 Back
BackOrganic Chemistry Foundations for Biochemistry: Carbon, Functional Groups, and Molecular Properties
Study Guide - Smart Notes

Organic Chemistry in Biochemistry
Importance of Carbon in Biology
Organic chemistry is the study of carbon-containing compounds, which form the structural basis of all biomolecules. Carbon's unique bonding capabilities allow it to create a vast array of molecular frameworks essential for life, including carbohydrates, fats, proteins, and nucleic acids.
Carbon is the most abundant element in biological organisms.
It forms the backbone of organic molecules found in cells.
Consumer products such as plastics, medicines, and textiles are also based on organic chemistry.

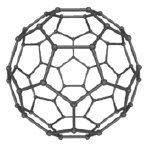
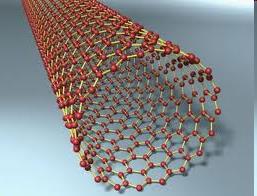
Allotropes of Carbon
Carbon exists in several forms (allotropes), each with distinct physical properties. These include diamond, graphite, buckyballs (fullerenes), and carbon nanotubes.
Diamond: Hard, transparent, and used in jewelry and cutting tools.
Graphite: Soft, black, and used in pencils and as a lubricant.
Buckyballs (C60): Spherical molecules with unique chemical properties.
Carbon nanotubes: Cylindrical structures with remarkable strength and conductivity.


Atomic Structure and Bonding of Carbon
Electronic Configuration and Bonding
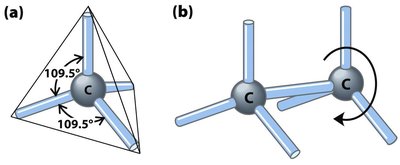
Carbon is the sixth element in the periodic table, with an atomic number of 6 and a mass number of 12. Its ground state electronic configuration is 1s2 2s2 2p2. Carbon follows the octet rule, forming four covalent bonds to achieve a stable electron configuration.
Carbon shares electrons rather than easily accepting or donating them.
This sharing leads to covalent bonds and allows for diverse molecular structures.

Bonding Combinations
Carbon can form single, double, and triple bonds, resulting in various bonding combinations:
Four single bonds
One double and two single bonds
Two double bonds
One single and one triple bond
Carbon Geometries and Molecular Diversity
Versatility of Carbon Frameworks
Carbon's ability to form bonds with itself leads to straight chains, branched chains, and rings. This versatility is the foundation for the diversity of organic molecules.
Even with only single bonds, a 20-carbon network can form over 360,000 different compounds.
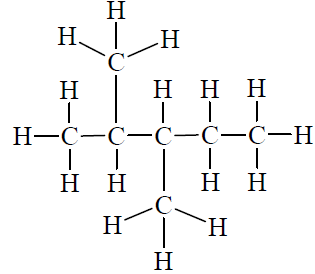
Alkanes and Skeletal Formulas
Alkanes are hydrocarbons containing only single bonds. Skeletal formulas are used to represent these compounds, omitting carbon symbols and showing bonds as lines.
Each corner or end of a line represents a carbon atom.
Isomerism and Chirality
Isomerisation
Isomers are molecules with the same atoms and bonds but different arrangements. Their properties can vary greatly, especially in biological contexts.
Structural isomers: Differ in connectivity of atoms.
Stereoisomers: Same connectivity, different spatial arrangement.
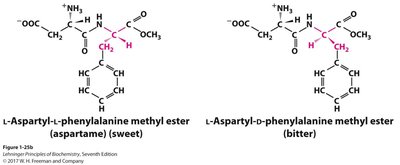
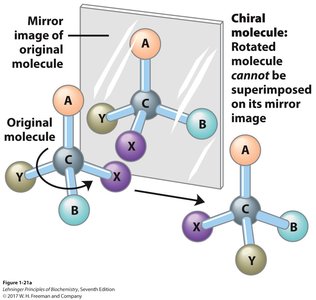
Chirality
A chiral carbon has four different groups attached, resulting in non-superimposable mirror images (enantiomers). Chirality is crucial in biochemistry, as enantiomers can have very different biological effects.
