 Back
BackIsomerism and Stereochemistry: A Biochemistry Study Guide
Study Guide - Smart Notes

Isomerism in Organic and Biological Chemistry
Introduction to Isomers
Isomers are compounds that share the same molecular formula but differ in the arrangement of their atoms. This difference can be in the connectivity of atoms or their spatial orientation, leading to distinct physical and chemical properties.
Constitutional (Structural) Isomers: Compounds with the same molecular formula but different bonding arrangements.
Stereoisomers: Compounds with the same molecular and structural formulas but different spatial arrangements of atoms.
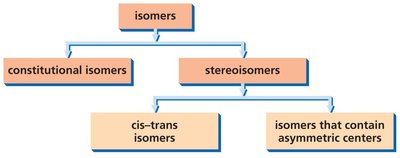
Types of Isomers
Constitutional Isomers: Differ in the way atoms are connected. Examples include ethanol and dimethyl ether, or acetone and propionaldehyde.
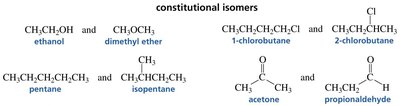
Stereoisomers: Atoms are connected in the same order but differ in spatial arrangement. Stereoisomerism includes cis–trans (geometric) isomerism and optical isomerism (enantiomers and diastereomers).
Stereochemistry and Stereoisomerism
Stereochemistry Overview
Stereochemistry is the study of the three-dimensional arrangement of atoms in molecules. Stereoisomers have the same molecular formula and sequence of bonded atoms but differ in the orientation of their atoms in space.
Geometric (cis–trans) Isomerism: Occurs due to restricted rotation around double bonds or in cyclic structures.
Optical Isomerism: Occurs when molecules have chiral centers, leading to non-superimposable mirror images (enantiomers).
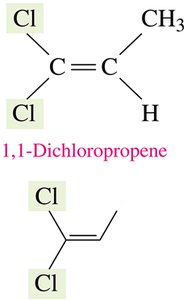
Geometric (Cis–Trans) Isomerism
Geometric isomerism arises in alkenes and cyclic compounds due to restricted rotation around double bonds or ring structures. The two main forms are cis (same side) and trans (opposite sides).
Cis Isomer: Substituents are on the same side of the double bond or ring.
Trans Isomer: Substituents are on opposite sides.
Geometric isomers have different physical and chemical properties.
Cis–trans isomerism is not possible if either carbon of the double bond has two identical groups.
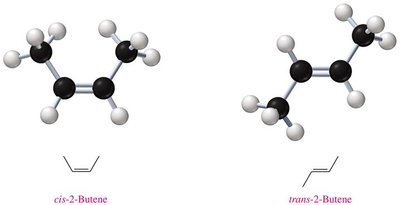
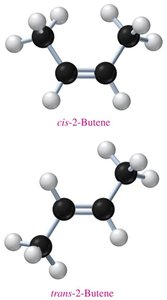
Examples and Applications
2-Butene: Exists as cis-2-butene and trans-2-butene, differing in the position of methyl groups relative to the double bond.
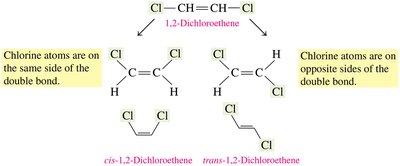
1,2-Dichloroethene: Cis isomer has both Cl atoms on the same side; trans isomer has them on opposite sides.
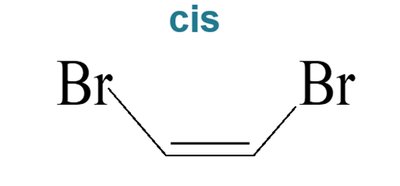
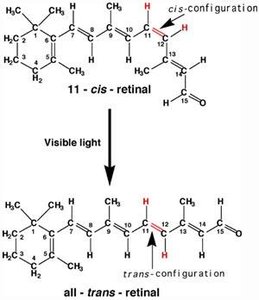
Biological Relevance: Vision
Cis–trans isomerism is crucial in biological systems. For example, the conversion of 11-cis-retinal to all-trans-retinal in the eye is essential for vision.
Optical Isomerism and Chirality
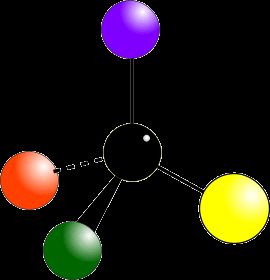
Optical isomerism arises when molecules have chiral centers—typically a carbon atom bonded to four different groups. Such molecules exist as two non-superimposable mirror images called enantiomers.
Chiral Molecule: Not superimposable on its mirror image (like left and right hands).
Achiral Molecule: Superimposable on its mirror image (has a plane of symmetry).


Properties of Enantiomers
Enantiomers have identical physical properties (melting point, boiling point, density) except for the direction in which they rotate plane-polarized light.
One enantiomer rotates light to the right (dextrorotatory, +), the other to the left (levorotatory, –).
A 50:50 mixture of enantiomers is called a racemic mixture and is optically inactive.
Chirality in Biological Systems
Many biomolecules (e.g., amino acids, sugars) are chiral, and their biological activity depends on their configuration.
Enantiomers can have drastically different effects in biological systems (e.g., L-dopa is effective in treating Parkinson’s disease, D-dopa is not).
Identifying Chiral Centers
A carbon atom is a chiral center if it is tetrahedral and bonded to four different groups. Molecules with more than one chiral center can have multiple stereoisomers, including enantiomers and diastereomers.
Van’t Hoff Rule: The maximum number of stereoisomers is , where n is the number of chiral centers.
Meso Compounds: Molecules with chiral centers but a plane of symmetry, making them achiral and optically inactive.
Summary Table: Types of Isomers
Type | Definition | Example |
|---|---|---|
Constitutional Isomers | Same molecular formula, different connectivity | Ethanol and dimethyl ether |
Stereoisomers | Same connectivity, different spatial arrangement | Cis-2-butene and trans-2-butene |
Enantiomers | Non-superimposable mirror images | Lactic acid enantiomers |
Diastereomers | Stereoisomers not related as mirror images | Threonine stereoisomers |
Meso Compounds | Chiral centers, but achiral due to symmetry | Tartaric acid meso form |
Key Equations and Rules
Van’t Hoff Rule: possible stereoisomers for n chiral centers.
Cahn–Ingold–Prelog Priority Rules: Assign priorities to groups attached to a chiral center based on atomic number to determine R/S configuration.
Example: For a chiral carbon with groups F, N, C, and H, the priority order is F > N > C > H.
Conclusion
Understanding isomerism, especially stereochemistry, is fundamental in biochemistry. The spatial arrangement of atoms in molecules determines their physical properties, chemical reactivity, and biological function. Mastery of these concepts is essential for further study in organic and biological chemistry.
