 Back
BackGlucose Metabolism: Glycolysis, Fermentation, Gluconeogenesis, and the Pentose Phosphate Pathway
Study Guide - Smart Notes

Glucose Metabolism Overview
Introduction to Glucose Metabolism
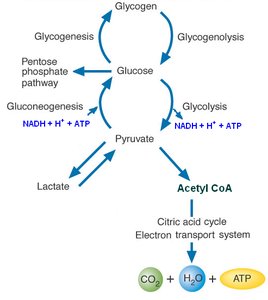
Glucose metabolism is central to cellular energy production and biosynthetic processes. It involves a series of interconnected pathways that convert glucose into energy, reducing equivalents, and metabolic intermediates for other biosynthetic routes.
Glycolysis converts glucose to pyruvate, generating ATP and NADH.
Pyruvate can be further metabolized aerobically to acetyl-CoA or anaerobically to lactate or ethanol.
Gluconeogenesis synthesizes glucose from non-carbohydrate precursors.
Pentose Phosphate Pathway (PPP) generates NADPH and ribose 5-phosphate for biosynthesis.
Historical Perspective: Otto Warburg and Metabolic Studies
Warburg's Contributions
Otto Warburg was a pioneer in the study of cellular metabolism. He developed techniques for studying metabolic reactions in tissue slices and discovered the catalytic role of iron porphyrins (heme groups) in biological oxidation, earning a Nobel Prize in 1931. Warburg's work laid the foundation for understanding the metabolic differences in cancer cells, known as the Warburg effect.
Glycolysis
Pathway Overview
Glycolysis is a ten-step pathway that occurs in the cytosol, converting one molecule of glucose into two molecules of pyruvate. This process generates a net gain of ATP and NADH, providing energy and reducing power for the cell.
Location: Cytosol
Net Reaction:
Key Steps: Energy investment (uses ATP), cleavage, energy payoff (produces ATP and NADH)
Detailed Steps of Glycolysis
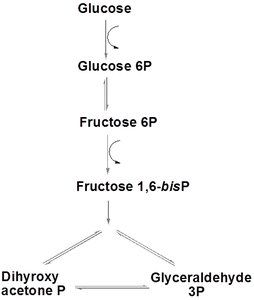
Glucose phosphorylation: Glucose is phosphorylated to glucose-6-phosphate by hexokinase, using ATP.
Isomerization: Glucose-6-phosphate is converted to fructose-6-phosphate by phosphoglucoisomerase.
Second phosphorylation: Fructose-6-phosphate is phosphorylated to fructose-1,6-bisphosphate by phosphofructokinase-1 (PFK-1), using ATP.
Cleavage: Aldolase splits fructose-1,6-bisphosphate into dihydroxyacetone phosphate (DHAP) and glyceraldehyde-3-phosphate (G3P).
Isomerization: Triose phosphate isomerase interconverts DHAP and G3P.
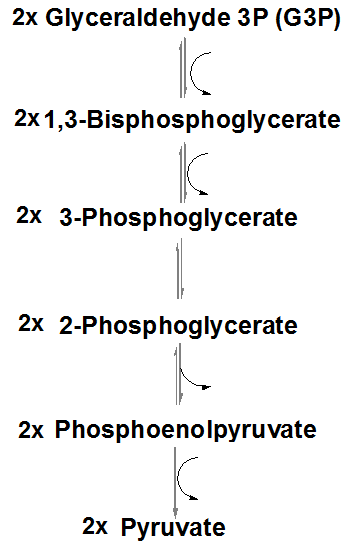
Oxidation and phosphorylation: G3P is oxidized and phosphorylated to 1,3-bisphosphoglycerate, generating NADH.
Substrate-level phosphorylation: 1,3-bisphosphoglycerate donates a phosphate to ADP, forming ATP and 3-phosphoglycerate.
Isomerization: 3-phosphoglycerate is converted to 2-phosphoglycerate.
Dehydration: 2-phosphoglycerate is dehydrated to phosphoenolpyruvate (PEP).
Second substrate-level phosphorylation: PEP donates a phosphate to ADP, forming ATP and pyruvate.
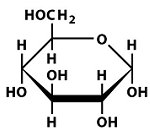
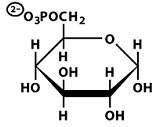
Structures of Key Intermediates
The following images show the chemical structures of glucose and its phosphorylated intermediates during glycolysis.
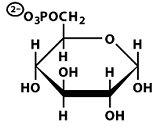
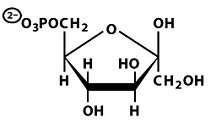
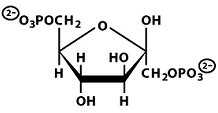
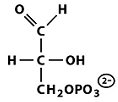
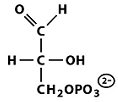
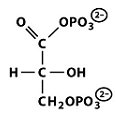
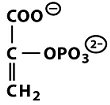
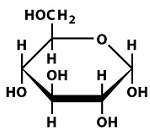
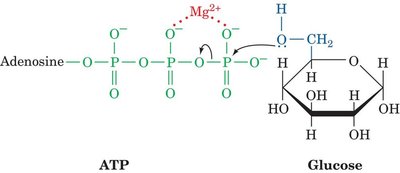
Mg2+-Mediated Phosphorylation
Hexokinase catalyzes the phosphorylation of glucose using ATP complexed with Mg2+. The Mg2+ ion shields the negative charges on ATP, making the γ-phosphorus more susceptible to nucleophilic attack by glucose.
