 Back
BackProtein Structure and Function: Comprehensive Study Notes
Study Guide - Smart Notes

Proteins: Structure and Function
Overview of Proteins
Proteins are essential biological macromolecules that serve as enzymes, hormones, receptors, channels, transporters, and antibodies. They provide structural support both inside and outside cells. Proteins are composed of 20 different amino acids, linked together in polymers, and classified according to the chemical properties of their side chains (R groups). The unique composition and sequence of amino acids in a polypeptide chain determine each protein's specific function in the cell.
Key Point 1: Proteins are exclusively composed of L-amino acids in living organisms.
Key Point 2: The sequence and chemical nature of amino acids dictate protein structure and function.
Example: Enzymes catalyze biochemical reactions, while antibodies recognize foreign molecules.
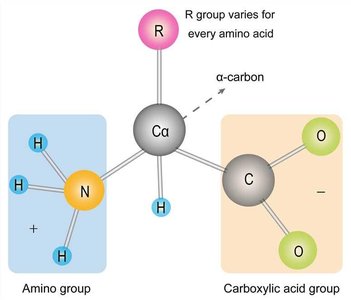
Amino Acids
Structure and Nomenclature
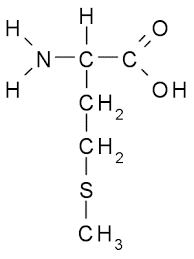
Amino acids are organic compounds containing an amino group, a carboxylic acid group, a hydrogen atom, and a unique side chain (R group) attached to a central α-carbon. The physical and chemical properties of amino acids are determined by their R groups. All amino acids except glycine have at least one asymmetrical (chiral) carbon atom.
Key Point 1: The α-carbon is the central carbon atom to which all groups are attached.
Key Point 2: Glycine is the only amino acid without optical activity due to its symmetric structure.
Example: The R group distinguishes each amino acid and determines its role in protein structure.
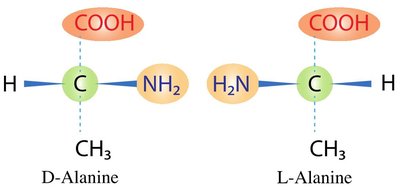
Classification of Amino Acids
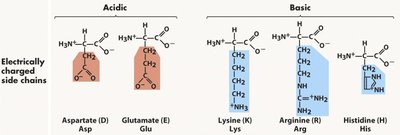
Amino acids are classified based on the properties of their side chains: nonpolar, polar, acidic, and basic. This classification affects their location in proteins and their interactions with water.
Key Point 1: Nonpolar amino acids have aliphatic or aromatic side chains and are typically found in the interior of globular proteins, away from water.
Key Point 2: Polar amino acids can form hydrogen bonds with water and are often found on the protein surface.
Key Point 3: Acidic and basic amino acids carry charges at physiological pH, contributing to protein solubility and function.
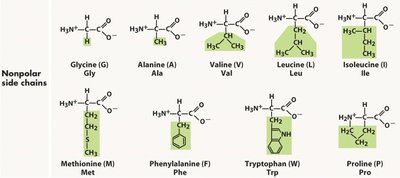
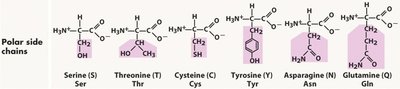
Essential Amino Acids
Essential amino acids cannot be synthesized by the human body and must be obtained from the diet. There are nine essential amino acids required for proper protein synthesis and metabolic function.
Key Point 1: Essential amino acids include histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine.
Key Point 2: Deficiency in essential amino acids can lead to impaired growth and metabolic disorders.
Protein Structure
Levels of Protein Structure
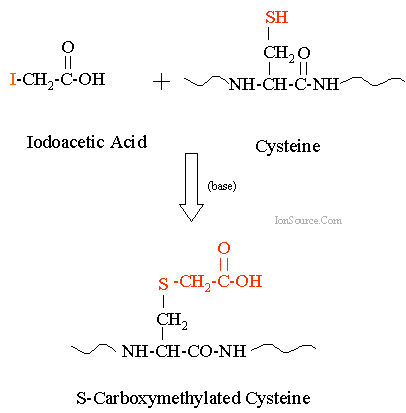
Proteins exhibit four levels of structural organization: primary, secondary, tertiary, and quaternary. Each level is defined by specific interactions and arrangements of amino acids.
Primary Structure: The linear sequence of amino acids in a polypeptide chain.
Secondary Structure: Local spatial arrangements such as α-helices and β-sheets, stabilized by hydrogen bonds.
Tertiary Structure: The overall three-dimensional shape of a single polypeptide, determined by interactions among side chains.
