 Back
BackIsomerism and Stereochemistry: Constitutional Isomers, Cis-Trans Isomers, and Optical Isomerism
Study Guide - Smart Notes

Isomerism in Organic Chemistry
Overview of Isomers
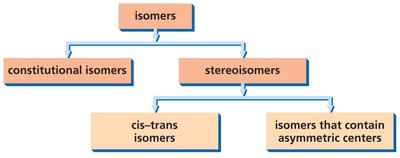
Isomers are compounds that share the same molecular formula but differ in their structural or spatial arrangements. Understanding isomerism is fundamental in biochemistry, as it affects molecular properties and biological activity.
Constitutional Isomers: Same molecular formula, different connectivity of atoms.
Stereoisomers: Same molecular formula and connectivity, but different spatial arrangement.
Constitutional Isomers
Definition and Examples
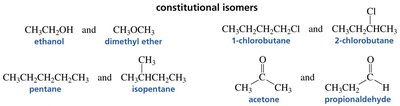
Constitutional isomers (also called structural isomers) differ in the way atoms are connected within the molecule. This results in distinct physical and chemical properties.
Key Point: Atoms are connected differently, leading to different functional groups or carbon skeletons.
Example: Ethanol (CH3CH2OH) and dimethyl ether (CH3OCH3) are constitutional isomers.
Stereoisomerism
Types of Stereoisomers
Stereoisomers have the same connectivity but differ in the spatial arrangement of atoms. The two main types are cis-trans (geometric) isomers and optical isomers (enantiomers).
Cis-Trans Isomers: Arise from restricted rotation around double bonds or rings.
Isomers with Asymmetric Centers: Arise from the presence of chiral (asymmetric) carbon atoms.
Cis-Trans (Geometric) Isomerism
Definition and Structural Basis
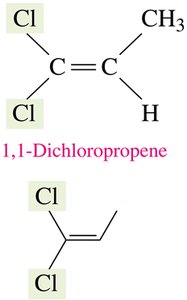
Cis-trans isomerism occurs in alkenes and cyclic compounds due to restricted rotation around double bonds or ring structures. The arrangement of substituents on either side of the bond or ring determines the isomer type.
Cis Isomer: Substituents are on the same side.
Trans Isomer: Substituents are on opposite sides.
Physical Properties: Cis and trans isomers often have different boiling points, melting points, and dipole moments.
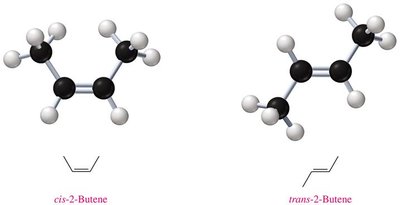
Requirements for Cis-Trans Isomerism
Cis-trans isomerism is only possible when each carbon of the double bond has two different groups attached. If both groups are identical, isomerism does not occur.
Example: 1-butene and 2-methylpropene do not exhibit cis-trans isomerism due to identical groups.
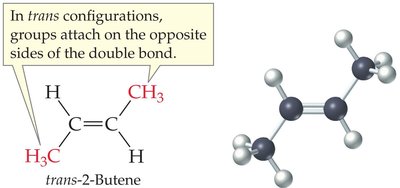
Drawing and Naming Cis-Trans Isomers
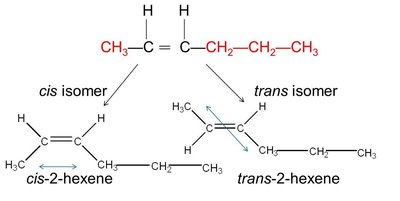
The prefix 'cis' or 'trans' is used in the compound's name to indicate the arrangement. For example, cis-2-hexene and trans-2-hexene.
Example: cis-2-hexene: Both hydrogens are on the same side; trans-2-hexene: Hydrogens are on opposite sides.
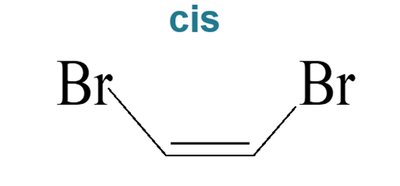
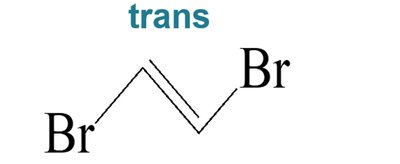
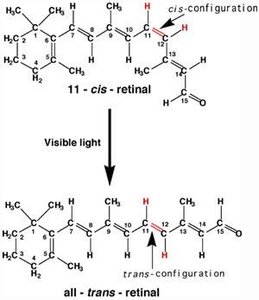
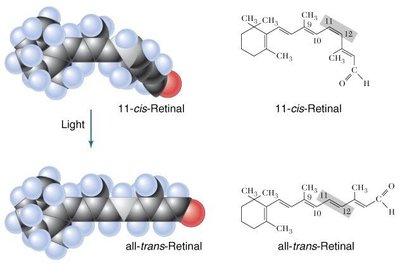
Biological Relevance: Cis-Trans Isomerism in Vision
Cis-trans isomerism is crucial in biological systems. For example, the conversion of 11-cis-retinal to all-trans-retinal in the eye is essential for vision.
Rhodopsin: Contains 11-cis-retinal; upon absorption of light, it isomerizes to all-trans-retinal, triggering a signal to the brain.
Optical Isomerism (Enantiomerism)
Chirality and Chiral Centers
Optical isomerism arises when molecules have chiral centers—carbon atoms bonded to four different groups. These molecules exist as non-superimposable mirror images called enantiomers.
Chiral Center: A tetrahedral carbon with four distinct substituents.
Enantiomers: Mirror-image isomers that cannot be superimposed.
Biological Importance: Many biomolecules, such as amino acids and sugars, are chiral.

