 Back
BackNucleotides and Nucleic Acids: Structure, Function, and Biological Roles
Study Guide - Smart Notes

Functions and Biological Roles of Nucleotides and Nucleic Acids
Overview of Nucleic Acids
Nucleic acids are essential biopolymers responsible for the storage, transmission, and expression of genetic information. They are composed of nucleotide monomers and play central roles in cellular metabolism, gene regulation, and protein synthesis.
DNA (Deoxyribonucleic Acid): Stores genetic information in all living organisms.
RNA (Ribonucleic Acid): Transmits genetic information (mRNA), processes genetic information (ribozymes), and is involved in protein synthesis (tRNA and rRNA).
Nucleotides (Monomeric Form): Serve as energy carriers (e.g., ATP), enzyme cofactors (e.g., NAD+), and signaling molecules (e.g., cAMP).
Structure of Nucleotides and Nucleosides
Basic Components
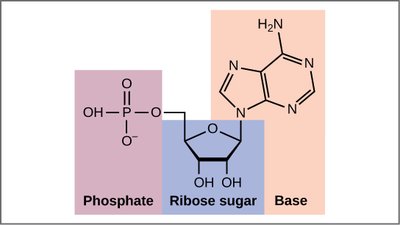
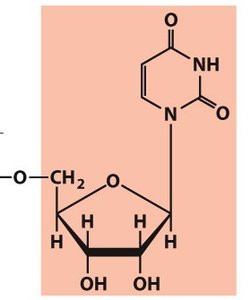
A nucleotide consists of three fundamental components: a nitrogenous base, a pentose sugar, and one or more phosphate groups. Nucleosides are similar but lack the phosphate group.
Nucleotide: Nitrogenous base + Pentose sugar + Phosphate group(s)
Nucleoside: Nitrogenous base + Pentose sugar
Numbering and Linkages
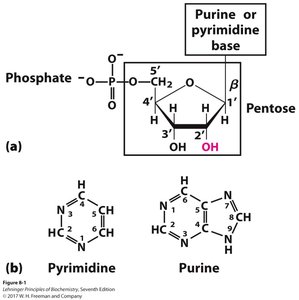
Carbons in the pentose sugar are numbered with primes (1', 2', 3', etc.) to distinguish them from atoms in the base.
The phosphate group is typically attached to the 5' carbon of the sugar, while the base is attached to the 1' carbon.
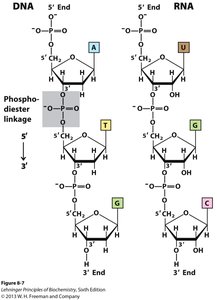
Nucleotides are linked via phosphodiester bonds between the 3' hydroxyl of one sugar and the 5' phosphate of the next.
Pentose Sugars in Nucleic Acids
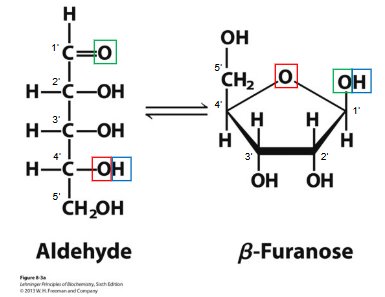
Ribose and Deoxyribose
The pentose sugar component differs between DNA and RNA, influencing their chemical properties and biological functions.
Ribose (RNA): Contains a hydroxyl group at the 2' position, making RNA more reactive and less stable.
2'-Deoxyribose (DNA): Lacks the 2' hydroxyl group, increasing DNA's stability for long-term genetic storage.
