 Back
BackGlycolysis: Pathway, Regulation, and Clinical Insights
Study Guide - Smart Notes

Glycolysis: Central Pathway in Glucose Metabolism
Introduction to Glycolysis
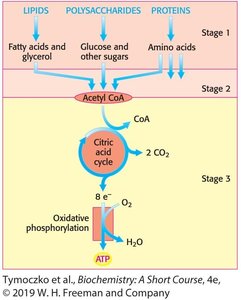
Glycolysis is a universal metabolic pathway found in nearly all prokaryotic and eukaryotic cells. It occurs in the cytosol and converts one molecule of glucose into two molecules of pyruvate. Glycolysis serves two major functions: generating ATP and providing intermediates for biosynthesis of amino acids and fatty acids.
ATP Generation: Glycolysis is a primary source of energy, especially under anaerobic conditions.
Biosynthetic Intermediates: Several glycolytic intermediates are precursors for other biomolecules.
Historical Perspective
The discovery of glycolysis involved key experiments with yeast and muscle extracts, leading to the identification of enzymes, cofactors, and intermediates. The pathway was fully elucidated by the early 1940s.
Fermentation: Pasteur showed that living cells (yeast, bacteria) are responsible for fermentation.
Enzyme Isolation: Harden, Young, and Myerhof contributed to the identification of glycolytic enzymes and intermediates.
ATP Discovery: ATP was identified as the main energy transfer molecule in 1941.
Overview of Glycolysis and Glucose Metabolism
Digestion and Uptake of Carbohydrates
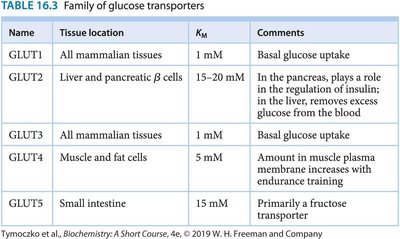
Dietary carbohydrates are broken down into monosaccharides, primarily glucose, which is then transported into cells via specific glucose transporters.
Enzymatic Breakdown: Amylases, lactase, sucrase, and maltase degrade polysaccharides and disaccharides.
Transporters: GLUT family proteins mediate glucose uptake in different tissues.
Name | Tissue location | KM | Comments |
|---|---|---|---|
GLUT1 | All mammalian tissues | 1 mM | Basal glucose uptake |
GLUT2 | Liver and pancreatic β cells | 15–20 mM | Regulation of insulin; removes excess glucose from blood |
GLUT3 | All mammalian tissues | 1 mM | Basal glucose uptake |
GLUT4 | Muscle and fat cells | 5 mM | Amount increases with endurance training |
GLUT5 | Small intestine | 15 mM | Primarily a fructose transporter |
Glycolytic Pathway: Steps and Enzymes
Stages of Glycolysis
Glycolysis is divided into two stages: Stage 1 traps and modifies glucose; Stage 2 oxidizes 3-carbon compounds to pyruvate, generating ATP.
Stage 1: Energy investment phase; glucose is phosphorylated and split.
Stage 2: Energy payoff phase; ATP and NADH are produced.
Stepwise Reactions of Glycolysis
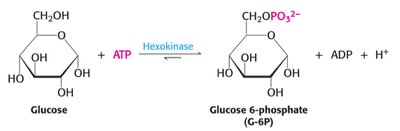
Step 1: Phosphorylation of glucose by hexokinase to form glucose-6-phosphate (G6P). This traps glucose in the cell.
Step 2: Isomerization of G6P to fructose-6-phosphate (F6P) by phosphoglucose isomerase.
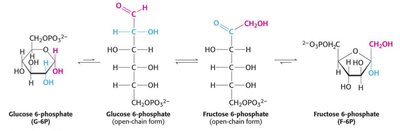
Step 3: Phosphorylation of F6P to fructose-1,6-bisphosphate (F1,6BP) by phosphofructokinase (PFK). This is a key regulatory step.

Step 4: Cleavage of F1,6BP by aldolase to yield dihydroxyacetone phosphate (DHAP) and glyceraldehyde-3-phosphate (GAP).
