 Back
BackNucleosides, Nucleotides, and Polynucleotides: Structure, Properties, and Analogues
Study Guide - Smart Notes

Nucleic Acids: Structure and Components
Introduction to Nucleic Acids
Nucleic acids are essential biopolymers found in all living cells, responsible for the storage and transmission of genetic information. The two primary types are deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). Both are composed of repeating units called nucleotides, which consist of a nitrogenous base, a pentose sugar, and a phosphate group.
Nitrogenous Bases
Pyrimidine and Purine Bases
Nitrogenous bases are heterocyclic aromatic amines, classified as either pyrimidines or purines. Pyrimidines are single-ring structures (cytosine, thymine, uracil), while purines are double-ring structures (adenine, guanine). - Pyrimidines: Thymine (T), Cytosine (C), Uracil (U) - Purines: Adenine (A), Guanine (G) - Minor bases: Modified forms such as 5-methylcytosine, 5-bromouracil, N6-methyladenosine
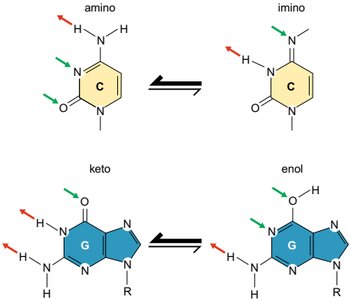
Tautomerism in Nitrogenous Bases
The aromatic nature of these bases allows for tautomeric shifts between amino/imino and keto/enol forms, affecting hydrogen bonding and base pairing. These shifts can lead to mutations during DNA synthesis.
Amino-imino tautomerism: Cytosine can shift between amino and imino forms.
Keto-enol tautomerism: Guanine and thymine can shift between keto and enol forms.
UV Absorption
Due to their aromaticity, nitrogenous bases absorb ultraviolet light at 260 nm, a property used in nucleic acid quantification.
Pentose Sugars in Nucleic Acids
Structure and Types
The pentose sugar in nucleic acids is always a five-carbon furanose. - β-D-ribose: Found in RNA - β-D-2-deoxyribose: Found in DNA (lacks an OH group at the 2' position) - The sugar atoms are numbered with a prime (') to distinguish from base atoms. 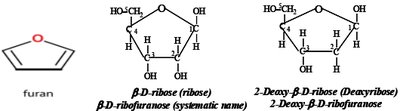
Nucleosides
Formation and Structure
Nucleosides are formed when a nitrogenous base is linked to the anomeric carbon (C1') of a pentose sugar via a β-N-glycosidic bond. - Purine nucleosides: Link at N9 of the base - Pyrimidine nucleosides: Link at N1 of the base
Classification and Nomenclature
Nucleosides are classified by their base and sugar: - Ribonucleosides: Contain ribose (e.g., adenosine, guanosine, cytidine, uridine) - Deoxyribonucleosides: Contain deoxyribose (e.g., deoxyadenosine, deoxyguanosine, deoxycytidine, deoxythymidine)
Base | Nucleoside | Common Name |
|---|---|---|
Adenine | Adenine ribonucleoside | Adenosine |
Guanine | Guanine ribonucleoside | Guanosine |
Uracil | Uracil ribonucleoside | Uridine |
Cytosine | Cytosine ribonucleoside | Cytidine |
Adenine | Adenine deoxyribonucleoside | Deoxyadenosine |
Guanine | Guanine deoxyribonucleoside | Deoxyguanosine |
Thymine | Thymine deoxyribonucleoside | Deoxythymidine |
Cytosine | Cytosine deoxyribonucleoside | Deoxycytidine |
Syn and Anti Conformations
Nucleosides can adopt syn or anti conformations based on the rotation around the glycosidic bond. - Anti conformation: More stable, base points away from sugar - Syn conformation: Less stable, base is above the sugar
Nucleotides
Structure and Ionization
Nucleotides are phosphoric acid esters of nucleosides, consisting of a nitrogenous base, pentose sugar, and phosphate group. The phosphate group can exist in different ionization states depending on pH: 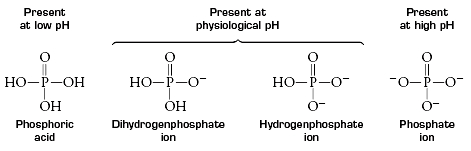
Naming and Structure
Nucleotides are named as phosphate substitutes of nucleosides, e.g., adenosine 5'-monophosphate (AMP), cytidine 5'-monophosphate (CMP). 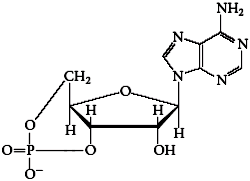
Cyclic Nucleotides
Cyclic nucleotides (e.g., cAMP, cGMP) are formed when phosphate forms two ester bonds with the sugar, playing key roles in cell signaling.
Nucleotide Triphosphates
Addition of more phosphate groups forms diphosphates (ADP) and triphosphates (ATP), with phosphoanhydride bonds between phosphates.
