 Back
BackProtein Structure and Function: Hemoglobin, Myoglobin, and Immunoglobulins
Study Guide - Smart Notes

Protein Structure and Function
Overview of Protein Structure
Proteins are essential macromolecules that perform a wide variety of functions in biological systems. Their structure determines their function, and understanding the architecture of key proteins such as hemoglobin, myoglobin, and immunoglobulins is fundamental in biochemistry.
Hemoglobin and Myoglobin
Structural Comparison
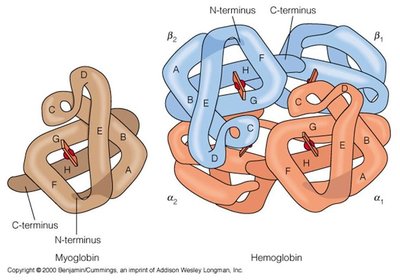
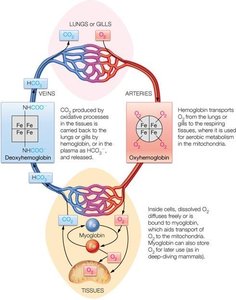
Hemoglobin and myoglobin are oxygen-binding proteins with distinct structures and physiological roles.
Myoglobin is a monomeric protein found in muscle tissue, responsible for storing oxygen.
Hemoglobin is a tetrameric protein in red blood cells, specialized for oxygen transport in the bloodstream.
Both proteins contain a heme group that binds oxygen, but hemoglobin's quaternary structure allows for cooperative binding and allosteric regulation.
Oxygen Transport and Storage
Hemoglobin and myoglobin work together to ensure efficient oxygen delivery from the lungs to tissues and storage within muscle cells.
Hemoglobin exhibits cooperative binding, meaning its affinity for oxygen increases as more oxygen molecules bind.
Myoglobin has a higher affinity for oxygen at low partial pressures, facilitating oxygen transfer from hemoglobin to muscle tissue.
Immunoglobulins (Antibodies)
Structure of Immunoglobulins
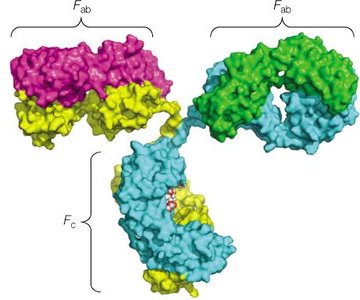
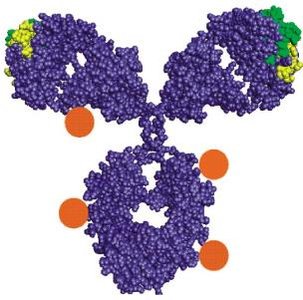
Immunoglobulins, or antibodies, are Y-shaped glycoproteins produced by B cells. They play a central role in the adaptive immune response by specifically recognizing and binding antigens.
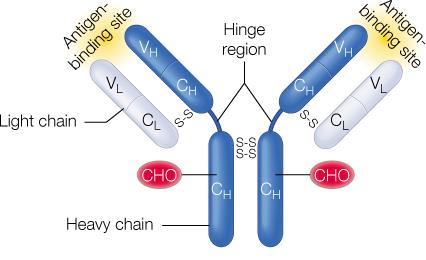
Each antibody consists of two heavy chains and two light chains, connected by disulfide bonds.
The Fab (Fragment antigen-binding) regions bind to antigens, while the Fc (Fragment crystallizable) region mediates immune effector functions.
Variable regions at the tips of the Y confer antigen specificity.
Classes of Immunoglobulins
There are five main classes of immunoglobulins, each with distinct structures and functions:
Class | Definition | Diagram |
|---|---|---|
IgM | Produced early in immune response; pentameric structure; effective in complement activation. |
|
IgG | Most abundant in circulation; monomeric; crosses placenta; triggers complement system. |
|
IgA | Found in secretions (saliva, milk); dimeric or monomeric; protects mucosal surfaces. |
|
IgE/IgD | IgE involved in allergic responses; IgD found on B cell surfaces; both are monomeric. |
|
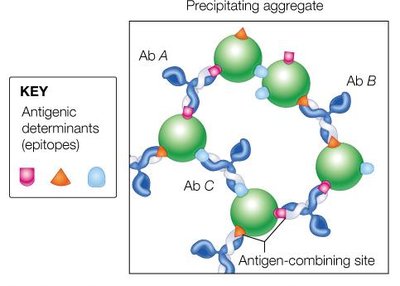
Antibody-Antigen Interaction
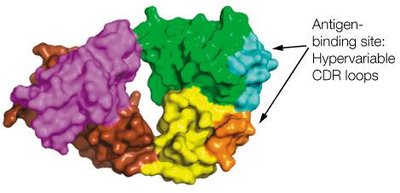
Antibodies recognize specific regions (epitopes) on antigens through their variable regions. The binding involves non-covalent interactions such as hydrogen bonds, van der Waals forces, and electrostatic interactions.