Chiral molecules participate in similar chemical reactions but may behave differently in biological systems.
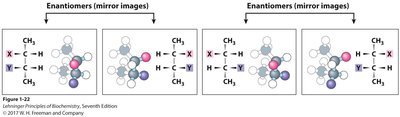
Enantiomers and Diastereomers
Enantiomers are mirror images, while diastereomers are not. The number of possible stereoisomers depends on the number of chiral centers: a molecule with n chiral centers has 2n stereoisomers.
Enantiomers differ at all chiral centers.
Diastereomers differ at some, but not all, chiral centers.
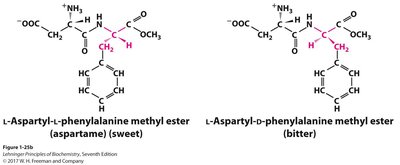
Geometric Isomerism
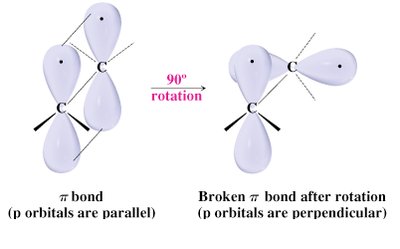
Bond Rotation and π Bonds
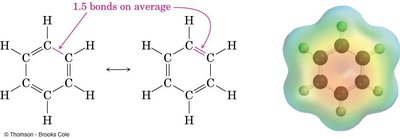
Rotation around C=C double bonds is restricted due to the π bond, which must be broken for rotation to occur. This restriction leads to geometric isomerism.
Single bond rotational barrier: 12 kJ/mol
Double bond rotational barrier: 260 kJ/mol
Geometric Isomers of Alkenes
Alkenes can have geometric isomers (cis/trans) when each carbon in the double bond is attached to two different groups. These isomers have the same molecular formula and connectivity but differ in spatial arrangement.
Cis isomer: Functional groups on the same side of the double bond.
Trans isomer: Functional groups on opposite sides.
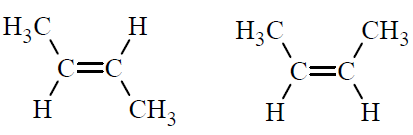
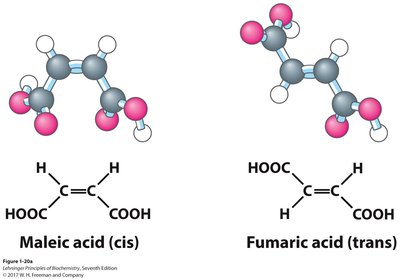
Biological Relevance of Geometric Isomers
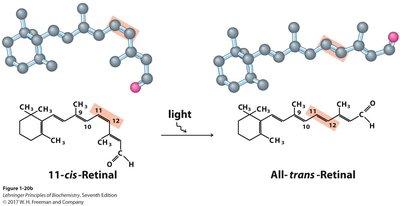
Geometric isomerism is important in biological systems, such as vision, where light converts 11-cis-retinal to all-trans-retinal, triggering a nerve impulse.
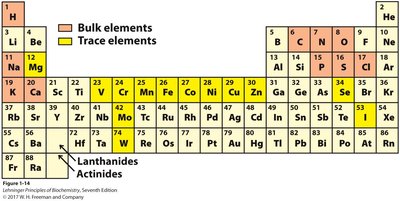
Elements Essential for Life
Bulk and Trace Elements
Besides carbon, elements such as hydrogen, oxygen, nitrogen, phosphorus, and sulfur are common in biomolecules. Metal ions (e.g., K+, Na+, Ca2+, Mg2+, Zn2+, Fe2+) play important roles in metabolism.
Polarity and Electronegativity
Polarity and Physical Properties
Physical properties such as solubility, melting point, and boiling point depend on intermolecular forces, which are influenced by molecular polarity.
Polar molecules: Experience strong dipole-dipole interactions and may form hydrogen bonds.
Non-polar molecules: Held together by weak dispersion forces.
Biomolecules often have both polar and non-polar components.