Nitrogenous Bases
Classification and Properties
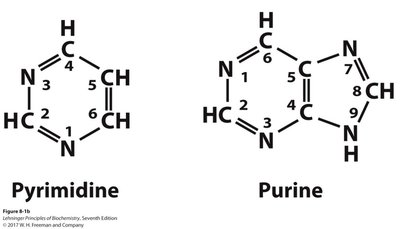
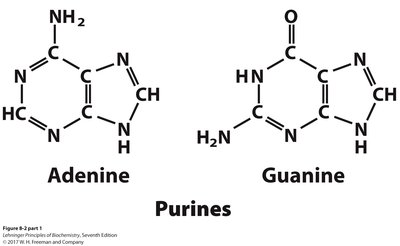
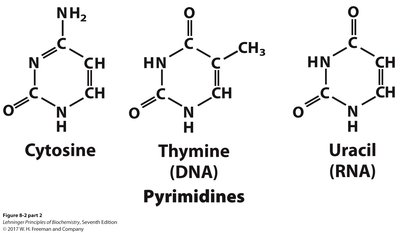
Nitrogenous bases are aromatic, heterocyclic molecules classified as purines or pyrimidines. They are planar, absorb UV light, and participate in hydrogen bonding.
Pyrimidines: Cytosine, Thymine (DNA), Uracil (RNA)
Purines: Adenine, Guanine (both DNA and RNA)
Chemical Bonds in Nucleic Acids
Types of Bonds and Their Functions
Phosphodiester Bonds: Covalent bonds forming the sugar-phosphate backbone, providing stability and directionality (5' → 3').
β-N-Glycosidic Bond: Covalent bond linking the base to the sugar (N1 in pyrimidines, N9 in purines).
Hydrogen Bonds: Non-covalent bonds between complementary bases (A=T/U, G≡C), stabilizing double-stranded structures.
Base Stacking Interactions: Van der Waals and hydrophobic forces between adjacent bases, enhancing stability.
Ionic Interactions: Electrostatic interactions between phosphate groups and metal ions or proteins, aiding DNA packaging.
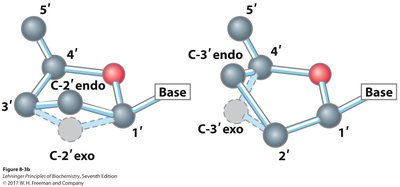
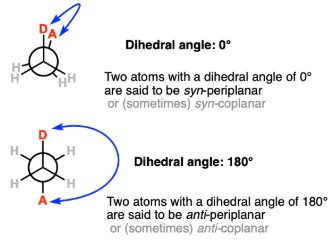
Conformation Around the N-Glycosidic Bond
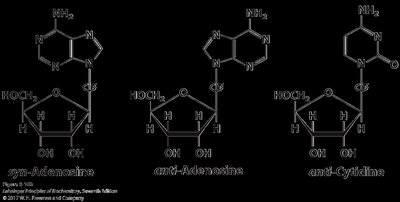
Syn and Anti Conformations
The orientation of the base relative to the sugar ring (syn or anti) affects nucleic acid structure and function. The anti conformation is predominant in DNA and RNA, allowing proper base pairing and stacking.
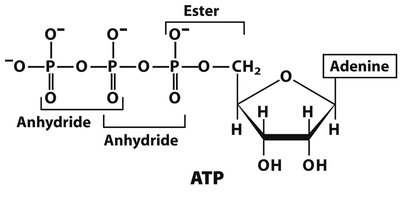
Phosphate Groups and High-Energy Bonds
Phosphoanhydride Bonds in ATP
Phosphate groups are negatively charged at neutral pH and are typically attached to the 5' position of the sugar. Nucleotides like ATP contain high-energy phosphoanhydride bonds, which release energy upon hydrolysis, fueling cellular processes.
ATP Hydrolysis:
Standard free energy change:
Nomenclature of Nucleotides
Ribonucleotides and Deoxyribonucleotides
Nucleotide names are based on their nitrogenous base, sugar type, and number of phosphate groups. Deoxyribonucleotides are distinguished by the prefix 'deoxy-' and a lowercase 'd' in abbreviations.


