect on Km | Effect on Vmax | Reversibility |
|---|---|---|---|---|
Competitive | Active site | Increases | Unchanged | Reversible |
Non-Competitive | Other site | Unchanged | Decreases | Often Irreversible |
Medical Applications of Enzyme Inhibition
Enzyme inhibitors are used as drugs (e.g., AZT for AIDS, lovastatin for atherosclerosis, captopril for hypertension).
Some poisons act by inhibiting essential enzymes.
Conclusion
Enzymes are central to biochemistry, with their properties, mechanisms, and regulation underpinning all physiological and metabolic processes. Understanding enzyme kinetics and inhibition is crucial for medical, industrial, and research applications.