Electronegativity
Electronegativity is the ability of an atom to attract electrons. Polarity arises from bonds between atoms with different electronegativities.
Low electronegativity: C, H
High electronegativity: O, N, S, P, Cl
More electronegative atoms withdraw electrons, creating partial charges (δ+, δ–).
Functional Groups in Biomolecules
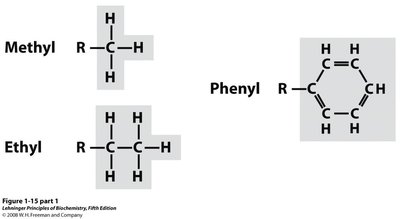
Non-Polar Groups: Hydrocarbons
Non-polar groups are composed entirely of carbon and hydrogen, such as methyl, ethyl, propyl, and butyl. Aromatic rings (phenyl groups) are also important hydrocarbon structures.
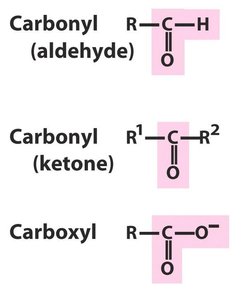
Polar Groups Containing Oxygen
Polar functional groups containing oxygen include hydroxyl (alcohol), carbonyl (aldehyde and ketone), carboxyl, and ester groups. These groups impart polarity and reactivity to biomolecules.
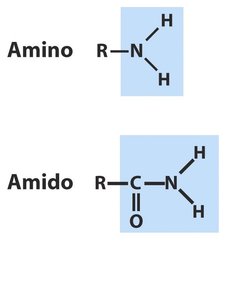
Polar Groups Containing Nitrogen
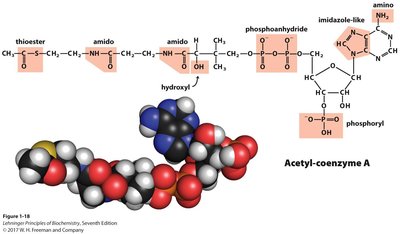
Nitrogen-containing groups include amines and amides, which are key components of proteins and nucleic acids.
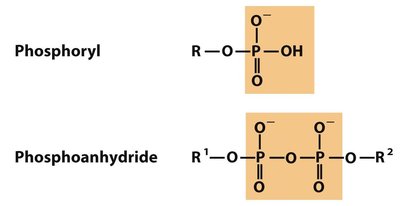
Polar Groups Containing Phosphorus
Phosphoryl and phosphoanhydride groups are important in energy transfer and nucleic acid structure.
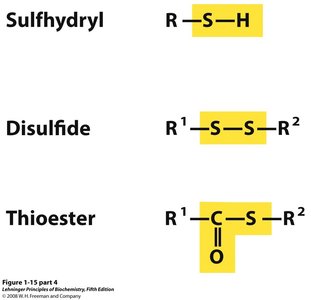
Polar Groups Containing Sulfur
Sulfur-containing groups include sulfhydryl, disulfide, and thioester, which are important in protein structure and enzyme function.
Biomolecule Functional Groups and Their Roles
Functional Groups in Biomolecules
Biomolecules often contain multiple functional groups, which determine their chemical and physical properties.
Sugars: Carbonyls and hydroxyls
DNA: Sugars (carbonyls/hydroxyls), phosphodiesters
Fats/oils: Hydrocarbons, carboxyls, esters
Proteins: Amines and amides
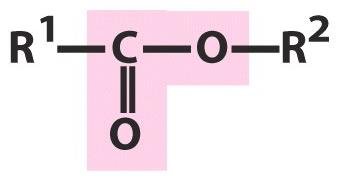
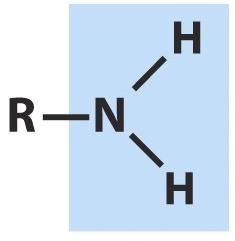
Alcohols, Aldehydes, Ketones, Carboxylic Acids, Esters, Amines, and Amides
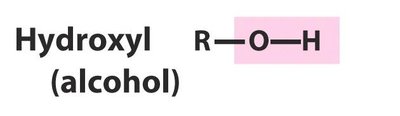
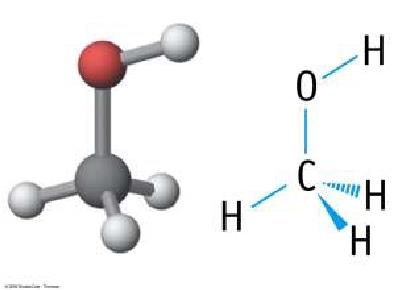
Alcohols (–OH)
Alcohols contain a hydroxyl group attached to a saturated carbon. They are named with the suffix 'ol' (e.g., methanol, ethanol).