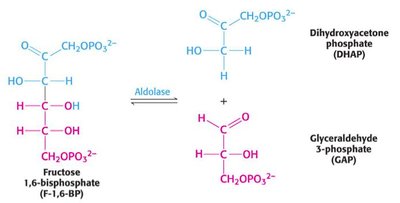
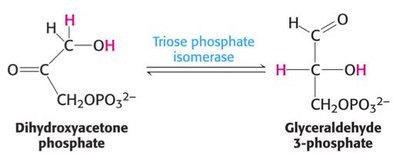
Step 5: Interconversion of DHAP and GAP by triose phosphate isomerase.
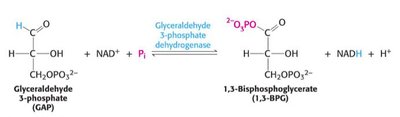
Step 6: Oxidation of GAP to 1,3-bisphosphoglycerate (1,3-BPG) by GAP dehydrogenase; NAD+ is reduced to NADH.
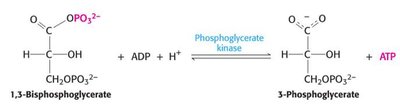
Step 7: Substrate-level phosphorylation: 1,3-BPG donates a phosphate to ADP, forming ATP and 3-phosphoglycerate (3PG).
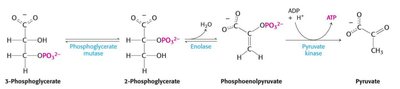
Steps 8-10: Conversion of 3PG to 2-phosphoglycerate (2PG), then to phosphoenolpyruvate (PEP), and finally to pyruvate by pyruvate kinase, producing another ATP.
Net Reaction of Glycolysis
The overall reaction for glycolysis is:
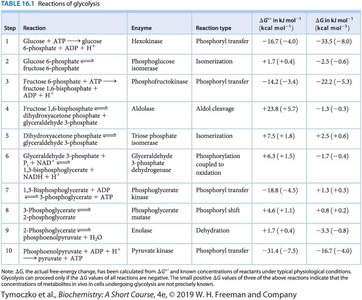
Step | Reaction | Enzyme | Reaction type | ΔG°' (kJ/mol) |
|---|---|---|---|---|
1 | Glucose + ATP → Glucose 6-phosphate + ADP + H+ | Hexokinase | Phosphoryl transfer | -16.7 |
2 | Glucose 6-phosphate → Fructose 6-phosphate | Phosphoglucose isomerase | Isomerization | +1.7 |
3 | Fructose 6-phosphate + ATP → Fructose 1,6-bisphosphate + ADP + H+ | Phosphofructokinase | Phosphoryl transfer | -14.2 |
4 | Fructose 1,6-bisphosphate → DHAP + GAP | Aldolase | Aldol cleavage | +23.8 |
5 | DHAP → GAP | Triose phosphate isomerase | Isomerization | +7.5 |
6 | GAP + Pi + NAD+ → 1,3-BPG + NADH + H+ | GAP dehydrogenase | Oxidation | +6.3 |
7 | 1,3-BPG + ADP + H+ → 3PG + ATP | Phosphoglycerate kinase | Phosphoryl transfer | -18.5 |
8 | 3PG → 2PG | Phosphoglycerate mutase | Phosphoryl shift | +4.4 |
9 | 2PG → PEP + H2O | Enolase | Dehydration | +7.5 |
10 | PEP + ADP + H+ → Pyruvate + ATP | Pyruvate kinase | Phosphoryl transfer | -31.4 |
Fates of Pyruvate
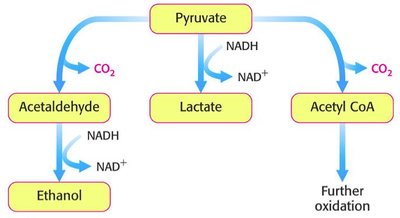
Metabolic Pathways of Pyruvate
Pyruvate, the end product of glycolysis, can follow different metabolic fates depending on cellular conditions and organism type.