Quaternary Structure: The arrangement of multiple polypeptide subunits in a protein complex.
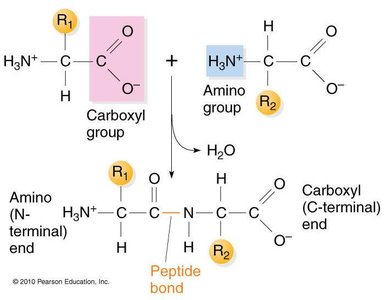
Primary Structure: Peptide Bonds
The primary structure is defined by the order in which amino acids are linked together via peptide bonds. Peptide bonds are formed between the amino group of one amino acid and the carboxyl group of another, releasing water in a condensation reaction.
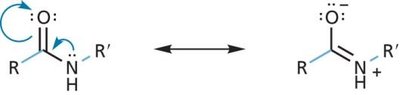
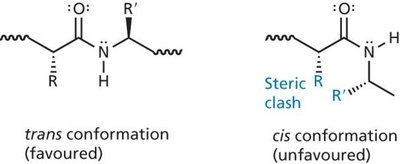
Key Point 1: Peptide bonds have partial double bond character due to resonance, restricting rotation.
Key Point 2: The polypeptide chain grows from the amino (N) terminus to the carboxyl (C) terminus.
Example: The sequence of amino acids determines the protein's unique properties and function.

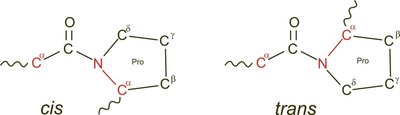
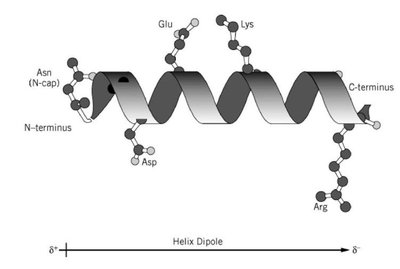
Secondary Structure: α-Helix and β-Sheet
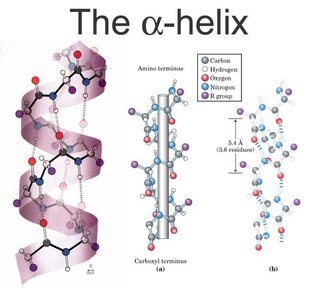
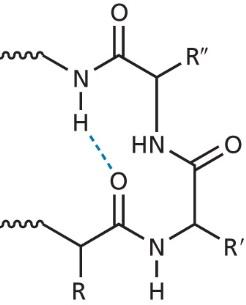
The secondary structure refers to regular, repeating patterns within the protein backbone, primarily α-helices and β-sheets. These structures are stabilized by hydrogen bonds between backbone atoms.
α-Helix: A right-handed coil stabilized by hydrogen bonds between the carbonyl oxygen and amide hydrogen four residues apart. Proline introduces kinks due to its rigid structure.
β-Sheet: Extended strands connected by hydrogen bonds, forming parallel or antiparallel arrangements. The side chains alternate above and below the sheet, minimizing steric clashes.
β-Turn: Allows the polypeptide chain to reverse direction, stabilized by hydrogen bonds, often involving glycine or proline.
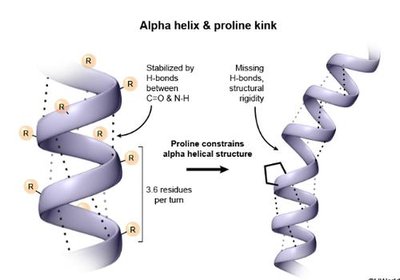
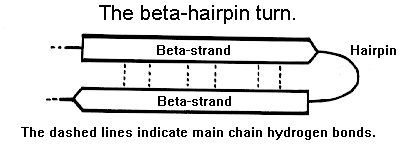
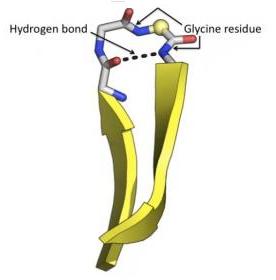
Tertiary Structure: Three-Dimensional Folding
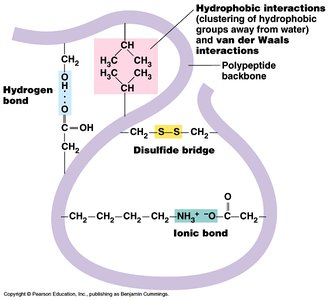
The tertiary structure is the overall three-dimensional shape of a protein, determined by interactions among side chains, including hydrogen bonds, ionic bonds, hydrophobic interactions, van der Waals forces, and covalent disulfide bonds.
