 Back
BackIntroduction to Biochemistry: Structure, Function, and Organization of Life
Study Guide - Smart Notes

Introduction to Biochemistry
Definition and Scope
Biochemistry is the study of the structures and processes—both physical and chemical—that occur in living organisms. It seeks to answer fundamental questions such as “What are we made of?” and “How do we work?” - Physical processes: No changes in chemical composition (e.g., melting or evaporating water). - Chemical processes: Changes in chemical composition (e.g., electrolysis of water). - Multidisciplinary nature: Biochemistry integrates biology, chemistry, physics, and other sciences. - Applications: Medical, industrial, agricultural, technological, nutritional, and life sciences.
Example: Physical vs. Chemical Processes
- Physical Process: Melting and evaporating water involve phase changes without altering the molecular structure. 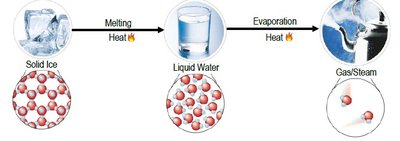 - Chemical Process: Electrolysis of water splits water molecules into hydrogen and oxygen gases, changing chemical composition.
- Chemical Process: Electrolysis of water splits water molecules into hydrogen and oxygen gases, changing chemical composition. 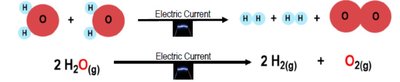
Characteristics of Living Systems
Biological Hierarchy and Cellular Organization
All organisms exhibit a hierarchy of biological organization, with cells as the fundamental unit of life. Living systems are highly ordered and composed of one or more cells. - Order: Organisms are highly organized. - Energy and Nutrient Acquisition: Dynamic metabolism. - Reproduction: Ability to produce offspring or divide. - Response to Stimuli: React to environmental changes. - Homeostasis: Maintain internal stability. - Heredity: Transmission of genetic information. - Evolution: Adaptation and change over time.
Cell Types and Organization
Prokaryotic Cells
Prokaryotes (bacteria and archaea) are unicellular organisms lacking internal membrane-bound compartments. - Key features: Nucleoid region, ribosomes, plasma membrane, cell wall, outer membrane, flagella, pili. 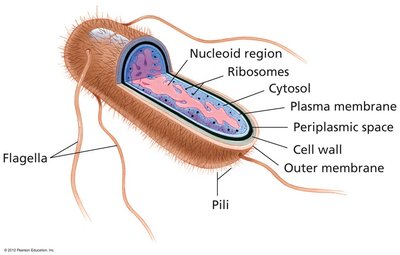
Eukaryotic Cells
Eukaryotes possess complex internal structures, including membrane-bound organelles. - Animal cells: Nucleus, cytosol, endoplasmic reticulum, mitochondrion, Golgi apparatus, vesicles, cytoskeleton, lysosome, peroxisome, plasma membrane. 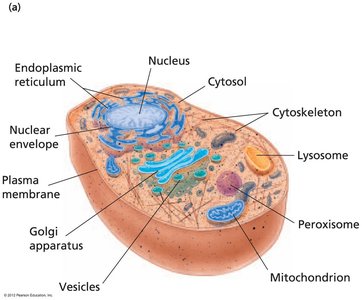 - Plant cells: Nucleus, cytosol, endoplasmic reticulum, mitochondrion, Golgi apparatus, vesicles, vacuole, cell wall, plasma membrane, peroxisome, chloroplasts.
- Plant cells: Nucleus, cytosol, endoplasmic reticulum, mitochondrion, Golgi apparatus, vesicles, vacuole, cell wall, plasma membrane, peroxisome, chloroplasts. 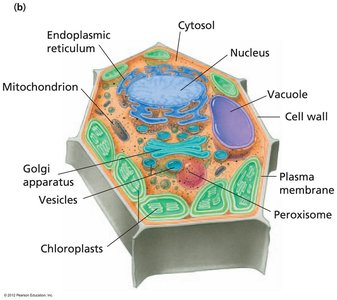
Specialization in Multicellular Organisms
Multicellular organisms consist of specialized cells that perform distinct functions, yet each cell retains the fundamental characteristics of life.