Energy Balance of Glycolysis
The net energy yield from glycolysis per molecule of glucose is:
ATP used: 2
ATP produced: 4
Net ATP gain: 2
NADH produced: 2
Overall reaction:
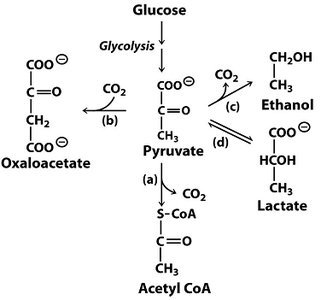
The Fate of Pyruvate
Pathways for Pyruvate Utilization
Pyruvate, the end product of glycolysis, can follow several metabolic fates depending on cellular conditions:
Aerobic conditions: Oxidized to acetyl-CoA, entering the citric acid cycle for further oxidation and ATP production.
Anaerobic conditions (muscle, RBCs): Reduced to lactate via lactate dehydrogenase (homolactic fermentation).
Anaerobic conditions (yeast): Converted to ethanol and CO2 (alcoholic fermentation).
Gluconeogenesis: Used as a substrate for glucose synthesis in the liver and kidney.
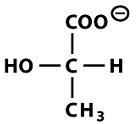
Homolactic Fermentation (Pyruvate to Lactate)
In the absence of oxygen, pyruvate is reduced to lactate by lactate dehydrogenase, regenerating NAD+ for glycolysis to continue.
Alcoholic Fermentation (Pyruvate to Ethanol)
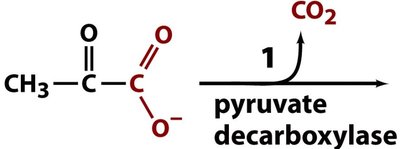
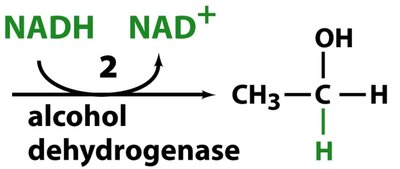
In yeast and some bacteria, pyruvate is converted to ethanol and CO2 in two steps:
Pyruvate decarboxylase converts pyruvate to acetaldehyde and CO2 (requires thiamine pyrophosphate, TPP).
Alcohol dehydrogenase reduces acetaldehyde to ethanol, regenerating NAD+.
Gluconeogenesis
Overview and Importance
Gluconeogenesis is the synthesis of glucose from non-carbohydrate precursors, primarily in the liver and kidney. It is essential during fasting or intense exercise when glucose reserves are depleted.
Major precursors: Lactate, most amino acids (especially alanine), and glycerol.
Net reaction:
Gluconeogenesis is not a simple reversal of glycolysis; it uses unique enzymes to bypass the irreversible steps of glycolysis.
Key Differences: Gluconeogenesis vs. Glycolysis
Different enzymes catalyze the irreversible steps in each pathway.
Gluconeogenesis requires more energy input (ATP and GTP).
Phosphatases (e.g., glucose-6-phosphatase, fructose-1,6-bisphosphatase) replace kinases in glycolysis for dephosphorylation steps.
Regulation of Glycolysis and Gluconeogenesis
Key Regulatory Points
Regulation ensures that glycolysis and gluconeogenesis do not occur simultaneously in the same cell, preventing a futile cycle.
Hexokinase, phosphofructokinase-1 (PFK-1), and pyruvate kinase catalyze irreversible steps in glycolysis and are tightly regulated.
PFK-1 is the primary control point, inhibited by ATP and citrate, and activated by AMP, ADP, and fructose 2,6-bisphosphate (F2,6BP).
Hexokinase is inhibited by its product, glucose-6-phosphate.
When ATP is needed, glycolysis is activated; when ATP is abundant, glycolysis is suppressed.
The Pentose Phosphate Pathway (PPP)
Overview and Functions
The pentose phosphate pathway is an alternative route for glucose metabolism, producing NADPH and ribose 5-phosphate. NADPH is essential for reductive biosynthesis (e.g., fatty acids, steroids), and ribose 5-phosphate is required for nucleotide synthesis.
Active in tissues: Liver, adipose tissue, adrenal glands, mammary glands, and red blood cells.
Net reaction:
All enzymes of the PPP are located in the cytosol.
Summary Table: Glycolysis vs. Gluconeogenesis
Feature | Glycolysis | Gluconeogenesis |
|---|---|---|
Main function | Breakdown of glucose to pyruvate, ATP, and NADH | Synthesis of glucose from non-carbohydrate precursors |
Location | Cytosol (all cells) | Liver, kidney (cytosol and mitochondria) |
Key regulatory enzyme | Phosphofructokinase-1 (PFK-1) | Fructose-1,6-bisphosphatase |
Energy yield | Net gain of 2 ATP, 2 NADH per glucose | Consumes 4 ATP, 2 GTP, 2 NADH per glucose |
Major precursors | Glucose | Lactate, alanine, glycerol, other amino acids |