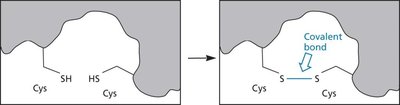
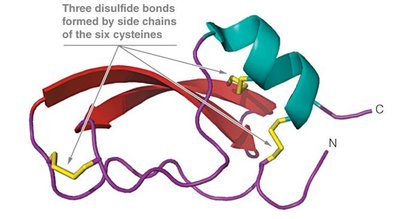
Key Point 1: Disulfide bonds between cysteine residues stabilize the folded structure.
Key Point 2: Hydrophobic side chains cluster in the protein's interior, while hydrophilic side chains interact with water on the surface.
Key Point 3: Ionic bonds (salt bridges) and hydrogen bonds contribute to stability and specificity.

Protein Denaturation and Stability
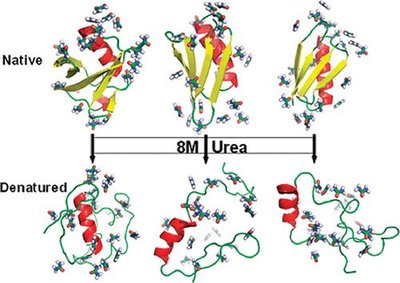
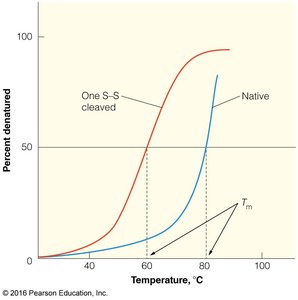
Protein denaturation involves the loss of native structure and biological activity, often caused by heat, pH extremes, or chemical agents like urea. Disulfide bonds greatly enhance protein stability, as seen in bovine pancreatic trypsin inhibitor (BPTI).
Key Point 1: Urea disrupts hydrogen bonds and weakens secondary and tertiary structure.
Key Point 2: Disulfide bonds increase the melting temperature (Tm) and resistance to denaturation.
Example: BPTI remains stable at high temperatures and acidic pH due to its disulfide bonds.
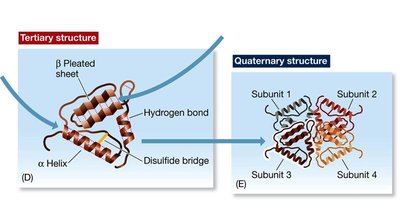
Quaternary Structure: Protein Complexes
Quaternary structure refers to the arrangement of multiple polypeptide subunits in a protein complex. Interactions between subunits include hydrophobic, van der Waals, and ionic bonds. Only proteins with multiple subunits exhibit quaternary structure.
Key Point 1: Subunits may be identical or different, and their arrangement is crucial for function.
Key Point 2: Hydrophobic regions may be exposed on the surface, promoting dimerization or oligomerization.
Example: Hemoglobin is a tetrameric protein with two α and two β subunits.
Protein Folding and Thermodynamics
Folding Pathways and Stability
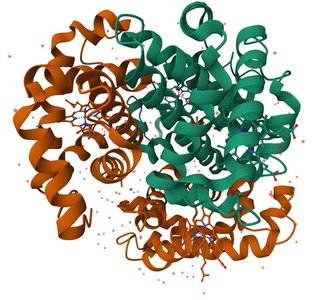
Protein folding is directed by the amino acid sequence and influenced by thermodynamic factors. The process involves a decrease in conformational entropy, enthalpic stabilization from intramolecular interactions, and the hydrophobic effect, which releases ordered water molecules.
Key Point 1: The nucleotide sequence of mRNA determines the amino acid sequence, which dictates folding.
Key Point 2: Only a single conformational form is associated with a functional protein, despite millions of possibilities.
Key Point 3: Three thermodynamic factors influence folding: enthalpic stabilization, loss of conformational entropy, and gain of solvent entropy from the hydrophobic effect.
Equation:
Amino Acid Classification | Side Chain Type | Example Amino Acids | Properties |
|---|---|---|---|
Nonpolar | Aliphatic/Aromatic | Gly, Ala, Val, Leu, Ile, Met, Phe, Trp, Pro | Hydrophobic, found in protein interior |
Polar | Hydroxyl, Amide, Sulfhydryl | Ser, Thr, Cys, Tyr, Asn, Gln | Hydrophilic, can form H-bonds |
Acidic | Carboxyl | Asp, Glu | Negatively charged at pH 7.4 |
Basic | Amino, Imidazole | Lys, Arg, His | Positively charged at pH 7.4 |
Additional info: Academic context was added to clarify the role of amino acid side chains, the importance of peptide bond resonance, and the thermodynamic principles underlying protein folding and stability.