Smaller alcohols are more water-like; larger alcohols are more hydrocarbon-like.
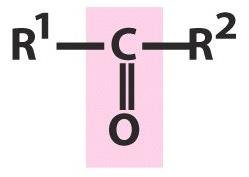
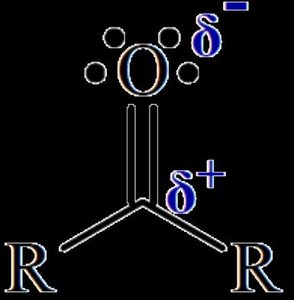
Carbonyl Compounds
Carbonyl groups (C=O) are present in aldehydes, ketones, carboxylic acids, and esters. The polarity of the C=O bond influences their properties.
Aldehydes
Aldehydes have an alkyl group and a hydrogen bonded to a carbonyl group. They are named with the suffix 'al' (e.g., methanal, ethanal).
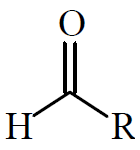
Ketones
Ketones have two alkyl groups bonded to a carbonyl group. They are named with the suffix 'one' (e.g., propanone/acetone).
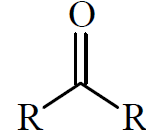
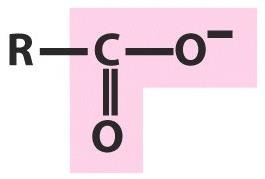
Carboxylic Acids
Carboxylic acids contain both a hydroxyl and a carbonyl group attached to the same carbon. They are named with the suffix 'oic acid' (e.g., methanoic acid, butanoic acid).
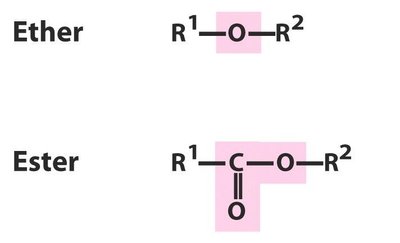
Esters
Esters are formed by the reaction of an alcohol with a carboxylic acid (condensation reaction). They are named with the suffix 'oate' or 'ate' (e.g., methyl butanoate).
Amines and Amides
Amines are organic derivatives of ammonia, classified as primary, secondary, or tertiary based on the number of alkyl groups. Amides are formed by condensation reactions between amines and carboxylic acids, linking amino acids in proteins.
Summary Table: Functional Groups and Their Roles
Functional Group | Structure | Role in Biomolecules |
|---|---|---|
Hydroxyl (Alcohol) | R–OH | Carbohydrates, solubility |
Carbonyl (Aldehyde/Ketone) | R–C=O–R | Sugars, reactivity |
Carboxyl | R–COOH | Fatty acids, amino acids |
Ester | R–COOR | Fats, oils |
Amino | R–NH2 | Amino acids, proteins |
Amido | R–CONH2 | Protein linkages |
Phosphoryl | R–PO4 | DNA, energy transfer |
Sulfhydryl | R–SH | Protein structure |
Key Concepts and Applications
Carbon's versatility enables the diversity of organic molecules.
Isomerism (structural, geometric, and chiral) is crucial for biological function.
Polarity and electronegativity affect solubility and intermolecular interactions.
Functional groups define the chemical properties and biological roles of biomolecules.
Condensation and hydrolysis reactions are fundamental in biomolecule synthesis and breakdown.
Additional info: Expanded explanations and context were added to ensure completeness and clarity for biochemistry students.