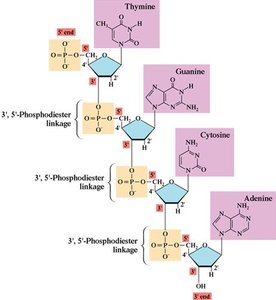
Phosphodiester Bonds, Oligonucleotides, and Polynucleotides
Phosphodiester Linkages
Nucleotides are joined by phosphodiester bonds between the 3' hydroxyl of one sugar and the 5' phosphate of the next, forming the backbone of nucleic acids. 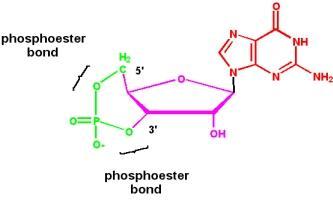
Formation of Dinucleotides and Polynucleotides
The condensation reaction between nucleotides releases pyrophosphate, forming oligonucleotides (short chains) and polynucleotides (long chains). 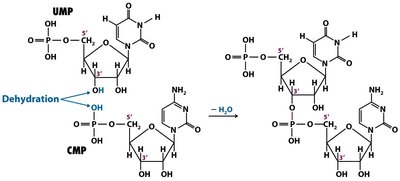
Polarity and Directionality
Nucleic acid chains have inherent directionality, with a 5' end (phosphate) and a 3' end (hydroxyl). Sequences are written from 5' to 3'. 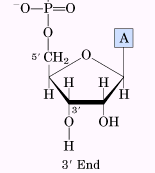
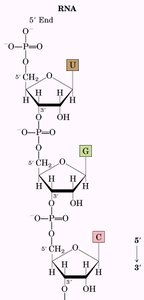
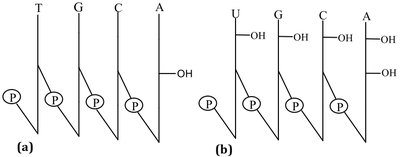
Primary Structure and Shorthand Notation
The primary structure of nucleic acids is the sequence of bases. Shorthand notations use single-letter abbreviations and symbols for phosphate and sugar. 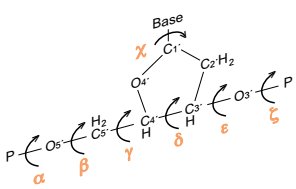
Nucleoside and Nucleotide Analogues
Antimetabolites and Therapeutic Applications
Nucleoside and nucleotide analogues are modified compounds used as antimetabolites in antiviral and anticancer therapies. They mimic natural nucleotides and interfere with nucleic acid metabolism. 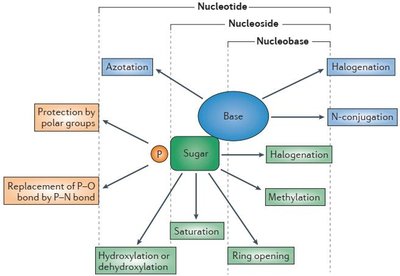
Examples of Therapeutically Active Analogues
Modifications can occur in the sugar, base, or phosphate moieties, resulting in compounds like Zidovudine (AZT), Lamivudine (3TC), Acyclovir, Ribavirin, and Sofosbuvir. 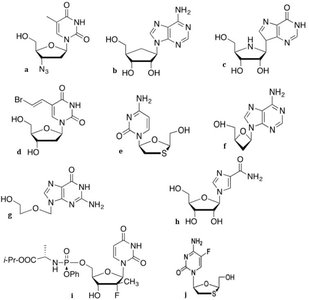
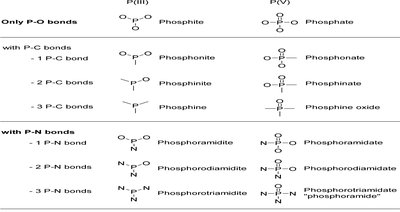
Phosphate Moiety Modifications
Pronucleotides are nucleoside monophosphate prodrugs with masked phosphate groups to enhance cellular uptake and activity.
P(III) Moieties | P(V) Moieties |
|---|---|
Phosphite | Phosphate |
Phosphonite | Phosphonate |
Phosphine | Phosphinate |
Phosphoramidite | Phosphoramidate |
Phosphorodiamidite | Phosphorodiamidate |
Phosphorotriamidite | Phosphorotriamidate |
L-Nucleoside Analogues
L-nucleosides are stereoisomers of natural D-nucleosides, recognized by viral but not mammalian enzymes, offering greater metabolic stability and reduced toxicity. Example: Emtricitabine (FTC) is an L-nucleoside used in HIV treatment.
Summary
Nucleic acids are complex biopolymers built from nucleotides, each comprising a nitrogenous base, pentose sugar, and phosphate group. Their structure, properties, and modifications are fundamental to biochemistry, genetics, and therapeutic drug design. Key concepts: - Nitrogenous base tautomerism affects base pairing and mutation rates - Pentose sugars define DNA and RNA - Nucleosides and nucleotides are named based on their base and sugar - Phosphodiester bonds create the backbone and directionality of nucleic acids - Analogues are crucial in medicine for antiviral and anticancer therapies