Chemical Composition of Cells
Elements of Life
- Major elements: Carbon (C), Hydrogen (H), Nitrogen (N), Oxygen (O), Phosphorus (P), and Sulfur (S) account for 97% of the mass of most organisms. - Trace elements: Additional 23 elements contribute to the remaining 3%.
Cellular Components
- Solvent and small solutes: Water (H2O), inorganic ions (Na+, K+, Ca2+, Mg2+, Cl-, etc.), dissolved gases (O2, N2, CO2, H2). - Small organic molecules: Monosaccharides, amino acids, nucleotides, lipids, cofactors (e.g., heme, NADH, FAD, SAM). - Organic macromolecules: Nucleic acids, proteins, polysaccharides, membranes.
Water in Cells
- Water content: 60–90% of cell mass is water, providing an aqueous environment for biochemical reactions.
Major Classes of Biological Molecules
Nucleic Acids
Nucleic acids (DNA and RNA) are polymers of nucleotides. - DNA: Composed of adenine (A), cytosine (C), guanine (G), thymine (T), and 2'-deoxyribose sugar, linked by phosphodiester bonds. - RNA: Contains adenine (A), cytosine (C), guanine (G), uracil (U), and ribose sugar. - Genome: The complete set of genes in an organism.
Proteins
Proteins are polymers of amino acids linked by peptide bonds. - 20 natural amino acids: Differ by their side chains (R groups). - Function: The sequence of amino acids determines protein shape and function.
Lipids
Lipids are hydrophobic molecules that form biological membranes. - Phospholipids: Composed of a head group, acyl chains (saturated and unsaturated fatty acids), glycerol, and phosphate. - Membrane structure: Lipids assemble into bilayers, forming boundaries between cellular compartments. - Other lipids: Cholesterol and glycolipids contribute to membrane diversity and function.
Carbohydrates
Carbohydrates (saccharides) are molecules with the general formula Cm(H2O)n. - Monosaccharides: Single sugar units (e.g., glucose). - Oligosaccharides: Few sugar units. - Polysaccharides: Many sugar units, can be linear or branched, homopolymers or heteropolymers. - Glycoproteins and proteoglycans: Carbohydrates covalently linked to proteins or lipids.
Cellular Compartmentalization
Prokaryotic vs. Eukaryotic Cells
- Prokaryotes: Encased in one or two membranes and a cell wall; lack internal membrane-bound compartments. - Eukaryotes: Possess specialized internal organelles (nucleus, ER, Golgi, mitochondria, chloroplasts).
Structure – The Cytoskeleton
Components and Functions
The cytoskeleton is a network of protein assemblies providing structural support and facilitating vesicle trafficking. - Microtubules, microfilaments, and intermediate filaments are constantly assembled and disassembled. - Cell division: Cytoskeleton remodeling is essential for separating cells during division.
Dynamic Cellular Processes
Metabolism and Regulation
Cells undergo multiple dynamic processes, including synthesis, trafficking, remodeling, and metabolism. - Metabolic pathways: Interconnected reactions produce, use, or degrade cellular components. - Regulation: Feedback and feed-forward loops control metabolic activity.
Interactions and Homeostasis
Feedback Mechanisms and Steady State
- Homeostasis: Maintenance of steady physical and chemical conditions through interactions between genes, chemical content, and feedback mechanisms. - Dynamic steady state: Matter and energy are constantly transformed, but overall content remains relatively constant.
Preview of Biochemistry Course Structure
Units Overview
- Unit 1: Structures and properties of biological molecules, with a focus on proteins. - Unit 2: Enzymes as biological catalysts, their function, and regulation. - Unit 3: Survey of metabolism, energy acquisition and utilization, metabolic pathways. - Unit 4: Biological information, gene encoding, DNA replication, and protein expression.
Practice Questions
Sample Questions
Which of the following is a chemical process?
a) Freezing water
b) Tearing paper
c) Rotating the bonds of a molecule
d) Mixing acids and bases
Which of the following is a physical process?
a) Digesting food in the stomach
b) Rusting of iron
c) Baking a cake
d) A protein chain folding
Summary
Biochemistry is the foundation for understanding the molecular basis of life, encompassing the structure, function, and organization of biological molecules and cells. The course will explore the complexity of living systems, the chemical composition of cells, and the dynamic processes that sustain life. Understanding molecular interactions is essential for mastering biochemistry.