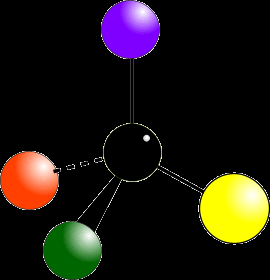
Chiral and Achiral Objects
Chirality is illustrated by objects like hands or gloves, which are mirror images but not superimposable. Achiral objects, such as a glass, are identical to their mirror images.
Chiral: No plane of symmetry; mirror images are not superimposable.
Achiral: Has a plane of symmetry; mirror images are superimposable.


Optical Activity
Chiral compounds rotate plane-polarized light, a property measured using a polarimeter. Enantiomers rotate light in equal magnitude but opposite directions.
Dextrorotatory (+): Rotates light clockwise.
Levorotatory (–): Rotates light counterclockwise.
Racemic Mixture: A 50:50 mixture of enantiomers; optically inactive.
Enantiomers in Biological Systems
Enantiomers often have different biological effects. For example, L-dopa is effective in treating Parkinson's disease, while D-dopa is not.
Example: Carvone enantiomers have different smells: spearmint vs. caraway.
Fischer Projections and Naming Enantiomers
Fischer projections are used to represent chiral molecules in two dimensions. The Cahn-Ingold-Prelog system assigns R or S configuration based on the priority of groups attached to the chiral center.
R Configuration: Clockwise arrangement of priorities.
S Configuration: Counterclockwise arrangement.
Rule: Assign priorities by atomic number; orient the lowest priority group away from the viewer.
Multiple Chiral Centers: Diastereomers and Meso Compounds
Compounds with more than one chiral center can have enantiomers, diastereomers, or meso compounds. The maximum number of stereoisomers is given by , where n is the number of chiral centers.
Enantiomers: Opposite configuration at all chiral centers.
Diastereomers: Opposite configuration at some, but not all, chiral centers.
Meso Compounds: Have chiral centers but are achiral due to a plane of symmetry.
Summary Table: Types of Isomers
Type | Definition | Example |
|---|---|---|
Constitutional Isomers | Different connectivity | Ethanol vs. Dimethyl Ether |
Cis-Trans Isomers | Different spatial arrangement around double bond/ring | cis-2-Butene vs. trans-2-Butene |
Enantiomers | Non-superimposable mirror images | Lactic acid enantiomers |
Diastereomers | Non-mirror image stereoisomers | Threonine stereoisomers |
Meso Compounds | Achiral with chiral centers | Tartaric acid meso form |
Additional info: The notes above expand on the original content by providing definitions, examples, and biological relevance, ensuring completeness and academic quality for biochemistry students.