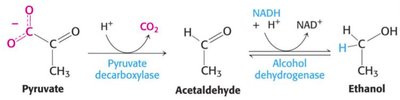
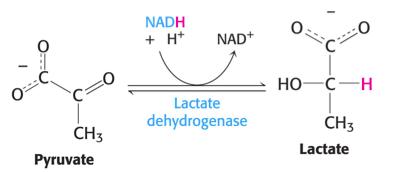
Conversion to Ethanol: In yeast and some microorganisms, pyruvate is converted to ethanol, regenerating NAD+.
Conversion to Lactate: In muscle and certain bacteria, pyruvate is reduced to lactate, also regenerating NAD+.
Conversion to Acetyl CoA: Under aerobic conditions, pyruvate is converted to acetyl CoA, entering the citric acid cycle for further oxidation.
Entry of Other Sugars into Glycolysis
Fructose and Galactose Metabolism
Fructose and galactose, common dietary sugars, are converted into glycolytic intermediates through distinct pathways.
Galactose: Converted to glucose-6-phosphate via phosphorylation, transfer to UDP, and epimerization.
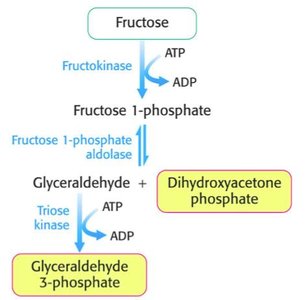
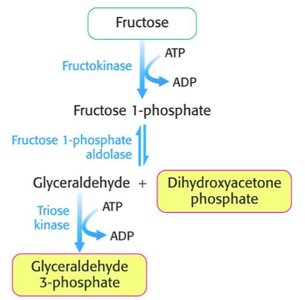
Fructose: In the liver, fructose is phosphorylated to fructose-1-phosphate, split into DHAP and glyceraldehyde, and then glyceraldehyde is phosphorylated to GAP.
Clinical Insight: Excessive Fructose Consumption
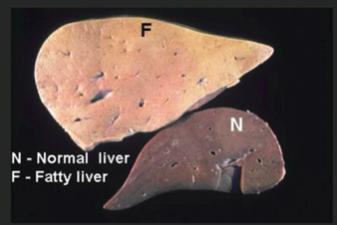
High fructose intake can lead to obesity, fatty liver, and type 2 diabetes due to bypassing key regulatory steps in glycolysis and promoting fat synthesis.
Regulation of Glycolysis
Key Regulatory Enzymes
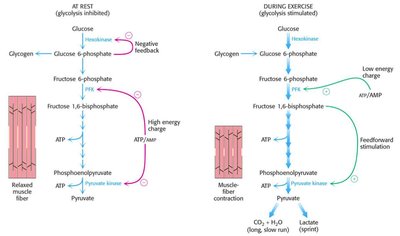
Glycolysis is tightly regulated at three irreversible steps catalyzed by hexokinase, phosphofructokinase, and pyruvate kinase. Regulation varies by tissue type.
Hexokinase: First step, not the committed step; glucose-6-P can enter other pathways.
Phosphofructokinase (PFK): Major control point, regulated allosterically by ATP, AMP, pH, and citrate.
Pyruvate kinase: Last step, regulated by ATP (inhibition) and fructose-1,6-bisphosphate (activation).
Regulation in Muscle vs. Liver
Muscle: Glycolysis provides ATP for contraction; regulated by energy state (ATP:AMP ratio) and pH.
Liver: Glycolysis maintains blood glucose and provides biosynthetic precursors; regulated by citrate and fructose-2,6-bisphosphate.
Glucokinase in Liver
Higher Km: Glucokinase has a higher Km for glucose, allowing liver to process excess glucose after meals.
Not inhibited by product: Glucokinase is not inhibited by glucose-6-P, facilitating efficient glucose utilization.
Summary
Glycolysis is a central metabolic pathway for energy production and biosynthesis.
It is regulated at key steps to meet cellular and organismal needs.
Clinical conditions such as diabetes and fatty liver are linked to dysregulation of glycolytic and related pathways.