The antigen-binding site is formed by the variable domains of both heavy and light chains.
Antigen-antibody complexes can precipitate, neutralize pathogens, or mark them for destruction by other immune cells.
Immune System Overview
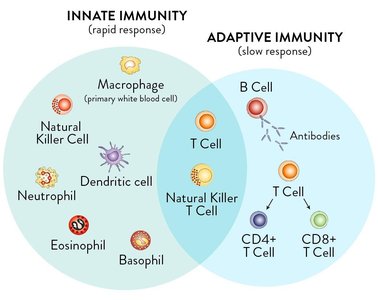
The immune system is divided into innate and adaptive branches. Antibodies are central to the adaptive immune response, which is slower but highly specific.
Innate immunity provides rapid, non-specific defense mechanisms.
Adaptive immunity involves B cells (antibody production) and T cells (cell-mediated responses).
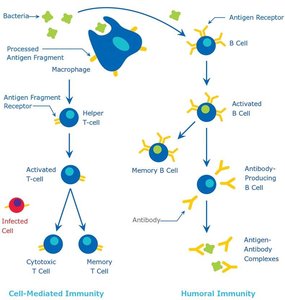
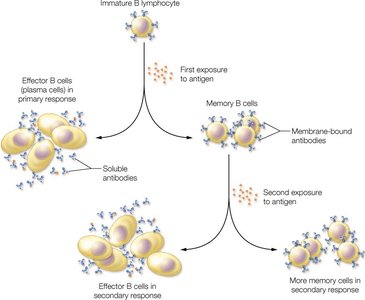
B Cell Activation and Memory
B cells differentiate into plasma cells (which secrete antibodies) and memory B cells (which provide long-term immunity) upon exposure to antigens.
Primary response: Initial exposure leads to effector and memory cell formation.
Secondary response: Subsequent exposures elicit a faster and stronger antibody response due to memory cells.
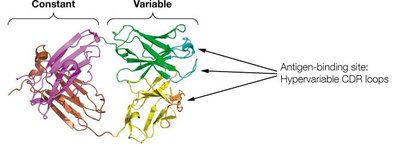
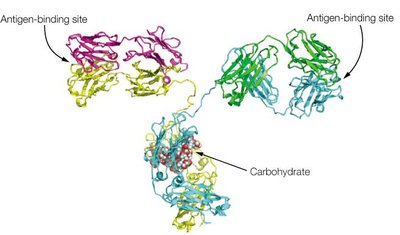
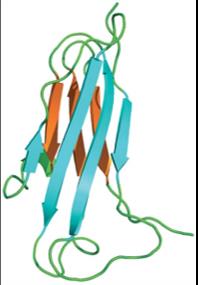
Antibody Structure: Domains and Variability
Antibody molecules are composed of constant and variable domains. The variable domains contain hypervariable regions (complementarity-determining regions, CDRs) that determine antigen specificity.
Constant domains provide structural stability and mediate effector functions.
Variable domains form the antigen-binding site and confer diversity.
Antibody Engineering and Applications
Antibodies can be engineered for therapeutic purposes, such as antibody-drug conjugates (ADCs), which deliver cytotoxic drugs specifically to target cells.
ADCs combine the specificity of antibodies with the potency of drugs, linked by a stable chemical linker.
This approach is used in targeted cancer therapies.
Summary Table: Key Protein Functions
Protein | Function | Structure |
|---|---|---|
Hemoglobin | Oxygen transport in blood | Tetramer (2α, 2β subunits) |
Myoglobin | Oxygen storage in muscle | Monomer |
Immunoglobulin (Antibody) | Antigen recognition and immune defense | Y-shaped, 2 heavy + 2 light chains |
Additional info: The notes above integrate foundational biochemistry concepts from protein structure (Ch. 5, 6, 7) and protein function (Ch. 7), with a focus on oxygen-binding proteins and immunoglobulins, as well as their roles in immunity and clinical applications.