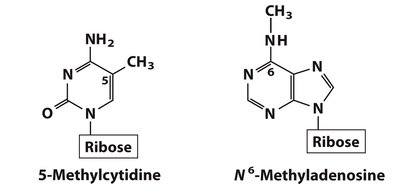
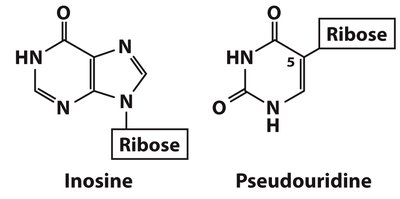
Minor Nucleosides and Modified Bases
DNA and RNA Modifications
Minor nucleosides are modified after nucleic acid synthesis and play roles in gene regulation, DNA protection, and repair. Examples include 5-methylcytidine (epigenetic regulation) and N6-methyladenosine (bacterial gene regulation).
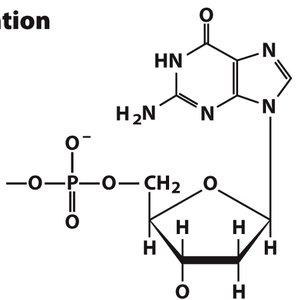
Alternative Phosphate Positions and Regulatory Nucleotides
cAMP and Its Formation
Phosphorylation can occur at different positions on the sugar, as in cyclic AMP (cAMP), a regulatory nucleotide formed from ATP by adenylyl cyclase. cAMP is crucial in signal transduction pathways.
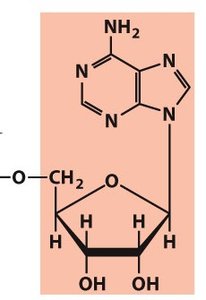
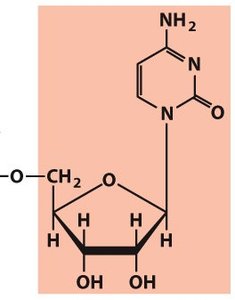
Polynucleotides: DNA and RNA Structure
Backbone and Directionality
Polynucleotides are linear polymers of nucleotides linked by phosphodiester bonds. The backbone is negatively charged and exhibits directionality (5' to 3'). DNA is more stable than RNA due to the absence of the 2' hydroxyl group.
Primary and Secondary Structure of DNA
Base Pairing and Double Helix
DNA's primary structure is its nucleotide sequence. The secondary structure is the double helix, stabilized by complementary base pairing (A=T, G≡C) and base stacking. The two strands are antiparallel and complementary.
Historical Discoveries and the Double Helix Model
Key Contributors and Experimental Evidence
The structure of DNA was elucidated through the work of Miescher, Levene, Franklin, Wilkins, Watson, and Crick. X-ray diffraction data and chemical analysis led to the double helix model, with major and minor grooves and specific base pairing.
DNA Denaturation and Renaturation
Thermal and Chemical Denaturation
Denaturation disrupts the secondary structure (hydrogen bonds and base stacking) but leaves the primary structure (covalent bonds) intact. The melting temperature (Tm) depends on GC content, DNA length, and ionic strength. Renaturation (annealing) is possible under suitable conditions.
RNA Structure and Function
Types of RNA and Their Roles
RNA molecules are single-stranded but can form complex secondary structures (hairpins, bulges, internal loops, pseudoknots) through intramolecular base pairing. Types of RNA include:
mRNA (Messenger RNA): Carries genetic code from DNA to ribosomes.
tRNA (Transfer RNA): Brings amino acids to ribosomes during translation.
rRNA (Ribosomal RNA): Structural and catalytic component of ribosomes.
miRNA (Micro-RNA): Regulates gene expression post-transcriptionally.
Summary Table: Key Bonds in DNA and RNA
Bond Type | DNA | RNA | Function |
|---|---|---|---|
Phosphodiester (Covalent) | Present | Present | Forms sugar-phosphate backbone |
β-N-Glycosidic (Covalent) | Present | Present | Links base to sugar |
Hydrogen Bonds (Non-covalent) | Between strands | In secondary structures | Stabilizes base pairing |
Base Stacking (Van der Waals) | Present | Present (weaker) | Structural stability |
Ionic Interactions | Present | Present | Neutralizes backbone charge |
Conclusion
Nucleotides and nucleic acids are fundamental to life, serving as the blueprint for genetic information, mediators of cellular energy, and regulators of gene expression. Their diverse structures and chemical properties underpin the complexity and adaptability of biological systems.
